Abstract
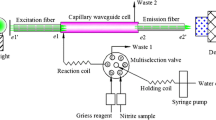
A novel small-volume fiber-optic evanescent-wave absorption sensor based on the Griess–Ilosvay reaction has been developed and evaluated for nitrite determination. The sensor was constructed by inserting a decladded optical fiber into a transparent capillary to form an annular column microchannel. The Evanescent wave (EW) field produced on the optical fiber core surface penetrated into the surrounding medium and interacted with the azo dye, which was generated by the reaction of nitrite and nitrite-sensitive reagents. The detector was designed to be parallel to the axis of the optical fiber. The defined absorbance was linear with the concentration of nitrite in the range from 0.05 to 10 mg L−1, and the detection limit was 0.02 mg L−1 (3σ) with the relative standard deviation (RSD) of 2.6% (n = 8). The present sensor was successfully used to determine nitrite in real samples of mineral water, tap water, rain water, and seawater. The results were consistent with the data obtained by standard spectrophotometric method, showing potential of the proposed sensor for practical application.

Schematic diagram of the evanescent wave in the sensor.




Similar content being viewed by others
References
Wolff IA, Wassennan AE (1972) Nitrates, nitrites, and nitrosamines. Science 177:15–19
Nelieu S, Shankar MV, Kerhoas L (2008) Phototransformation of monuron induced by nitrate and nitrite ions in water: Contribution of photonitration. J Photochem Photobiol, A 193:1–9
World Health Organization (2004) http://www.who.int/water_sanitation_health/dwq/gdwq3/en/index.html
Apyari VV, Dmitrienko SG, Ostrovskaya VM (2008) Use of polyurethane foam and 3-hydroxy-7, 8-benzo-1, 2, 3, 4-tetrahydroquinoline for determination of nitrite by diffuse reflectance spectroscopy and colorimetry. Anal Bioanal Chem 391:1977–1982
Ranjit KT, Viswanathan B (2003) Photoelectrochemical reduction of nitrite ions to ammonia on CdS photocatalysts. J Photochem Photobiol, A 154:299–302
Miró M, Cladera A, Estela JM, Cerdà V (2000) Sequential injection spectrophotometric analysis of nitrite in natural waters using an on-line solid-phase extraction and preconcentration method. Analyst 125:943–948
Nagaraja P, Kumar MSH, Rangappa KS (2001) Dapsone and iminodibenzyl as novel reagents for the spectrophotometric determination of trace amounts of nitrite in water samples. Anal Sci 17:439–442
Abbas MN, Mostafa GA (2000) Determination of traces of nitrite and nitrate in water by solid phase spectrophotometry. Anal Chim Acta 410:185–192
Zuo YG, Wang CJ, Van T (2006) Simultaneous determination of nitrite and nitrate in dew, rain, snow and lake water samples by ion-pair high-performance liquid chromatography. Talanta 70:281–285
Kodamatani H, Yamazaki S, Saito K (2009) Selective determination method for measurement of nitrite and nitrate in water samples using high-performance liquid chromatography with post-column photochemical reaction and chemiluminescence detection. J Chromatogr A 1216:3163–3167
Mubarak AT, Mohamed AA, Fawy KF (2007) A novel kinetic determination of nitrite based on the perphenazine–bromate redox reaction. Microchim Acta 157:99–105
Mao L, Yamamoto K, Zhou W, Jin L (2000) Electrochemical nitric oxide sensors based on electropolymerized film of M(salen) with central ions of Fe, Co, Cu, and Mn. Electroanalysis 12:72–77
Yang W, Bai Y, Li Y, Sun C (2005) Amperometric nitrite sensor based on hemoglobin/colloidal gold nanoparticles immobilized on a glassy carbon electrode by a titania sol–gel film. Anal Bioanal Chem 382:44–50
Ishio N, Fukushi K, Michiba K (2002) Optimum conditions for effective use of the terminating ion in transient isotachophoresis for capillary zone electrophoretic determination of nitrite and nitrate in seawater, with artificial seawater as background electrolyte. Anal Bioanal Chem 374:1165–1169
He DY, Zhang ZJ, Huang Y (2007) Chemiluminescence microflow injection analysis system on a chip for the determination of nitrite in food. Food Chem 101:667–672
Wang L, Dong L, Bian GR (2005) Using organic nanoparticle fluorescence to determine nitrite in water. Anal Bioanal Chem 382:1300–1303
Harrick NJ (1967) Internal reflection spectroscopy. Wiley, New York
Busse S, Scheumann V, Menges B, Mittler S (2002) Sensitivity studies for specific binding reactions using the biotin/streptavidin system by evanescent optical methods. Biosens Bioelectron 17:704–710
Ge Z, Brown CW, Sun L, Yang SC (1993) Fiber-optic pH sensor based on evanescent wave absorption spectroscopy. Anal Chem 65:2335–2338
Vogt F, Karlowatz M, Jakusch M, Mizaikoff B (2003) The automated sample preparation system MixMaster for investigation of volatile organic compounds with mid-infrared evanescent wave spectroscopy. Analyst 128:397–403
DeGrandpre MD, Burgess LW (1988) Long path fiber-optic sensor for evanescent field absorbance measurements. Anal Chem 60:2582–2586
Xiong Y, Xu J, Wang J, Guan Y (2009) A fiber-optic evanescent wave sensor for dissolved oxygen detection based on novel hybrid fluorinated xerogels immobilized with [Ru(bpy)3]2+. Anal Bioanal Chem 394:919–923
Kumar PS, Lee ST, Vallabhan CPG, Nampoori VPN, Radhakrishnan P (2002) Design and development of an LED based fiber optic evanescent wave sensor for simultaneous detection of chromium and nitrite traces in water. Opt Commun 214:25–30
Lee ST, George NA, Kumar PS, Radhakrishnan P, Nampoori VPN, Vallabhan CPG (2001) Chemical sensing with microbent optical fiber. Opt Lett 26:1541–1543
Saltzman BE (1954) Colorimetric microdetermination of nitrogen dioxide in atmosphere. Anal Chem 26:1949–1955
Acknowledgment
This work was supported by the Chinese Academy of Sciences, contract no. KZCX1-YW-14-3, and the Ministry of Science and Technology of China on High Tech Program, contract no. 2008AA09Z110.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiong, Y., Zhu, Dq., Duan, Cf. et al. Small-volume fiber-optic evanescent-wave absorption sensor for nitrite determination. Anal Bioanal Chem 396, 943–948 (2010). https://doi.org/10.1007/s00216-009-3246-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-009-3246-2