Abstract
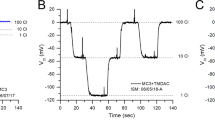
Self-referencing with ion-selective microelectrodes (ISMs) is a useful approach for monitoring near-real-time ion flux near single cells and across epithelia. While ISMs for H+, Ca2+, and K+ have been optimized for use with self-referencing, ISMs for two other primary inorganic ions, Na+ and Cl−, have not. In this study, we have characterized ISMs based on three Na+ ionophores (I, VI, and X) and one Cl− ionophore to assess their suitability for use with self-referencing. ISMs constructed with Na+ ionophore VI have short response times (≈100 ms) but possess nearly an order of magnitude less selectivity for Na+ over K+ than ISMs constructed with Na+ ionophore X. The Na+ ionophore X mixture was enhanced to give it a shorter response time while not compromising its selectivity. A Cl−-selective microelectrode was constructed and characterized with superior anionic selectivity compared with previously reported Cl− ISMs used with self-referencing. This Cl−-selective microelectrode, however, has a relatively slow response time (≈3 s), thus requiring changes to the self-referencing protocol. Self-referencing with these ISMs will enable near-real-time ion flux measurements for Na+ and Cl−.



Similar content being viewed by others
References
Smith PJS, Sanger RH, Messerli MA (2007) Principles, development and applications of self-referencing electrochemical microelectrodes to the determination of fluxes at cell membranes. In: Michael AC, Borland LM (eds) Electrochemical methods for neuroscience. CRC, Boca Raton, pp 373–405
Messerli MA, Robinson KR, Smith PJS (2006) Electrochemical sensor applications to the study of molecular physiology and analyte flux in plants. In: Volkov AG (ed) Plant electrophysiology. Springer, Berlin, pp 73–107
Messerli MA, Smith PJS, Lewis RC, Robinson KR (2004) Chloride fluxes in lily pollen tubes: a critical reevaluation. Plant J 40:799–812
Smith PJS, Hammar K, Porterfield DM, Sanger RH, Trimarchi JR (1999) Self-referencing, non-invasive, ion selective electrode for single cell detection of trans-plasma membrane calcium flux. Micros Res Tech 46:398–417
Umezawa Y, Bühlmann P, Umezawa K, Tohda K, Amemiya S (2000) Potentiometic selectivity coefficients of ion-selective electrodes Part I. Inorganic cations. Pure Appl Chem 72:1851–1856
Steiner RA, Oehme M, Ammann D, Simon W (1979) Neutral carrier sodium ion-selective microelectrode for intracellular studies. Anal Chem 51:351–353
Tamura H, Kimura K, Shono T (1982) Coated wire sodium- and potassium-selective electrodes based on bis(crown ether) compounds. Anal Chem 54:1224–1227
Cadogan AM, Diamond D, Smyth MR, Deasy M, McKervey MA, Harris S J (1989) Sodium-selective polymeric membrane electrodes based on calix[4]arene ionophores. Analyst 114:1551–1554
Phillips F, Kaczor K, Gandhi N, Pendley BD, Danish RK, Neuman MR, Tóth B, Horváth V, Lindner E (2007) Measurement of sodium ion concentration in undiluted urine with cation-selective polymeric membrane electrodes after the removal of interfering compounds. Talanta 74:255–264
Noël J, Pouysségur J (1995) Hormonal regulation, pharmacology, and membrane sorting of vertebrate Na+/H+ exchanger isoforms, Hormonal regulation, pharmacology, and membrane sorting of vertebrate Na+/H+ exchanger isoforms. Am J Physiol Cell Physiol 268:C283–C296
Garber SS, Messerli MA, Hubert M, Lewis R, Hammar K, Indyk E, Smith PJS (2005) Monitoring Cl- movement in single cells exposed to hypotonic solution. J Memb Biol 203:101–110
Bratov A, Abramova N, Domínguez C (2004) Investigation of chloride sensitive ISFETs with different membrane compositions suitable for medical applications. Anal Chim Acta 514:99–106
Acknowledgements
This research was funded by NIH-NCRR grant P41 RR001395 to P.J.S. Smith and by NIH Grants DK077162, DK07789, and DK058563, and DK063125 to I. Kurtz.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Messerli, M.A., Kurtz, I. & Smith, P.J.S. Characterization of optimized Na+ and Cl− liquid membranes for use with extracellular, self-referencing microelectrodes. Anal Bioanal Chem 390, 1355–1359 (2008). https://doi.org/10.1007/s00216-007-1804-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-007-1804-z