Abstract
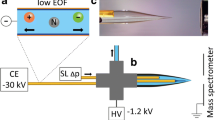
Detailed information on the geochemical behavior of radioactive and toxic metal ions under environmental conditions (in geological matrices and aquifer systems) is needed in order to assess the long-term safety of waste repositories. This includes knowledge of the mechanisms of relevant geochemical reactions, as well as associated thermodynamic and kinetic data. Several previous studies have shown that humic acid can play an important role in the immobilization or mobilization of metal ions due to complexation and colloid formation. In our project we investigate the complexation behavior of (purified Aldrich) humic acid and its influence on the migration of the lanthanides europium and gadolinium (homologs of the actinides americium and curium) in the ternary system consisting of these heavy metals, humic acid and kaolinite (KGa-1b) under almost natural conditions. Capillary electrophoresis (CE, Beckman Coulter P/ACE MDQ), with its excellent separation performance, was hyphenated with a homemade interface to inductively coupled plasma mass spectrometry (ICP–MS, VG Elemental PlasmaQuad 3) giving a system that is highly sensitive to the rare-earth element species of europium and gadolinium with humic acid. The humic acid used was also halogenated with iodine, which acted as an ICP–MS marker. To couple CE to ICP–MS, a fused silica CE capillary was flexibly fitted into a MicroMist 50 μl nebulizer with a Cinnabar cyclonic spray chamber in the external homemade interface. The chamber was chilled to a temperature of 4 °C to optimize the sensitivity. 200 ppb of cesium were added to the CE separation buffer so that the capillary flow could be observed. A make-up fluid including 4 ppb Ho as an internal standard was combined with the flow from the capillary within the interface in order to get a fluid throughput high enough to maintain continuous nebulization. Very low detection limits were achieved: 125 ppt for 153Eu and 250 ppt for 158Gd. Using this optimized CE–ICP–MS coupling system it was possible to quantify metal concentrations from the detection limit up to approximately 1 ppm (the linear range). This set-up was used to separate metal/humic acid-species in a 100 mM acetic acid/10 mM acetate buffer system. Using humic acid as the complexing ligand, uncomplexed metal ion species could be separated from metal–humate complexes on a time-resolved scale.






Similar content being viewed by others
References
Katz JJ, Seaborg GT, Morss LR (eds) (1986) The chemistry of the actinide elements. Chapman & Hall, New York
Kersting AB, Efurd DW, Finnegan DL, Rokop DJ, Smith DK, Thompson JL (1999) Nature 397:56–59
Stevenson FJ (1994) Humus chemistry. Wiley, New York
Orlov DS (1995) Humic substances of soils and general theory of humification. Balkema, Brookfeld, VT
Sachs S, Schmeide K, Reich T, Brendler V, Heise KH, Bernhard G (2005) Radiochim Acta 93:17–25
Marquardt CM (ed) (2000) Influence of humic acids on the migration behaviour of radioactive and non-radioactive substances under conditions close to nature: final report. Wissenschaftliche Berichte, FZKA 6557, Forschungszentrum Karlsruhe, Karlsruhe, Germany
Marquardt CM (ed) (2004) Investigations on the complexation behaviour of humic acids and their influence on the migration of radioactive and non-radioactive substances under conditions close to nature. Wissenschaftliche Berichte, FZKA-6999, Forschungszentrum Karlsruhe, Karlsruhe, Germany
Plaschke M, Rothe J, Denecke M, Fanghänel T (2004) J Electron Spectrosc 135:53–62
Kim HJ, Baek K, Kim BK, Yang JW (2005) J Hazard Mater 122:31–36
Wrobel K, Sadi BB, Wrobel K, Castillo JR, Caruso JA (2003) Anal Chem 75:761–767
Gehmecker H, Trautmann N, Herrmann G (1986) Radiochim Acta 40:81–88
Coates JT, Fjeld RA, Paulenova A, DeVol T (2001) J Radioanal Nucl Chem 248:501–506
Röllin S, Eklund UB (2000) J Chromatogr A 884:131–141
Barnes RM (1998) Fresenius J Anal Chem 361:246–251
Michalke B (1999) J Anal Atom Spectrom 14:1297–1302
Prange A, Schaumlöffel D (1999) Fresenius J Anal Chem 364:452–456
Schaumloffel D, Prange A, Marx G, Heumann KG, Bratter P (2002) Anal Bioanal Chem 372:155–163
Kuczewski B, Marquardt CM, Seibert A, Geckeis H, Kratz JV, Trautmann N (2003) Anal Chem 75:6769–6774
Pröfrock D, Leonhard P, Ruck W, Prange A (2005) Anal Bioanal Chem 381:194–204
Braun D, Wirth HO (1961) Patentschrift 1 088 227, Deutsches Patentamt
Acknowledgement
This work was carried out within the BMWA (German Federal Ministry of Economics and Labour) project “Investigations on the complexation behaviour of humic acids and their influence on the migration of radioactive and non-radioactive substances under conditions close to nature”. The authors therefore thank the BMWA for financial support through grant no. 02 E 9683.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kautenburger, R., Nowotka, K. & Beck, H.P. Online analysis of europium and gadolinium species complexed or uncomplexed with humic acid by capillary electrophoresis–inductively coupled plasma mass spectrometry. Anal Bioanal Chem 384, 1416–1422 (2006). https://doi.org/10.1007/s00216-006-0299-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-006-0299-3