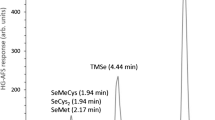
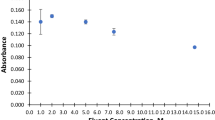
Abstract
Different chemical modifiers for use with electrothermal atomic absorption spectrometry (ET AAS) were investigated in relation to determining the selenium in human urine samples. The samples were diluted in a solution containing 1% v/v HNO3 and 0.02% m/v cetyltrimethylammonium chloride (CTAC). Studying the modifiers showed that the use of either Ru or Ir as the permanent modifier gave low sensitivity to Se and the peak shape was very noisy, while Zr or Rh gave no peak at all. The same occurred when Zr was used in solution. For mixtures of permanent modifiers, Ir plus Rh or Zr plus Rh gave very low sensitivity, Zr plus Rh with co-injection of Ir in solution was also not efficient, Zr plus Rh in solution gave good sensitivity, but the best results were obtained with a mixture of Zr and Rh as the permanent modifier and co-injection of Rh in solution. Using this last modifier, the following dilutions with the HNO3 and CTAC were studied: 1:1, 1:2, 1:3 and 1:4. The best dilution was 1:1, which promoted good sensitivity and a more defined peak shape and made it possible to correct for the background using a deuterium arc lamp. Under these conditions, a characteristic mass of 26±0.2 pg was obtained for Se in aqueous solution. Six certified urine samples were analyzed using matrix matching calibration and the measured concentrations were in agreement with the certified values, according to a t-test at the 95% confidence level. Recovery tests were carried out and the recoveries were in the range 100–103%, with relative standard deviation better than 9%. The limit of detection (LOD, 3 sd, n=10) was 3.0 μg L−1 in the sample. The treated graphite tube could be used for at least 600 atomization cycles without significant alteration of the analytical signal.





Similar content being viewed by others
References
Oga S (1996) Fundamentos de toxicologia. Atheneu Editora, São Paulo, SP, Brasil
Robberecht H, Deelstra H (1994) J Trace Elem Electrolytes Health Dis 8:129–143
Alaejos MS, Romero FJD, Romero CD (2000) Nutrition 16:376–383
Alaejos MS, Romero CD (1993) Clin Chem 39:2040–2052
Welz B, Sperling M (1999) Atomic absorption spectrometry, 3rd edn. Wiley-VCH, Germany
Tiran B, Tiran A, Rossipal E, Lorenz O (1993) J Trace Elem Electrolytes Health Dis 7:211–216
Navarro M, Lopez H, Lopez MC, Perez V (1996) J AOAC 79:773–776
Silva JBB, Giacomelli MBO, Souza IG, Curtius AJ (1998) Microchem J 60:249–257
Tyson JF, Sundin NG, Hanna CP, McIntosh SA (1997) Spectrochim Acta Part B 52:1773–1781
Bulska E, Thybusch B, Ortner HM (2001) Spectrochim Acta Part B 56:363–373
Drake EN, Hain TD (1994) Anal Biochem 220:336–339
Harrison I, Littlejohn D, Fell GS (1996) Analyst 121:1641–1646
Liang YZ, Li M, Rao Z (1996) Anal Sci 12:629–633
Campillo N, Vinas P, Lopez-Garcia I, Hernandez-Cordoba M (2000) Anal Biochem 280:195–200
Wang HC, Peng HW, Kuo MS (2001) Anal Sci 17527–532
Correia PRM, Oliveira E, Oliviera PV (2002) Talanta 57:527–535
Tsalev DL, Lampugnani L, D’Ulivo A, Petrov II Jr, Georgieva R, Marcucci K, Zamboni R (2001) Microchem J 70:103–113
Nunes BR, Magalhães CG, da Silva JBB (2002) J Anal Atom Spectrom 17:1335–1338
Gammelgaard B, Jons O (1999) J Anal Atom Spectrom 14:867–874
Nixon DE, Neubauer KR, Eckdahl SJ, Butz JA, Burrit MF (2003) Spectrochim Acta Part B 58:97–110
LaFuente JMG, Marchante-Gayon JM, Sanchez MLF, Sanz-Medel A (1999) Talanta 50:207–217
Silva JBB, Silva MAM, Curtius AJ, Welz B (1999) J Anal Atom Spectrom 14:1737–1742
Becker-Ross H, Florek S, Heitmann U (2000) J Anal Atom Spectrom 15:137–141
Volynsky AB, Wennrich R (2002) Spectrochim Acta Part B 57:1301–1316
Tsalev DL, Slaveykova VI, Lampugnani L, D’Ulivo A, Georgieva R (2000) Spectrochim Acta Part B 55:473–490
Volynsky AB (2004) Spectrochim Acta Part B 59:1799–1821
Acknowledgements
The authors are thankful to Conselho Nacional de Pesquisa e Desenvolvimento Tecnológico (CNPq, Brazil) and to Pró-Reitoria de Pesquisa (UFMG, Brazil) for financial support and scholarships. F.G. Pinto, T.D. Saint'Pierre, A. J. Curtius, F. R. Amorim and J.B.B. da Silva have scholarships from CNPq.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pinto, F.G., Andrada, D., Magalhães, C.G. et al. Direct determination of selenium in urine samples by electrothermal atomic absorption spectrometry using a Zr plus Rh-treated graphite tube and co-injection of Rh as chemical modifier. Anal Bioanal Chem 383, 825–832 (2005). https://doi.org/10.1007/s00216-005-0070-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-005-0070-1