Abstract
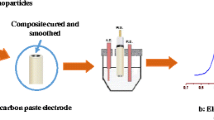
Cobalt-phthalocyanine-modified carbon paste electrodes are shown to be excellent indicators for electrocatalytic amperometric measurements of triazolic herbicides such as amitrole, at low oxidation potentials (+0.40 V). The detection and determination of amitrole in flow injection analysis with a modified carbon paste electrode with Co-phthalocyanine is described. The concentrations of amitrole in 0.1 M NaOH solutions were determined using the electrocatalytic oxidation signal corresponding to the Co(II)/Co(III) redox process. A detection limit of 0.04 µg mL–1 (4 ng amitrole) was obtained for a sample loop of 100 µL at a fixed potential of +0.55 V (vs. Ag/AgCl) in 0.1 M NaOH and a flow rate of 4.0 mL min–1. Furthermore, the modified carbon paste electrodes offers reproducible responses in such a system, and the relative standard deviation was 3.3% using the same surface, 5.1% using different surface, and 6.9% using different pastes. The performance of the cobalt-phthalocyanine-modified carbon paste electrodes is illustrated here for the determination of amitrole in commercial formulations. The response of the electrodes is stable, with more than 80% of the initial retained activity after 50 min of continuous use.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Chicharro, M., Zapardiel, A., Bermejo, E. et al. Electrocatalytic amperometric determination of amitrole using a cobalt-phthalocyanine-modified carbon paste electrode. Anal Bioanal Chem 373, 277–283 (2002). https://doi.org/10.1007/s00216-002-1345-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-002-1345-4