Abstract
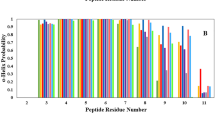
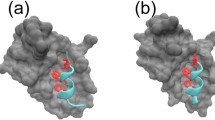
Protein P53 is involved in more than 50% of the human cancers and the P53–MDM2 complex is a target for anticancer drug design. It is possible to engineer small P53 mimics that would be expected to disrupt the P53–MDM2 complex, and release P53 to initiate cell-cycle arrest or apoptosis. These small peptides should bind to the functional epitopes of the protein–protein interface, and prevent the interaction between P53 and MDM2. Here, we apply an improved computational alanine scanning mutagenesis method, which allows the determination of the hot spots present in both monomers, P53 and MDM2, of three protein complexes (the P53-binding domain of human MDM2, its analogue from Xenopus laevis, and the structure of human MDM2 in complex with an optimized P53 peptide). The importance of the hydrogen bonds formed by the protein backbone has been neglected due to the difficulty of measuring experimentally their contribution to the binding free energy. In this study we present a computational approach that allows the estimation of the contribution to the binding free energy of the C=O and N–H groups in the backbone of the P53 and MDM2 proteins. We have noticed that the hydrogen bond between the HE1 atom of the hot spot Trp23 and the O atom of the residue Leu54, as well as the NH-pi hydrogen bond between the Ile57 and Met58 were observed in the Molecular dynamics simulation, and their contribution to the binding free energy measured. This study not only shows the reliability of the computational mutagenesis method to detect hot spots but also demonstrates an excellent correlation between the quantitative calculated binding free energy contribution of the C=O and N–H backbone groups of the interfacial residues and the qualitative values expected for this kind of interaction. The study also increases our understanding of the P53–MDM2 interaction.
Similar content being viewed by others
References
Kussie PH, Gorina S, Marechal V, Elenbaas B, Moreau J, Levine AJ, Pavletich NP (1996) Science 274: 948–953
Chène P (2004) Mol Cancer Res 2: 20–28
Soussi T, Dehouche K, Beroud C (2000) Hum Mutat 2: 105–213
Chi CW, Lee SH, Do-Hyoung K, Min-Jung A, Jae-Sung K, Jin-Young W, Takuya T, Masatsune K, Kyou-Hoon H (2005) J Biol Chem 280: 38795–38802
Welburn JPI, Endicott JA (2005) Sem Cell Devel Biol 16: 369–381
Mateu MG, Fersht A (1998) EMBO J 17: 2748–2758
Picksley SM, Lane DP (1993) Bioessays 2: 689–690
Wu X, Bayle JH, Olson D, Levine AJ (1993) Genes Dev 2: 1126–1132
Momand J, Zambetti GP, Olson DC, George D, Levine AJ (1992) Cell 2: 1237–1245
Zhong H, Carlson HA (2005) Proteins 58: 222–234
Lu F, Chi SW, kim DH, Han KH, kuntz ID, Guy RK (2006) J Comb Chem 8: 315–325
Grasberger BL, Schubert C, Koblish HK, Carver TE, Franks CF, Zhao SY, Lu T, LaFrance LV, Parks DJ (2005) J Med Chem 48: 909–912
Massova M, Kollman PA (1999) J Am Chem Soc 121: 36
Bottger A, Bottger V, Garcia-Echeverria C, Chene P, Hochkeppel HK, Sampson W, Ang K, Howard SF, Picksley SM, Lane DP (1997) J Mol Biol 269: 744–756
Ma B, Nussinov R (2207) Curr Top Med Chem 7: 999–1005
DeLano WL, Ultsch MH, de Vos AM, Wells JA (2000) Science 287: 1279–1283
Bogan AA, Thorn KS (1998) J Mol Biol 280: 1–9
Ma B, Wolfson HJ, Nussinov R (2001) Curr Opin Struct Biol 11: 364–369
Moreira IS, Fernandes PA, Ramos MJ (2006) Proteins 63: 811–21
Moreira IS, Fernandes PA, Ramos MJ (2006) J Phys Chem B 110: 10962–10969
Moreira IS, Fernandes PA, Ramos MJ (2007) 117:99–113
Case DA, Darden TA, Cheatham III TE, Simmerling CL, Wang J, Duke RE, Luo R, Merz HM, Wang B, Pearlman DA, Crowley M, Brozell S, Tsui V, Gohlke H, Mongan J, Hornak V, Cui G, Beroza P, Schafmeister C, Caldwell JW, Ross WS, Kollman PA (2004) AMBER 8, University of California, San Francisco
Tsui V, Case DA (2001) Biopolymers (Nucl Acid Sci) 56: 275–291
Cornell WD, Cieplak P, Bayly CI, Gould IR, Merz KM Jr., Ferguson DM, Spellmeyer DC, Fox T, Caldwell JW, Kollman PA (1995) J Am Chem Soc 117: 5179–5197
Ryckaert JP, Ciccotti G, Berendsen HJ (1977) J Comput Phys 23: 327–335
Pastor RW, Brooks BR, Szabo A (1988) Mol Phys 65: 1409–1419
Loncharich RJ, Brooks BR, Pastor RW (1992) Biopolymers 32: 523–535
Izaguirre JA, Catarello DP, Wozniak JM, Skeel RD (2001) J Chem Phys 114: 2090–2098
Huo S, Massova I, Kollman PA (2002) J Comput Chem 23: 15–27
Rocchia W, Sridharan S, Nicholls A, Alexov E, Chiabrera A, Honig B (2002) J Comp Chem 23: 128–137
Rocchia W, Alexov E, Honig B (2001) J Phys Chem B 105: 6507–6514
Moreira IS, Fernandes PA, Ramos MJ (2005) J Mol Struct (Theochem) 729: 11–18
Connolly ML (1983) J Appl Cryst 16: 548–558
Gohlke H, Case DA (2004) J Comput Chem 25: 238–250
Xu D, Tsai CJ, Nussinov R (1997) Protein Eng 10: 999–1012
Biot C, Buisine E,Kwasigroch JM,Wintiens R, RoomanM (2002) 277:40816–40822 (1997)
Author information
Authors and Affiliations
Corresponding author
Additional information
Contribution to the Nino Russo Special Issue.
Rights and permissions
About this article
Cite this article
Moreira, I.S., Fernandes, P.A. & Ramos, M.J. Protein–protein recognition: a computational mutagenesis study of the MDM2–P53 complex. Theor Chem Account 120, 533–542 (2008). https://doi.org/10.1007/s00214-008-0432-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-008-0432-9