Abstract
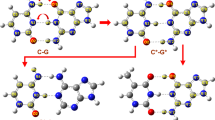
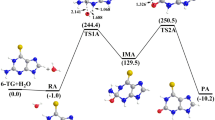
Two pathways involving proton catalyzed hydrolytic deamination of cytosine (to uracil) are investigated at the PCM-corrected B3LYP/6-311G(d,p) level of theory, in the presence of an additional catalyzing water molecule. It is concluded that the pathway involving initial protonation at nitrogen in position 3 of the ring, followed by water addition at C4 and proton transfer to the amino group, is a likely route to hydrolytic deamination. The rate determining step is the addition of water to the cytosine, with a calculated free energy barrier in aqueous solution of ΔG ≠=140 kJ/mol. The current mechanism provides a lower barrier to deamination than previous work based on OH − catalyzed reactions, and lies closer to the experimental barrier derived from rate constants (E a = 117 ± 4 kJ/mol).
Similar content being viewed by others
References
Burgur A (1983). A Guide to the Chemical Basis of Drug Design. Wiley, New York
Peng W and Shaw BR (1996). Biochemistry 35: 10172–10181
Florián J, Baumruk V and Leszczyñski J (1996). J Phys Chem 100: 5578–5589
Sponer J, Leszczynski J and Hobza P (1996). J Comput Chem 117: 841–850
Estrin DA, Paglieri L and Corongiu G (1994). J Phys Chem 98: 5653–5660
Scanlan MJ and Hillier IH (1984). J Am Chem Soc 106: 3737–3745
Chandra AK, Nguyen MT and Zeegers-Huyskens T (2000). J Mol Struct (Theochem) 519: 1–11
Chandra AK, Michalska D, Wysokinsky R and Zeegers-Huyskens T (2004). J Phys Chem A 108: 9593–9600
Civcir PÜ (2000). J Mol Struc (Theochem) 532: 157–169
Gould IR, Green DVS, Young P and Hillier IH (1992). J Org Chem 57: 4434–4437
Morpurgo S, Bossa M and Morpurgo GO (2000). Adv Quantum Chem 36: 169–183
Sambrano JR, Souza AR, Queralt JJ and Andrés J (2000). Chem Phys Lett 317: 437–443
Colominas C, Luque FJ and Orozco M (1996). J Am Chem Soc 118: 6811–6821
Fogarasi G (2002). J Phys Chem A 106: 1381–1390
Fogarasi G and Szalay PG (2002). Chem Phys Lett 356: 383–390
Person WB, Szczepaniak K, Szczesniak M, Kwiatkowski JS, Hernandez L and Czerminski R (1989). J Mol Struc (Theochem) 194: 239–258
Brown RD, Godfrey PD, McNaughton D and Pierlot AP (1989). J Am Chem Soc 111: 2308–2310
Monajjemi M, Ghiasi R, Ketabi S, Passdar H, Mollaamin F (2004) pp 11–18
Monajjemi M, Ghiasi R and Abedi A (2005). Theor Inorg Chem 50: 435–441
Burda J, Sponer J, Leszczynski J and Hobza P (1997). J Phys Chem B 101: 9670–9677
Sponer J, Burda JV, Sabat M, Leszczynski J and Hobza P (1998). J Phys Chem A 102: 5951–5957
Sponer JE, Miguel PJ, Rodruigez-Santiago L, Erxleben A, Krumm M, Sodupe M, Sponer J and Lippert B (2004). Angew Chem Int Ed 43: 5396–5399
Russo N, Toscano M and Grand A (2001). J Phys Chem B 105: 4735–4741
Russo N, Sicilia E, Toscano M and Grand A (2002). Int J Quantum Chem 90: 903–909
Russo N, Toscano M and Grand A (2001). J Am Chem Soc 123: 10272–10279
Russo N, Toscano M and Grand A (2003). J Mass Spectrom 38: 265–270
Russo N, Toscano M and Grand A (2003). J Phys Chem A 107: 11533–11538
Marino T, Toscano M, Russo N and Grand A (2004). Int J Quantum Chem 98: 347–354
Marino T, Mazzuca D, Toscano M, Russo N and Grand A (2007). Int J Quantum Chem 107: 311–317
Brown D and Phillips JH (1965). J Mol Biol 11: 663–671
Notari RE, Chin ML and Cardoni A (1970). J Pharm Sci 59: 27–32
Dreyfus M, Bensaude O, Dodin G and Dubois JE (1976). J Am Chem Soc 98: 6338–6349
Shapiro R and Klein R (1966). Biochemistry 5: 2358–2362
Chen H and Shaw BR (1994). Biochemistry 33: 4121–4129
Glaser R, Rayat S, Lewis M, Son M-S and Meyer S (1999). J Am Chem Soc 121: 6108–6119
Shapiro R and Klein R (1967). Biochemistry 6: 3576–3782
Frederico LA:, Kunkel TA and Shaw BR (1990). Biochemistry 29: 2532–2537
Lindahl T and Nyberg B (1974). Biochemistry 13: 3405–3410
Duncan BK and Miller JH (1980). Nature 287: 560–561
Almatarneh MH, Flinn CG, Poirier RA and Sokalski WA (2006). J Phys Chem A 110: 8227–8234
Yao L, Li Y, Wu Y, Liu A and Yan H (2005). Biochemistry 44: 5940–5947
Becke AD (1993). J Chem Phys 98: 5648–5652
Lee C, Yang W and Parr RG (1988). Phys Rev B 37: 785–789
McLean AD and Chandler GS (1980). J Chem Phys 72: 5639
Krishnan R, Binkley JS, Seeger R and Pople JA (1980). J Chem Phys 72: 650
Frisch MJ, Pople JA and Binkley JS (1984). J Chem Phys 80: 3265
Cancès MT, Mennucci B and Tomasi J (1997). J Chem Phys 107: 3032–3041
Mennucci B and Tomasi J (1997). J Chem Phys 106: 5151–5158
Cossi M, Scalmani G, Rega N and Barone V (2002). J Chem Phys 117: 43–54
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskortz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chem W, Wong MW, Gonzalez C, Pople JA. Gaussian 03, Revision C.02, Gaussian: Wallingford, 2004
Llano J and Eriksson LA (2002). J Chem Phys 117: 10193–10206
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Nino Russo on the occasion of his 60th birthday.
Electronic Supplementary Material
The Below is the Electronic Supplementary Material.
Rights and permissions
About this article
Cite this article
Labet, V., Grand, A., Morell, C. et al. Proton catalyzed hydrolytic deamination of cytosine: a computational study. Theor Chem Account 120, 429–435 (2008). https://doi.org/10.1007/s00214-008-0418-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-008-0418-7