Abstract.
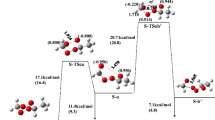
Theoretical studies on the thermolysis in the gas phase of alkyl N,N-dialkylcarbamates were carried out using ab initio theoretical methods, at the MP2/6-31G(d), MP2/6-31++G(d,p) and MP2/6-311++G(2d,p)//MP2/6-31G(d) levels. The reactions have two steps: the first one corresponds to the formation of an alkene and a neutral dialkylcarbamic acid intermediate via a six-membered cyclic transition state; the second one is the decarboxylation of this intermediate via a four-membered cyclic transition state, leading to carbon dioxide and the corresponding dialkylamine. The progress of the reactions was followed by means of the Wiberg bond indices. The results indicate that the transition states have character intermediate between reactants and products, and the calculated synchronicities show that the reactions are slightly asynchronous. The bond-breaking processes are more advanced than the bond-forming ones, indicating a bond deficiency in the transition states. The rate constants calculated for all the reactions agree very well with the available experimental data.
Similar content being viewed by others
Acknowledgments.
This work was supported by a research fund provided by the Comité de Investigación y Desarrollo Científico, Universidad Nacional de Colombia, Sede Medellín, and COLCIENCIAS (project no. 1118-05-1148), and the Spanish Dirección General de Investigación, Ministerio de Ciencia y Tecnología (project no. BQU20001499). The authors also thank COLCIENCIAS and C.S.I.C. for the joint project no. 2001CO0004, and the program Formación de Jóvenes Investigadores (Ederley Vélez)—COLCIENCIAS.
Author information
Authors and Affiliations
Corresponding author
Additional information
From the Proceedings of the 28th Congreso de Químicos Teóricos de Expresión Latina (QUITEL 2002)
Rights and permissions
About this article
Cite this article
Quijano, C., Notario, R., Quijano, J. et al. Theoretical study of the gas-phase thermolysis reaction of alkyl (ethyl, isopropyl, and tert-butyl) N,N-dimethylcarbamates and N,N-diethylcarbamates. Theor Chem Acc 110, 377–386 (2003). https://doi.org/10.1007/s00214-003-0491-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-003-0491-x