Abstract
Rationale
Risky choice can be measured using the risky decision task (RDT). In the RDT, animals choose between a large, risky option that is paired with probabilistic foot shock and a small, safe option that is never paired with shock. To date, studies examining the neurochemical basis of decision-making in the RDT have focused primarily on the dopaminergic system but have not focused on the glutamatergic system, which has been implicated in risky decision-making.
Objectives
Because glutamate is known to play a critical role in decision-making, we wanted to determine the contribution of the glutamatergic system to performance in the RDT.
Methods
In the experiment, 32 rats (16 male; 16 female) were tested in the RDT. The probability of receiving a foot shock increased across the session (ascending schedule) for half of the rats but decreased across the session (descending schedule) for half of the rats. Following training, rats received injections of the N-methyl-d-aspartate (NMDA) receptor competitive antagonist CGS 19755 (0, 1.0, 2.5, 5.0 mg/kg; s.c.) and the GluN2B-selective antagonist Ro 63-1908 (0, 0.1, 0.3, 1.0 mg/kg; s.c.).
Results
CGS 19755 (2.5 and 5.0 mg/kg) increased risky choice in males and females trained on the ascending schedule. Ro 63-1908 (1.0 mg/kg) decreased risky choice, but only in male rats trained on the ascending schedule.
Conclusions
Although NMDA receptor antagonists differentially alter risky choice in the RDT, the current results show that NMDA receptors are an important mediator of decision-making involving probabilistic delivery of positive punishment.





Similar content being viewed by others
Notes
In traditional discounting functions, A simply refers to reinforcer amount (typically the amount of the large magnitude reinforcer). This definition can be problematic in animal behavioral pharmacology experiments, in which pharmacological manipulations can drastically alter an animal’s preference for the large magnitude reinforcer, even when its delivery is immediate/guaranteed/delivered without shock.
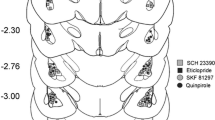
There were 26 rats that received all eight injections (208 injections), two rats that received 10 injections (due to retraining that occurred after receiving the second injection during the second set of injections; 20 injections), one rat that received three injections before losing stimulus control, and one rat that received one injection before losing stimulus control.
Because we used the percentage of receiving foot shock for the x-axis as opposed to the odds against receiving no shock, the proportion of responses for the large, risky option appears to decrease linearly as a function of probability. If we express the x-axis as odds against, the data are curvilinear, thus making the use of an exponential function appropriate.
References
Abela AR, Chudasama Y (2013) Dissociable contributions of the ventral hippocampus and orbitofrontal cortex to decision-making with a delayed or uncertain outcome. Eur J Neurosci 37:640–647. https://doi.org/10.1111/ejn.12071
Amador M, Dani JA (1991) MK-801 inhibition of nicotinic acetylcholine receptor channels. Synapse 7:207–215. https://doi.org/10.1002/syn.890070305
Bennett DA, Bernard PS, Amrick CL, Wilson DE, Liebman JM, Hutchinson AJ (1989) Behavioral pharmacological profile of CGS 19755, a competitive antagonist at N-methyl-D-aspartate receptors. J Pharmacol Exp Ther 250:454–460
Borges AM, Kuang J, Milhorn H, Yi R (2016) An alternative approach to calculating area-under-the-curve (AUC) in delay discounting research. J Exp Anal Behav 106:145–155. https://doi.org/10.1002/jeab.219
Brand M, Kalbe E, Labudda K, Fujiwara E, Kessler J, Markowitsch HJ (2005) Decision making impairments in patients with pathological gambling. Psychiatry Res 133:91–99. https://doi.org/10.1016/j.psychres.2004.10.003
Brevers D, Bechara A, Cleeremans A, Kornreich C, Verbank P, Noël X (2014) Impaired decision-making under risk in individuals with alcohol dependence. Alcohol Clin Exp Res 38:1924–1931. https://doi.org/10.1111/acer.12447
Cardinal RN, Howes NJ (2005) Effects of lesions of the nucleus accumbens core on choice between small certain rewards and large uncertain rewards in rats. BMC Neurosci 6:37. https://doi.org/10.1186/1471-2202-6-37
Carlezon WA Jr, Thomas MJ (2009) Biological substrates of reward and aversion: a nucleus accumbens activity hypothesis. Neuropharmacol 56:122–132. https://doi.org/10.1016/j.neuropharm.2008.06.075
Cottone P, Iemolo A, Narayan AR, Kwak J, Momaney D, Sabino V (2013) The uncompetitive NMDA receptor antagonists ketamine and memantine preferentially increase the choice for a small, immediate reward in low-impulsive rats. Psychopharmacology 226:127–138. https://doi.org/10.1007/s00213-012-2898-3
Deng JV, Orsini CA, Shimp KG, Setlow B (2018) MeCP2 expression in a rat model of risky decision making. Neuroscience 369:212–221. https://doi.org/10.1016/j.neuroscience.2017.11.016
Dobrovitsky V, West MO, Horvitz JC (2019) The role of the nucleus accumbens in learned approach behavior diminishes with training. Eur J Neurosci 50:3403–3415. https://doi.org/10.1111/ejn.14523
Evenden JL, Ryan CN (1996) The pharmacology of impulsive behaviour in rats: the effects of drugs on response choice with varying delays of reinforcement. Psychopharmacology 128:161–170. https://doi.org/10.1007/s002130050121
France CP, Winger GD, Woods JH (1990) Analgesic, anesthetic, and respiratory effects of the competitive N-methyl-D-aspartate (NMDA) antagonist CGS 19755 in rhesus monkeys. Brain Res 526:355–358. https://doi.org/10.1016/0006-8993(90)91247-e
Ferland JN, Winstanley CA (2017) Risk-preferring rats make worse decisions and show increased incubation of craving after cocaine self-administration. Addict Biol 22:991–1001. https://doi.org/10.1111/adb.12388
Green L, Myerson J (2004) A discounting framework for choice with delayed and probabilistic rewards. Psychol Bull 130:769–792. https://doi.org/10.1037/0033-2909.130.5.769
Gueorguieva R, Krystal JH (2004) Move over ANOVA: progress in analyzing repeated-measures data and its reflection in papers published in the Archives of General Psychiatry. Arch Gen Psychiatry 61:310–317. https://doi.org/10.1001/archpsyc.61.3.310
Higgins GA, Silenieks LB, MacMillan C, Sevo J, Zeeb FD, Thevarkunnel S (2016) Enhanced attention and impulsive action following NMDA receptor Glu2N2B-selective antagonist pretreatment. Behav Brain Res 311:1–14. https://doi.org/10.1016/j.bbr.2016.05.025
Higgins GA, Silenieks LB, MacMillan C, Zeeb FD, Thevarkunnel S (2018) Effects of the NMDA receptor antagonists dizocilpine and Ro 63-1908 on delay-discounting and risky decision-making in a gambling task. Behav Brain Res 348:201–210. https://doi.org/10.1016/j.bbr.2018.04.028
Jiménez-Sánchez L, Campa L, Auberson YP, Adell A (2014) The role of GluN2A and GluN2B subunits on the effects of NMDA receptor antagonists in modeling schizophrenia and treating refractory depression. Neuropsychopharmacology 39:2673–2680. https://doi.org/10.1038/npp.2014.123
Kalivas PW (2009) The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci 10:561–572. https://doi.org/10.1038/nrn2515
Kapur S, Seeman P (2002) NMDA receptor antagonists ketamine and PCP have direct effects on the dopamine D2 and serotonin 5-HT2 receptors—implications for models of schizophrenia. Mol Psychiatry 7:837–844. https://doi.org/10.1038/sj.mp.4001093
Kawabe K, Iwasaki T, Ichitani Y (2007) Repeated treatment with N-methyl-d-aspartate antagonists in neonatal, but not adult, rats causes long-term deficits of radial-arm maze learning. Brain Res 1169:77–86. https://doi.org/10.1016/j.brainres.2007.06.062
Kwan D, Craver CF, Green L, Myerson J, Rosenbaum RS (2013) Dissociations in future thinking following hippocampal damage: evidence from discounting and time perspective in episodic amnesia. J Exp Psychol Gen 142:1355–1369. https://doi.org/10.1037/a0034001
Laurie DJ, Seeburg PH (1994) Ligand affinities at recombinant N-methyl-D-aspartate receptors depend on subunit composition. Eur J Pharmacol 268:335–345. https://doi.org/10.1016/0922-4106(94)90058-2
Lehmann J, Hutchinson AJ, McPherson SE, Mondadori C, Schmutz M, Sinton CM, Tsai C, Murphy DE, Steel DJ, Williams M (1988) CGS 19755, a selective and competitive N-methyl-D-aspartate-type excitatory amino acid receptor antagonist. J Pharmacol Exp Ther 246:65–75
Lenth R, Singmann H, Love J, Buerkner P, Herve M (2019) emmeans: estimated marginal means, aka least-squares means, R Foundation for Statistical Computing, Vienna, Austria. https://CRAN.R-project.org/package=emmeans
Li HB, Matsumoto K, Yamamoto M, Watanabe H (1997) NMDA but not AMPA receptor antagonists impair the delay-interposed radial maze performance of rats. Pharmacol Biochem Behav 58:249–253. https://doi.org/10.1016/s0091-3057(97)00015-4
Li JT, Su YA, Guo CM, Feng Y, Yang Y, Huang RH, Si TM (2011) Persisting cognitive deficits induced by low-dose, subchronic treatment with MK-801 in adolescent rats. Eur J Pharmacol 652:65–72. https://doi.org/10.1016/j.ejphar.2010.10.074
Lima-Ojeda JM, Vogt MA, Pfeiffer N, Dormann C, Köhr G, Sprengel R, Gass P, Inta D (2013) Pharmacological blockade of GluN2B-containing NMDA receptors induces antidepressant-like effects lacking psychotomimetic action and neurotoxicity in the perinatal and adult rodent brain. Prog Neuro-Psychopharmacol Biol Psychiatry 45:28–33. https://doi.org/10.1016/j.pnpbp.2013.04.017
Madden GJ, Petry NM, Johnson PS (2009) Pathological gamblers discount probabilistic rewards less steeply than matched controls. Exp Clin Psychopharmacol 17:283–290. https://doi.org/10.1037/a0016806
Mendez IA, Damborsky JC, Winzer-Serhan UH, Bizon JL, Setlow B (2013) ⍺4β2 and ⍺7 nicotinic acetylcholine receptor binding predicts choice preference in two cost benefit decision making tasks. Neuroscience 230:121–131. https://doi.org/10.1016/j.neuroscience.2012.10.067
Mitchell MR, Weiss VG, Beas BS, Morgan D, Bizon JL, Setlow B (2014) Adolescent risk taking, cocaine self-administration, and striatal dopamine signaling. Neuropsychopharmacology 39:955–962. https://doi.org/10.1038/npp.2013.295
Monaghan DT, Cotman CW (1985) Distribution of N-methyl-D-aspartate-sensitive L-[3H]glutamate-binding sites in rat brain. J Neurosci 5:2909–2919. https://doi.org/10.1523/JNEUROSCI.05-11-02909.1985
Moscarello JM, Ben-Shahar O, Ettenberg A (2007) Dynamic interaction between medial prefrontal cortex and nucleus accumbens as a function of both motivational state and reinforcer magnitude: a c-Fos immunocytochemistry study. Brain Res 1169:69–76. https://doi.org/10.1016/j.brainres.2007.06.064
Myerson J, Green L, Warusawitharana M (2001) Area under the curve as a measure of discounting. J Exp Anal Behav 76:235–243. https://doi.org/10.1901/jeab.2001.76-235
National Research Council (2011) Guide for the care and use of laboratory animals, 8th edn. National Academies Press, Washington
Nishijima K, Kashiwa A, Nishikawa T (1994) Preferential stimulation of extracellular release of dopamine in rat frontal cortex to striatum following competitive inhibition of the N-methyl-D-aspartate receptor. J Neurochem 63:375–378. https://doi.org/10.1046/j.1471-4159.1994.63010375.x
Orsini CA, Trotta RT, Bizon JL, Setlow B (2015) Dissociable roles for the basolateral amygdala and orbitofrontal cortex in decision-making under risk of punishment. J Neurosci 35:1368–1379. https://doi.org/10.1523/JNEUROSCI.3586-14.2015
Orsini CA, Willis ML, Gilbert RJ, Bizon JL, Setlow B (2016) Sex differences in a rat model of risky decision making. Behav Neurosci 130:50–61. https://doi.org/10.1037/bne0000111
Orsini CA, Heshmati SC, Garman TS, Wall SC, Bizon JL, Setlow B (2018) Contributions of medial prefrontal cortex to decision making involving risk of punishment. Neuropharmacol 139:205–216. https://doi.org/10.1016/j.neuropharm.2018.07.018
Orsini CA, Blaes SL, Dragone RJ, Betzhold SM, Finner AM, Bizon JL, Setlow B (2020) Distinct relationships between risky decision making and cocaine self-administration under short- and long-access conditions. Prog Neuro-Psychopharmacol Biol Psychiatry 98:109791. https://doi.org/10.1016/j.pnpbp.2019.109791
Pettorruso M, De Risio L, Martinotti G, Di Nicola M, Ruggeri F, Conte G, Di Giannantonio M, Janiri L (2014) Targeting the glutamatergic system to treat pathological gambling: current evidence and future perspectives. Biomed Res Int 2014:109786–109711. https://doi.org/10.1155/2014/109786
Phillips AG, Vacca G, Ahn S (2008) A top-down perspective on dopamine, motivation and memory. Pharmacol Biochem Behav 90:236–249. https://doi.org/10.1016/j.pbb.2007.10.014
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2019) nlme: linear and nonlinear mixed effects models. R Foundation for statistical computing, Vienna https://CRAN.R-project.org/package=nlme
Rachlin H, Raineri A, Cross D (1991) Subjective probability and delay. J Exp Anal Behav 55:233–244. https://doi.org/10.1901/jeab.1991.55-233
Rammes G, Rupprecht R, Ferrari U, Zieglgänsberger W, Parsons CG (2001) The N-methyl-D-aspartate receptor channel blockers memantine, MRZ 2/579 and other amino-alkyl-cyclohexanes antagonize 5-HT(3) receptor currents in cultured HEK-293 and N1E-115 cell systems in a non-competitive manner. Neurosci Lett 306:81–84. https://doi.org/10.1016/s0304-3940(01)01872-9
Ranganathan M, DeMartinis N, Huguenel B, Gaudreault F, Bednar MM, Shaffer CL, Gupta S, Cahill J, Sherif MA, Mancuso J, Zumpano L, D’Souza DC (2017) Attenuation of ketamine-induced impairment in verbal learning and memory in healthy volunteers by the AMPA receptor potentiator PF-04958242. Mol Psychiatry 22:1633–1640. https://doi.org/10.1038/mp.2017.6
Schutter DJ, van Bokhoven I, Vanderschuren LJ, Lochman JE, Matthys W (2011) Risky decision making in substance dependent adolescents with a disruptive behavior disorder. J Abnorm Child Psychol 39:333–339. https://doi.org/10.1007/s10802-010-9475-1
Shimp KG, Mitchell MR, Beas BS, Bizon JL, Setlow B (2015) Affective and cognitive mechanisms of risky decision making. Neurobiol Learn Mem 117:60–70. https://doi.org/10.1016/j.nlm.2014.03.002
Simon NW, Gilbert RJ, Mayse JD, Bizon JL, Setlow B (2009) Balancing risk and reward: a rat model of risky decision making. Neuropsychopharmacology 34:2208–2217. https://doi.org/10.1038/npp.2009.48
St Onge JR, Floresco SB (2009) Dopaminergic modulation of risk-based decision making. Neuropsychopharmacology 34:681–697. https://doi.org/10.1038/npp.2008.121
Tang AH, Franklin SR (1983) Disruption of brightness discrimination in a shock avoidance task by phencyclidine and its antagonism in rats. J Pharmacol Exp Ther 225:503–508
Wang Y, Ma Y, Hu J, Cheng W, Jiang H, Zhang X, Li M, Ren J, Li X (2015) Prenatal chronic mild stress induces depression-like behavior and sex-specific changes in regional glutamate receptor expression patterns in adult rats. Neuroscience 301:363–374. https://doi.org/10.1016/j.neuroscience.2015.06.008
Winstanley CA, Floresco SB (2016) Deciphering decision making: variation in animal models of effort- and uncertainty-based choice reveals distinct neural circuitries underlying core cognitive processes. J Neurosci 36:12069–12079. https://doi.org/10.1523/JNEUROSCI.1713-16.2016
Yates JR (2019) Examining the neurochemical underpinnings of animal models of risky choice: methodological and analytic considerations. Exp Clin Psychopharmacol 27:178–201. https://doi.org/10.1037/pha0000239
Yates JR, Batten SR, Bardo MT, Beckmann JS (2015) Role of ionotropic glutamate receptors in delay and probability discounting in the rat. Psychopharmacology 232:1187–1196. https://doi.org/10.1007/s00213-014-3747-3
Yates JR, Brietenstein KA, Gunkel BT, Hughes MN, Johnson AB, Rogers KK, Sharpe SM (2016) Effects of NMDA receptor antagonists on probability discounting depend on the order of probability presentation. Pharmacol Biochem Behav 150-151:31–38. https://doi.org/10.1016/j.pbb.2016.09.004
Yates JR, Gunkel BT, Rogers KK, Hughes MN, Prior NA (2017a) Effects of N-methyl-D-aspartate receptor ligands to sensitivity to reinforcer magnitude and delayed reinforcement in a delay-discounting procedure. Psychopharmacology 234:461–473. https://doi.org/10.1007/s00213-016-4469-5
Yates JR, Rogers KK, Gunkel BT, Prior NA, Hughes MN, Sharpe SM, Campbell HL, Johnson AB, Keller MG, Breitenstein KA, Shults HN (2017b) Effects of group I metabotropic glutamate receptor antagonists on sensitivity to reinforcer magnitude and delayed reinforcement in a delay-discounting task in rats: contribution of delay presentation order. Behav Brain Res 322(Part A):29–33. https://doi.org/10.1016/j.bbr.2017.01.015
Yates JR, Prior NA, Chitwood MR, Day HA, Heidel JR, Hopkins SE, Muncie BT, Paradella-Bradley TA, Sestito AP, Vecchiola AN, Wells EE (2018) Effects of GluN2B-selective antagonists on delay and probability discounting in male rats: modulation by delay/probability presentation order. Exp Clin Psychopharmacol 26:525–540. https://doi.org/10.1037/pha0000216
Yates JR, Chitwood MR, Evans KE, Kappesser JL, Murray CP, Paradella-Bradley TA, Torline BT (2019a) Group I metabotropic receptor antagonists impair discriminability of reinforcer magnitude, but not risky choice, in a probability-discounting task. Behav Brain Res 365:77–81. https://doi.org/10.1016/j.bbr.2019.02.047
Yates JR, Day HA, Evans KE, Igwe HO, Kappesser JL, Miller AL, Murray CP, Torline BT, Ellis AL, Stacy WL (2019b) Effects of d-amphetamine and MK-801 on impulsive choice: modulation by schedule of reinforcement and delay length. Behav Brain Res 376:112228. https://doi.org/10.1016/j.bbr.2019.112228
Young ME (2017) Discounting: a practical guide to multilevel analysis of indifference data. J Exp Anal Behav 108:97–112. https://doi.org/10.1002/jeab.265
Young ME, Clark MH, Goffus A, Hoane MR (2009) Mixed effects modeling of Morris water maze data: advantages and cautionary notes. Learn Motiv 40:160–177. https://doi.org/10.1016/j.lmot.2008.10.004
Zeeb FD, Robbins TW, Winstanley CA (2009) Serotonergic and dopaminergic modulation of gambling behavior as assessed using a novel rat gambling task. Neuropsychopharmacology 34:2329–2343. https://doi.org/10.1038/npp.2009.62
Zhuo M (2017) Ionotropic glutamate receptors contribute to pain transmission and chronic pain. Neuropharmacology 112:228–234. https://doi.org/10.1016/j.neuropharm.2016.08.014
Acknowledgments
We would like to thank the NIMH Chemical Synthesis and Drug Supply Program for generously providing the CGS 19755 used in the current experiment. We would also like to thank Karson Evans and Kadyn Lilly for the technical assistance during the pilot studies that were important for the development of the current study.
Funding
The current study was supported by NIH grant R15DA047610 and NIGMS grant P20GM103436. The study was also supported by a Northern Kentucky University Faculty Project Grant and a Northern Kentucky University College of Arts and Sciences Professional Development Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yates, J.R., Horchar, M.J., Ellis, A.L. et al. Differential effects of glutamate N-methyl-d-aspartate receptor antagonists on risky choice as assessed in the risky decision task. Psychopharmacology 238, 133–148 (2021). https://doi.org/10.1007/s00213-020-05664-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05664-z