Abstract
Background
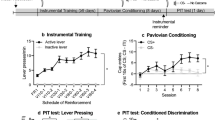
Pavlovian stimuli can influence instrumental behaviors via phenomena such as Pavlovian-to-instrumental transfer (PIT). PIT arises via dissociable processes as sensory-specific PIT (SS-PIT) and general PIT. The basolateral amygdala (BLA) mediates SS-PIT, but not general PIT. However, the specific BLA neuronal populations involved are unknown.
Aims
To determine the contribution of glutamatergic BLA neurons to the expression of SS-PIT and to the recall of sensory-specific properties of stimulus-outcome associations.
Methods
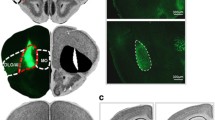
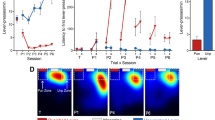
BLA neurons were transduced with virus containing either GFP or hM4Di, driven by the CamKII promoter. Rats were then tested for SS and general PIT and subsequently for expression of Pavlovian outcome devaluation effects and conditioned taste aversion following injections of vehicle or clozapine-N-oxide (CNO, the hM4Di agonist).
Results
CNO selectively blocked SS-PIT in the hM4Di-expressing group, but not controls, without altering expression of Pavlovian outcome devaluation or sensory-specific taste aversion in either group. Unexpectedly, CNO disrupted general PIT in both groups.
Conclusions
CamKII BLA neurons mediate the expression of SS-PIT by enabling Pavlovian stimuli to trigger recall of the correct action-outcome associations rather than by mediating recall of the sensory-specific properties of the stimulus-outcome association. Separately, our data demonstrate that CNO alone is sufficient to disrupt affective, but not sensory-specific processes, an effect that was not due to generalized motor disruption. This non-specific effect on general PIT may be related to CNO-induced shifts in internal state. Together, these data identify BLA CamKII neurons as critical for the expression of SS-PIT and reveal important considerations for using CNO to study general affective motivation.




Similar content being viewed by others
References
Alarcon D, Bonardi C (2016) The effect of conditioned inhibition on the specific Pavlovian-instrumental transfer effect. J Exp Psychol Anim Learn Cogn 42:82–94. https://doi.org/10.1037/xan0000087
Alarcon DE, Delamater AR (2019) Outcome-specific Pavlovian-to-instrumental transfer (PIT) with alcohol cues and its extinction. Alcohol (Fayetteville, NY) 76:131–146. https://doi.org/10.1016/j.alcohol.2018.09.003
Alarcon DE, Bonardi C, Delamater AR (2018) Associative mechanisms involved in specific Pavlovian-to-instrumental transfer in human learning tasks. Q J Exp Psychol 71:1607–1625. https://doi.org/10.1080/17470218.2017.1342671
Armbruster BN, Li X, Pausch MH, Herlitze S, Roth BL (2007) Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc Natl Acad Sci U S A 104:5163–5168. https://doi.org/10.1073/pnas.0700293104
Balleine B (1994) Asymmetrical interactions between thirst and hunger in Pavlovian-instrumental transfer. Q J Exp Psychol B 47:211–231
Baxter MG, Parker A, Lindner CC, Izquierdo AD, Murray EA (2000) Control of response selection by reinforcer value requires interaction of amygdala and orbital prefrontal cortex. J Neurosci 20:4311–4319
Berridge KC, Ho CY, Richard JM, Di Feliceantonio AG (2010) The tempted brain eats: pleasure and desire circuits in obesity and eating disorders. Brain Res 2:43–64
Blundell P, Hall G, Killcross S (2003) Preserved sensitivity to outcome value after lesions of the basolateral amygdala. J Neurosci 23:7702–7709
Boutelle KN, Bouton ME (2015) Implications of learning theory for developing programs to decrease overeating. Appetite 93:62–74. https://doi.org/10.1016/j.appet.2015.05.013
Chen X, Choo H, Huang XP, Yang X, Stone O, Roth BL, Jin J (2015) The first structure-activity relationship studies for designer receptors exclusively activated by designer drugs. ACS Chem Neurosci 6:476–484. https://doi.org/10.1021/cn500325v
Colwill RM, Motzkin DK (1994) Encoding of the unconditioned stimulus in Pavlovian conditioning. Anim Learn Behav 22:384–394. https://doi.org/10.3758/BF03209158
Corbit LH, Balleine BW (2005) Double dissociation of basolateral and central amygdala lesions on the general and outcome-specific forms of pavlovian-instrumental transfer. J Neurosci 25:962–970. https://doi.org/10.1523/jneurosci.4507-04.2005
Corbit LH, Balleine BW (2011) The general and outcome-specific forms of Pavlovian-instrumental transfer are differentially mediated by the nucleus accumbens core and shell. J Neurosci 31:11786–11794. https://doi.org/10.1523/JNEUROSCI.2711-11.2011
Corbit LH, Janak PH, Balleine BW (2007) General and outcome-specific forms of Pavlovian-instrumental transfer: the effect of shifts in motivational state and inactivation of the ventral tegmental area. Eur J Neurosci 26:3141–3149. https://doi.org/10.1111/j.1460-9568.2007.05934.x
Dagher A (2009) The neurobiology of appetite: hunger as addiction. Int J Obes (Lond) 33(Suppl 2):S30–S33. https://doi.org/10.1038/ijo.2009.69
De Tommaso M, Mastropasqua T, Turatto M (2018) Working for beverages without being thirsty: human Pavlovian-instrumental transfer despite outcome devaluation. Learn Motiv 63:37–48. https://doi.org/10.1016/j.lmot.2018.01.001
de Wit S, Dickinson A (2009) Associative theories of goal-directed behaviour: a case for animal-human translational models. Psychol Res 73:463–476. https://doi.org/10.1007/s00426-009-0230-6
Delamater AR (2012) On the nature of CS and US representations in Pavlovian learning. Learn Behav 40:1–23
Delamater AR, Holland PC (2008) The influence of CS-US interval on several different indices of learning in appetitive conditioning. J Exp Psychol Anim Behav Process 34:202–222. https://doi.org/10.1037/0097-7403.34.2.202
Delamater AR, Schneider K, Derman RC (2017) Extinction of specific stimulus-outcome (S-O) associations in Pavlovian learning with an extended CS procedure. J Exp Psychol Anim Learn Cogn 4
Derman RC, Ferrario CR (2018) Enhanced incentive motivation in obesity-prone rats is mediated by NAc core CP-AMPARs. Neuropharmacology 131:326–336
Derman RC, Ferrario CR (2019) Affective Pavlovian motivation is enhanced in obesity susceptible populations; implications for incentive motivation in obesity bioRxiv:657833. https://doi.org/10.1101/657833
Derman RC, Ferrario CR (In Press) Affective Pavlovian motivation is enhanced in obesity susceptible populations; implications for incentive motivation in obesity. Behav Brain Res
Gilroy KE, Everett EM, Delamater AR (2014) Response-outcome versus outcome-response associations in Pavlovian-to-instrumental transfer: effects of instrumental training context. Int J Comp Psychol 27:585–597
Gomez JL et al. (2017) Chemogenetics revealed: DREADD occupancy and activation via converted clozapine Science (New York) 357:503-507. https://doi.org/10.1126/science.aan2475
Gompf HS, Budygin EA, Fuller PM, Bass CE (2015) Targeted genetic manipulations of neuronal subtypes using promoter-specific combinatorial AAVs in wild-type animals. Front Behav Neurosci 9:152. https://doi.org/10.3389/fnbeh.2015.00152
Holland PC, Rescorla RA (1975) The effect of two ways of devaluing the unconditioned stimulus after first- and second-order appetitive conditioning. J Exp Psychol 1:355–363. https://doi.org/10.1037/0097-7403.1.4.355
Johnson AW, Gallagher M, Holland PC (2009) The basolateral amygdala is critical to the expression of pavlovian and instrumental outcome-specific reinforcer devaluation effects. J Neurosci 29:696–704. https://doi.org/10.1523/jneurosci.3758-08.2009
Jones EG, Huntley GW, Benson DL (1994) Alpha calcium/calmodulin-dependent protein kinase II selectively expressed in a subpopulation of excitatory neurons in monkey sensory-motor cortex: comparison with GAD-67 expression. J Neurosci 14:611–629
Konorski J (1967) Integrative activity of the brain: an interdisciplinary approach. University of Chicago Press, Chicago and London
Kosheleff AR, Araki J, Tsan L, Chen G, Murphy NP, Maidment NT, Ostlund SB (2018) Junk Food Exposure Disrupts Selection of Food-Seeking Actions in Rats. Frontiers Psychiatr 9:350. https://doi.org/10.3389/fpsyt.2018.00350
Lang EJ, Pare D (1998) Synaptic responsiveness of interneurons of the cat lateral amygdaloid nucleus. Neuroscience 83:877–889
Lichtenberg NT, Wassum KM (2017) Amygdala mu-opioid receptors mediate the motivating influence of cue-triggered reward expectations. Eur J Neurosci 45:381–387. https://doi.org/10.1111/ejn.13477
Lichtenberg NT, Pennington ZT, Holley SM, Greenfield VY, Cepeda C, Levine MS, Wassum KM (2017) Basolateral amygdala to orbitofrontal cortex projections enable cue-triggered reward Expectations. J Neurosci 37:8374–8384
Manvich DF, Webster KA, Foster SL, Farrell MS, Ritchie JC, Porter JH, Weinshenker D (2018) The DREADD agonist clozapine N-oxide (CNO) is reverse-metabolized to clozapine and produces clozapine-like interoceptive stimulus effects in rats and mice. Sci Rep 8:3840. https://doi.org/10.1038/s41598-018-22116-z
Ostlund SB, Liu AT, Wassum KM, Maidment NT (2017) Modulation of cue-triggered reward seeking by cholinergic signaling in the dorsomedial striatum. Eur J Neurosci 45:358–364. https://doi.org/10.1111/ejn.13462
Paxinos G, Watson CJ (2007) The rat brain in stereotaxic coordinates, sixth edition, 6th edn. Academic Press
Pickens CL, Saddoris MP, Setlow B, Gallagher M, Holland PC, Schoenbaum G (2003) Different roles for orbitofrontal cortex and basolateral amygdala in a reinforcer devaluation task. J Neurosci 23:11078. https://doi.org/10.1523/JNEUROSCI.23-35-11078.2003
Prager EM, Bergstrom HC, Wynn GH, Braga MF (2016) The basolateral amygdala gamma-aminobutyric acidergic system in health and disease. J Neurosci Res 94:548–567. https://doi.org/10.1002/jnr.23690
Robinson TE, Berridge KC (2008) Review. The incentive sensitization theory of addiction: some current issues. Philos Trans R Soc Lond B Biol Sci 363:3137–3146
Robinson MJ, Fischer AM, Ahuja A, Lesser EN, Maniates H (2016) Roles of “wanting” and “liking” in motivating behavior: gambling, food, and drug addictions. Curr Top Behav Neurosci 27:105–136
Sah P, Faber ES, Lopez De Armentia M, Power J (2003) The amygdaloid complex: anatomy and physiology. Physiol Rev 83:803–834. https://doi.org/10.1152/physrev.00002.2003
Shiflett MW, Balleine BW (2010) At the limbic-motor interface: disconnection of basolateral amygdala from nucleus accumbens core and shell reveals dissociable components of incentive motivation. Eur J Neurosci 32:1735–1743. https://doi.org/10.1111/j.1460-9568.2010.07439.x
Volkow ND, Wang GJ, Tomasi D, Baler RD (2013) Obesity and addiction: neurobiological overlaps. Obes Rev 14:2–18
Walker KC (1942) The effect of a discriminative stimulus transferred to a previously unassociated response. J Exp Psychol 31:312–321. https://doi.org/10.1037/h0062929
Watson P, Wiers RW, Hommel B, de Wit S (2014) Working for food you don’t desire. Cues interfere with goal-directed food-seeking. Appetite 79:139–148. https://doi.org/10.1016/j.appet.2014.04.005
Watson P, Wiers RW, Hommel B, de Wit S (2018) Motivational sensitivity of outcome-response priming: experimental research and theoretical models. Psychon Bull Rev 25:2069–2082. https://doi.org/10.3758/s13423-018-1449-2
Wellman LL, Gale K, Malkova L (2005) GABAA-mediated inhibition of basolateral amygdala blocks reward devaluation in macaques. J Neurosci 25:4577–4586. https://doi.org/10.1523/jneurosci.2257-04.2005
Woodruff AR, Sah P (2007) Networks of parvalbumin-positive interneurons in the basolateral amygdala. J Neurosci 27:553–563. https://doi.org/10.1523/jneurosci.3686-06.2007
Wyvell CL, Berridge KC (2001) Incentive sensitization by previous amphetamine exposure: increased cue-triggered “wanting” for sucrose reward. J Neurosci 21:7831. https://doi.org/10.1523/JNEUROSCI.21-19-07831.2001
Xiao X, Li J, Samulski RJ (1998) Production of high-titer recombinant adeno-associated virus vectors in the absence of helper adenovirus. J Virol 72:2224–2232
Acknowledgments
Viruses plasmids were obtained from Addgene as gifts from Bryan Roth (Addgene plasmid # 50469; http://n2t.net/addgene:50469; RRID: Addgene_50469; Addgene plasmid # 50477; http://n2t.net/addgene:50477; RRID: Addgene_50477). CNO was provided by the Drug Supply program of the National Institute on Drug Abuse.
Data accessibility
All data are freely available upon request. Please contact the corresponding author.
Funding
This work was supported by NIH-NIDDK 1F31-DK111194-01 awarded to RCD, Whitehall Foundation Grant 2017-12-98 and NIH-R21DA043190 awarded to CEB, and NIH-R01DK106188 and NIH-R01DK115526 awarded to CRF.
Author information
Authors and Affiliations
Contributions
RCD designed and executed the experiments, analyzed data, and wrote the manuscript. BEC generated the viruses and wrote the manuscript. CRF designed experiments, analyzed data, and wrote the manuscript. All authors have read and approve the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 506 kb)
Rights and permissions
About this article
Cite this article
Derman, R.C., Bass, C.E. & Ferrario, C.R. Effects of hM4Di activation in CamKII basolateral amygdala neurons and CNO treatment on sensory-specific vs. general PIT: refining PIT circuits and considerations for using CNO. Psychopharmacology 237, 1249–1266 (2020). https://doi.org/10.1007/s00213-020-05453-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05453-8