Abstract
Rationale
Oxycodone is one of the most widely prescribed painkillers in the USA. However, its use is complicated by high abuse potential. As sex differences have been described in drug addiction, the present study tested for sex differences in intravenous oxycodone self-administration in rats.
Methods
Male and female Sprague-Dawley rats were implanted with jugular vein catheters and trained to self-administer oxycodone (0.03 mg/kg/infusion) under fixed ratio 1 (FR1), FR2, and FR5 schedules of reinforcement followed by a dose-response study to assess sensitivity to the reinforcing effects of oxycodone. In separate rats, sucrose pellet self-administration was assessed under an FR1 schedule to determine whether sex differences in oxycodone self-administration could be generalized across reinforcers. In separate rats, oxycodone distribution to plasma and brain was measured after intravenous drug delivery.
Results
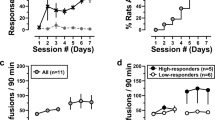
In the first 3 trials under an FR1 schedule of reinforcement, male rats self-administered more oxycodone than females. In contrast, females self-administered more sucrose pellets. Under FR2 and FR5 schedules, no significant sex differences in oxycodone intake were observed, although female rats had significantly more inactive lever presses. Male and female rats showed similar inverted U-shaped dose-effect functions, with females tending to self-administer more oxycodone than males at higher doses. No significant sex differences were observed in plasma or brain oxycodone levels, suggesting that sex differences in oxycodone self-administration behavior were not due to pharmacokinetics.
Conclusion
Our results suggest subtle sex differences in oxycodone self-administration, which may influence the abuse liability of oxycodone and have ramifications for prescription opioid addiction treatment.





Similar content being viewed by others
References
Bardo MT, Klebaur JE, Valone JM, Deaton C (2001) Environmental enrichment decreases intravenous self-administration of amphetamine in female and male rats. Psychopharmacology 155:278–284
Beardsley PM, Aceto MD, Cook CD, Bowman ER, Newman JL, Harris LS (2004) Discriminative stimulus, reinforcing, physical dependence, and antinociceptive effects of oxycodone in mice, rats, and rhesus monkeys. Exp Clin Psychopharmacol 12:163–172. doi:10.1037/1064-1297.12.3.163
Carr KD (2002) Augmentation of drug reward by chronic food restriction: behavioral evidence and underlying mechanisms. Physiol Behav 76:353–364
Carroll ME, Campbell UC, Heideman P (2001) Ketoconazole suppresses food restriction-induced increases in heroin self-administration in rats: sex differences. Exp Clin Psychopharmacol 9:307–316
Carroll ME, Morgan AD, Lynch WJ, Campbell UC, Dess NK (2002) Intravenous cocaine and heroin self-administration in rats selectively bred for differential saccharin intake: phenotype and sex differences. Psychopharmacology 161:304–313. doi:10.1007/s00213-002-1030-5
Chartoff EH, McHugh RK (2016) Translational studies of sex differences in sensitivity to opioid addiction. Neuropsychopharmacology 41:383–384. doi:10.1038/npp.2015.272
Cicero TJ, Aylward SC, Meyer ER (2003) Gender differences in the intravenous self-administration of mu opiate agonists. Pharmacol Biochem Behav 74:541–549
Collins D, Reed B, Zhang Y, Kreek MJ (2016) Sex differences in responsiveness to the prescription opioid oxycodone in mice. Pharmacol Biochem Behav 148:99–105. doi:10.1016/j.pbb.2016.06.006
Compton WM, Jones CM, Baldwin GT (2016) Relationship between nonmedical prescription-opioid use and heroin use. N Engl J Med 374:154–163. doi:10.1056/NEJMra1508490
Craft RM (2008) Sex differences in analgesic, reinforcing, discriminative, and motoric effects of opioids. Exp Clin Psychopharmacol 16:376–385. doi:10.1037/a0012931
Craft RM, Clark JL, Hart SP, Pinckney MK (2006) Sex differences in locomotor effects of morphine in the rat. Pharmacol Biochem Behav 85:850–858. doi:10.1016/j.pbb.2006.11.022
Craft RM, Kalivas PW, Stratmann JA (1996) Sex differences in discriminative stimulus effects of morphine in the rat. Behav Pharmacol 7:764–778
Craft RM, Stratmann JA, Bartok RE, Walpole TI, King SJ (1999) Sex differences in development of morphine tolerance and dependence in the rat. Psychopharmacology 143:1–7
Curtis KS, Davis LM, Johnson AL, Therrien KL, Contreras RJ (2004) Sex differences in behavioral taste responses to and ingestion of sucrose and NaCl solutions by rats. Physiol Behav 80:657–664. doi:10.1016/j.physbeh.2003.11.007
Hemsing N, Greaves L, Poole N, Schmidt R (2016) Misuse of prescription opioid medication among women: a scoping review. Pain Res Manag 2016:1754195. doi:10.1155/2016/1754195
Hernandez-Avila CA, Rounsaville BJ, Kranzler HR (2004) Opioid-, cannabis- and alcohol-dependent women show more rapid progression to substance abuse treatment. Drug Alcohol Depend 74:265–272. doi:10.1016/j.drugalcdep.2004.02.001
Hubscher CH, Brooks DL, Johnson JR (2005) A quantitative method for assessing stages of the rat estrous cycle. Biotech Histochem 80:79–87. doi:10.1080/10520290500138422
Kimishima A, Umihara H, Mizoguchi A, Yokoshima S, Fukuyama T (2014) Synthesis of (−)-oxycodone. Org Lett 16:6244–6247. doi:10.1021/ol503175n
Koob GF (1992) Drugs of abuse: anatomy, pharmacology and function of reward pathways. Trends Pharmacol Sci 13:177–184
Lu L, Shepard JD, Hall FS, Shaham Y (2003) Effect of environmental stressors on opiate and psychostimulant reinforcement, reinstatement and discrimination in rats: a review. Neurosci Biobehav Rev 27:457–491
Lynch WJ, Carroll ME (1999) Sex differences in the acquisition of intravenously self-administered cocaine and heroin in rats. Psychopharmacology 144:77–82
McHugh RK, Devito EE, Dodd D, Carroll KM, Potter JS, Greenfield SF, Connery HS, Weiss RD (2013) Gender differences in a clinical trial for prescription opioid dependence. J Subst Abus Treat 45:38–43. doi:10.1016/j.jsat.2012.12.007
Olkkola KT, Kontinen VK, Saari TI, Kalso EA (2013) Does the pharmacology of oxycodone justify its increasing use as an analgesic? Trends Pharmacol Sci 34:206–214. doi:10.1016/j.tips.2013.02.001
Piazza PV, Deminiere JM, Le Moal M, Simon H (1989) Factors that predict individual vulnerability to amphetamine self-administration. Science 245:1511–1513
Piazza PV, Deroche-Gamonent V, Rouge-Pont F, Le Moal M (2000) Vertical shifts in self-administration dose-response functions predict a drug-vulnerable phenotype predisposed to addiction. J Neurosci 20:4226–4232
Pravetoni M, Le Naour M, Harmon TM, Tucker AM, Portoghese PS, Pentel PR (2012) An oxycodone conjugate vaccine elicits drug-specific antibodies that reduce oxycodone distribution to brain and hot-plate analgesia. J Pharmacol Exp Ther 341:225–232. doi:10.1124/jpet.111.189506
Pravetoni M, Pentel PR, Potter DN, Chartoff EH, Tally L, LeSage MG (2014) Effects of an oxycodone conjugate vaccine on oxycodone self-administration and oxycodone-induced brain gene expression in rats. PLoS One 9:e101807. doi:10.1371/journal.pone.0101807
Puhl MD, Boisvert M, Guan Z, Fang J, Grigson PS (2013) A novel model of chronic sleep restriction reveals an increase in the perceived incentive reward value of cocaine in high drug-taking rats. Pharmacol Biochem Behav 109:8–15. doi:10.1016/j.pbb.2013.04.010
Russell SE, Rachlin AB, Smith KL, Muschamp J, Berry L, Zhao Z, Chartoff EH (2014) Sex differences in sensitivity to the depressive-like effects of the kappa opioid receptor agonist U-50488 in rats. Biol Psychiatry 76:213–222. doi:10.1016/j.biopsych.2013.07.042
Sclafani A, Hertwig H, Vigorito M, Feigin MB (1987) Sex differences in polysaccharide and sugar preferences in rats. Neurosci Biobehav Rev 11:241–251
Simoni-Wastila L, Ritter G, Strickler G (2004) Gender and other factors associated with the nonmedical use of abusable prescription drugs. Subst Use Misuse 39:1–23
Soderberg Lofdal KC, Andersson ML, Gustafsson LL (2013) Cytochrome P450-mediated changes in oxycodone pharmacokinetics/pharmacodynamics and their clinical implications. Drugs 73:533–543. doi:10.1007/s40265-013-0036-0
Stewart J, Woodside B, Shaham Y (1996) Ovarian hormones do not affect the initiation and maintenance of intravenous self-administration of heroin in the female rat. Psychobiology 24:154–159
Thomsen M, and Caine SB (2005) Chronic intravenous drug self-administration in rats and mice. Curr Protoc Neurosci Chapter 9, Unit 9 20. doi: 10.1002/0471142301.ns0920s32
Valenstein ES, Kakolewski JW, Cox VC (1967) Sex differences in taste preference for glucose and saccharin solutions. Science 156:942–943
van Hest A, van Haaren F, van de Poll NE (1989) Perseverative responding in male and female Wistar rats: effects of gonadal hormones. Horm Behav 23:57–67
Vander Weele CM, Porter-Stransky KA, Mabrouk OS, Lovic V, Singer BF, Kennedy RT, Aragona BJ (2014) Rapid dopamine transmission within the nucleus accumbens: dramatic difference between morphine and oxycodone delivery. Eur J Neurosci 40:3041–3054. doi:10.1111/ejn.12709
Wade CL, Vendruscolo LF, Schlosburg JE, Hernandez DO, Koob GF (2015) Compulsive-like responding for opioid analgesics in rats with extended access. Neuropsychopharmacology 40:421–428. doi:10.1038/npp.2014.188
Walker QD, Rooney MB, Wightman RM, Kuhn CM (2000) Dopamine release and uptake are greater in female than male rat striatum as measured by fast cyclic voltammetry. Neuroscience 95:1061–1070
Zacny JP, Drum M (2010) Psychopharmacological effects of oxycodone in healthy volunteers: roles of alcohol-drinking status and sex. Drug Alcohol Depend 107:209–214. doi:10.1016/j.drugalcdep.2009.10.012
Zhan B, Ma HY, Wang JL, Liu CB (2015) Sex differences in morphine-induced behavioral sensitization and social behaviors in ICR mice. Dongwuxue Yanjiu 36:103–108
Zhang Y, Mayer-Blackwell B, Schlussman SD, Randesi M, Butelman ER, Ho A, Ott J, Kreek MJ (2014) Extended access oxycodone self-administration and neurotransmitter receptor gene expression in the dorsal striatum of adult C57BL/6 J mice. Psychopharmacology 231:1277–1287. doi:10.1007/s00213-013-3306-3
Zhou L, Ghee SM, See RE, Reichel CM (2015) Oxytocin differentially affects sucrose taking and seeking in male and female rats. Behav Brain Res 283:184–190. doi:10.1016/j.bbr.2015.01.050
Acknowledgments
This study was supported by the Brooking Fellowship (MM). We thank Dr. Linda Valerie for the critical assistance with the statistical analysis of the results of this study. We also thank Dr. Maria Carreira for training MM in the jugular vein catheterization and relevant self-administration procedures, Dr. In-Jee You for the recommendations for setting up the drug self-administration paradigm, and Dr. Barak Caine and Dr. Morgane Thomsen for commenting on our data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All guidelines recommended by the Animal Care and Use Committee of McLean Hospital were followed.
ᅟ
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mavrikaki, M., Pravetoni, M., Page, S. et al. Oxycodone self-administration in male and female rats. Psychopharmacology 234, 977–987 (2017). https://doi.org/10.1007/s00213-017-4536-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4536-6