Abstract
Rationale
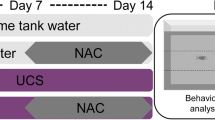
Several model organisms have been employed to study the impacts of stress on biological systems. Different models of unpredictable chronic stress (UCS) have been established in rodents; however, these protocols are expensive, long-lasting, and require a large physical structure. Our group has recently reported an UCS protocol in zebrafish with several advantages compared to rodent models. We observed that UCS induced behavioral, biochemical, and molecular changes similar to those observed in depressed patients, supporting the translational relevance of the protocol.
Objectives
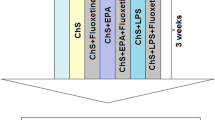
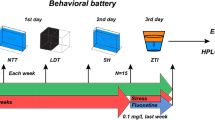
Considering that a pharmacological assessment is lacking in this zebrafish model, our aim was to evaluate the effects of anxiolytic (bromazepam) and antidepressant drugs (fluoxetine and nortriptyline) on behavioral (novel tank test), biochemical (whole-body cortisol), and molecular parameters (cox-2, tnf-α, il-6, and il-10 gene expression) in zebrafish subjected to UCS.
Results
We replicated previous data showing that UCS induces behavioral and neuroendocrine alterations in zebrafish, and we show for the first time that anxiolytic and antidepressant drugs are able to prevent such effects. Furthermore, we extended the molecular characterization of the model, revealing that UCS increases expression of the pro-inflammatory markers cox-2 and il-6, which was also prevented by the drugs tested.
Conclusions
This study reinforces the use of zebrafish as a model organism to study the behavioral and physiological effects of stress. The UCS protocol may also serve as a screening tool for evaluating new drugs that can be used to treat psychiatric disorders with stress-related etiologies.




Similar content being viewed by others
References
Banasr M, Valentine GW, Li X-Y, et al. (2007) Chronic unpredictable stress decreases cell proliferation in the cerebral cortex of the adult rat. Biol Psychiatry 62:496–504. doi:10.1016/j.biopsych.2007.02.006
Bartholomä P, Erlandsson N, Kaufmann K, et al. (2002) Neuronal cell death induced by antidepressants: lack of correlation with Egr-1, NF-kappa B and extracellular signal-regulated protein kinase activation. Biochem Pharmacol 63:1507–1516
Beesdo K, Pine DS, Lieb R, Wittchen H-U (2010) Incidence and risk patterns of anxiety and depressive disorders and categorization of generalized anxiety disorder. Arch Gen Psychiatry 67:47–57. doi:10.1001/archgenpsychiatry.2009.177
Belzer K, Schneier FR (2004) Comorbidity of anxiety and depressive disorders: issues in conceptualization, assessment, and treatment. J Psychiatr Pract 10:296–306
Borsini F, Podhorna J, Marazziti D (2002) Do animal models of anxiety predict anxiolytic-like effects of antidepressants? Psychopharmacology 163:121–141. doi:10.1007/s00213-002-1155-6
Bustin SA, Benes V, Garson J, et al. (2013) The need for transparency and good practices in the qPCR literature. Nat Methods 10:1063–1067. doi:10.1038/nmeth.2697
Calabrese JR, Skwerer RG, Barna B, et al. (1986) Depression, immunocompetence, and prostaglandins of the E series. Psychiatry Res 17:41–47
Castanon N, Médina C, Mormède C, Dantzer R (2004) Chronic administration of tianeptine balances lipopolysaccharide-induced expression of cytokines in the spleen and hypothalamus of rats. Psychoneuroendocrinology 29:778–790. doi:10.1016/S0306-4530(03)00142-2
Chakravarty S, Reddy BR, Sudhakar SR, et al. (2013) Chronic unpredictable stress (CUS)-induced anxiety and related mood disorders in a zebrafish model: altered brain proteome profile implicates mitochondrial dysfunction. PLoS One 8:e63302. doi:10.1371/journal.pone.0063302
Crowley SK, Girdler SS (2014) Neurosteroid, GABAergic and hypothalamic pituitary adrenal (HPA) axis regulation: what is the current state of knowledge in humans? Psychopharmacology 231:3619–3634. doi:10.1007/s00213-014-3572-8
D’Aquila PS, Brain P, Willner P (1994) Effects of chronic mild stress on performance in behavioural tests relevant to anxiety and depression. Physiol Behav 56:861–867
Dantzer R (2006) Cytokine, sickness behavior, and depression. Neurol Clin 24:441–460. doi:10.1016/j.ncl.2006.03.003
Dantzer R, Kelley KW (2007) Twenty years of research on cytokine-induced sickness behavior. Brain Behav Immun 21:153–160. doi:10.1016/j.bbi.2006.09.006
Djavadian RL (2004) Serotonin and neurogenesis in the hippocampal dentate gyrus of adult mammals. Acta Neurobiol Exp (Warsz) 64:189–200
Dowlati Y, Herrmann N, Swardfager W, et al. (2010) A meta-analysis of cytokines in major depression. Biol Psychiatry 67:446–457. doi:10.1016/j.biopsych.2009.09.033
Egan RJ, Bergner CL, Hart PC, et al. (2009) Understanding behavioral and physiological phenotypes of stress and anxiety in zebrafish. Behav Brain Res 205:38–44. doi:10.1016/j.bbr.2009.06.022
Eyre HA, Air T, Proctor S, et al. (2015) A critical review of the efficacy of non-steroidal anti-inflammatory drugs in depression. Prog Neuro-Psychopharmacol Biol Psychiatry 57:11–16. doi:10.1016/j.pnpbp.2014.10.003
Faikoh EN, Hong Y-H, S-Y H (2014) Liposome-encapsulated cinnamaldehyde enhances zebrafish (Danio rerio) immunity and survival when challenged with Vibrio Vulnificus and Streptococcus agalactiae. Fish Shellfish Immunol 38:15–24. doi:10.1016/j.fsi.2014.02.024
Gałecki P, Talarowska M, Bobińska K, Szemraj J (2014) COX-2 gene expression is correlated with cognitive function in recurrent depressive disorder. Psychiatry Res 215:488–490. doi:10.1016/j.psychres.2013.12.017
Global Burden of Disease Study 2013 Collaborators (2015) Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the global burden of disease study 2013. Lancet. doi:10.1016/S0140-6736(15)60692-4
Gray VC, Hughes RN (2015) Drug-, dose- and sex-dependent effects of chronic fluoxetine, reboxetine and venlafaxine on open-field behavior and spatial memory in rats. Behav Brain Res 281:43–54. doi:10.1016/j.bbr.2014.12.023
Hanisch U-K, Kettenmann H (2007) Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci 10:1387–1394. doi:10.1038/nn1997
Hannestad J, DellaGioia N, Bloch M (2011) The effect of antidepressant medication treatment on serum levels of inflammatory cytokines: a meta-analysis. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 36:2452–2459. doi:10.1038/npp.2011.132
Haroon E, Raison CL, Miller AH (2012) Psychoneuroimmunology meets neuropsychopharmacology: translational implications of the impact of inflammation on behavior. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 37:137–162. doi:10.1038/npp.2011.205
Herculano AM, Maximino C (2014) Serotonergic modulation of zebrafish behavior: towards a paradox. Prog Neuro-Psychopharmacol Biol Psychiatry 55:50–66. doi:10.1016/j.pnpbp.2014.03.008
Hinson RM, Williams JA, Shacter E (1996) Elevated interleukin 6 is induced by prostaglandin E2 in a murine model of inflammation: possible role of cyclooxygenase-2. Proc Natl Acad Sci U S A 93:4885–4890
Hou R, Baldwin DS (2012) A neuroimmunological perspective on anxiety disorders. Hum Psychopharmacol 27:6–14. doi:10.1002/hup.1259
Kenis G, Maes M (2002) Effects of antidepressants on the production of cytokines. Int J Neuropsychopharmacol Off Sci J Coll Int Neuropsychopharmacol CINP 5:401–412. doi:10.1017/S1461145702003164
Kessler RC, Chiu WT, Demler O, et al. (2005) Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry 62:617–627. doi:10.1001/archpsyc.62.6.617
Kubera M, Kenis G, Bosmans E, et al. (2004) Stimulatory effect of antidepressants on the production of IL-6. Int Immunopharmacol 4:185–192. doi:10.1016/j.intimp.2003.11.006
Kubera M, Obuchowicz E, Goehler L, et al. (2011) In animal models, psychosocial stress-induced (neuro)inflammation, apoptosis and reduced neurogenesis are associated to the onset of depression. Prog Neuro-Psychopharmacol Biol Psychiatry 35:744–759. doi:10.1016/j.pnpbp.2010.08.026
Leite CE, Maboni Lde O, Cruz FF, Rosemberg DB, Zimmermann FF, Pereira TC, Bogo MR, Bonan CD, Campos MM, Morrone FB, Battastini AM (2013) Involvement of purinergic system in inflammation and toxicity induced by copper in zebrafish larvae. Toxicol Appl Pharmacol 272(3):681–689. doi:10.1016/j.taap.2013.08.001
Leonard BE (2014) Impact of inflammation on neurotransmitter changes in major depression: an insight into the action of antidepressants. Prog Neuro-Psychopharmacol Biol Psychiatry 48:261–267. doi:10.1016/j.pnpbp.2013.10.018
Leonard B, Maes M (2012) Mechanistic explanations how cell-mediated immune activation, inflammation and oxidative and nitrosative stress pathways and their sequels and concomitants play a role in the pathophysiology of unipolar depression. Neurosci Biobehav Rev 36:764–785. doi:10.1016/j.neubiorev.2011.12.005
Levin ED, Bencan Z, Cerutti DT (2007) Anxiolytic effects of nicotine in zebrafish. Physiol Behav 90:54–58. doi:10.1016/j.physbeh.2006.08.026
Li R, Zhao D, Qu R, et al. (2015) The effects of apigenin on lipopolysaccharide-induced depressive-like behavior in mice. Neurosci Lett 594:17–22. doi:10.1016/j.neulet.2015.03.040
Maes M (2011) Depression is an inflammatory disease, but cell-mediated immune activation is the key component of depression. Prog Neuro-Psychopharmacol Biol Psychiatry 35:664–675. doi:10.1016/j.pnpbp.2010.06.014
Maes M, Kubera M, Obuchowiczwa E, et al. (2011) Depression’s multiple comorbidities explained by (neuro) inflammatory and oxidative & nitrosative stress pathways. Neuro Endocrinol Lett 32:7–24
Manuel R, Gorissen M, Zethof J, et al. (2014) Unpredictable chronic stress decreases inhibitory avoidance learning in Tuebingen long-fin zebrafish: stronger effects in the resting phase than in the active phase. J Exp Biol 217:3919–3928. doi:10.1242/jeb.109736
Martinowich K, Lu B (2007) Interaction between BDNF and serotonin: role in mood disorders. Neuropsychopharmacology 33:73–83. doi:10.1038/sj.npp.1301571
McEwen BS (2007) Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev 87:873–904. doi:10.1152/physrev.00041.2006
McEwen BS, Brinton RE, Sapolsky RM (1988) Glucocorticoid receptors and behavior: implications for the stress response. Adv Exp Med Biol 245:35–45
McEwen BS, Bowles NP, Gray JD, et al. (2015) Mechanisms of stress in the brain. Nat Neurosci 18:1353–1363. doi:10.1038/nn.4086
Miller AH, Maletic V, Raison CL (2009) Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry 65:732–741. doi:10.1016/j.biopsych.2008.11.029
Mineur YS, Belzung C, Crusio WE (2006) Effects of unpredictable chronic mild stress on anxiety and depression-like behavior in mice. Behav Brain Res 175:43–50. doi:10.1016/j.bbr.2006.07.029
Müller N, Myint A-M, Schwarz MJ (2009) The impact of neuroimmune dysregulation on neuroprotection and neurotoxicity in psychiatric disorders—relation to drug treatment. Dialogues Clin Neurosci 11:319–332
Muscat R, Papp M, Willner P (1992) Reversal of stress-induced anhedonia by the atypical antidepressants, fluoxetine and maprotiline. Psychopharmacology 109:433–438
Myint AM, O’Mahony S, Kubera M, et al. (2007) Role of paroxetine in interferon-alpha-induced immune and behavioural changes in male Wistar rats. J Psychopharmacol Oxf Engl 21:843–850. doi:10.1177/0269881107077165
Nazimek K, Kozlowski M, Bryniarski P, et al. (2016a) Repeatedly administered antidepressant drugs modulate humoral and cellular immune response in mice through action on macrophages. Exp Biol Med Maywood NJ 241:1540–1550. doi:10.1177/1535370216643769
Nazimek K, Strobel S, Bryniarski P, et al. (2016b) The role of macrophages in anti-inflammatory activity of antidepressant drugs. Immunobiology. doi:10.1016/j.imbio.2016.07.001
O’Sullivan JB, Ryan KM, Curtin NM, et al. (2009) Noradrenaline reuptake inhibitors limit neuroinflammation in rat cortex following a systemic inflammatory challenge: implications for depression and neurodegeneration. Int J Neuropsychopharmacol Off Sci J Coll Int Neuropsychopharmacol CINP 12:687–699. doi:10.1017/S146114570800967X
Pace TWW, Hu F, Miller AH (2007) Cytokine-effects on glucocorticoid receptor function: relevance to glucocorticoid resistance and the pathophysiology and treatment of major depression. Brain Behav Immun 21:9–19. doi:10.1016/j.bbi.2006.08.009
Pavlidis M, Theodoridi A, Tsalafouta A (2015) Neuroendocrine regulation of the stress response in adult zebrafish, Danio rerio. Prog Neuro-Psychopharmacol Biol Psychiatry 60:121–131. doi:10.1016/j.pnpbp.2015.02.014
Piato ÂL, Capiotti KM, Tamborski AR, et al. (2011) Unpredictable chronic stress model in zebrafish (Danio rerio): behavioral and physiological responses. Prog Neuro-Psychopharmacol Biol Psychiatry 35:561–567. doi:10.1016/j.pnpbp.2010.12.018
Piwowarska J, Chimiak A, Matsumoto H, et al. (2012) Serum cortisol concentration in patients with major depression after treatment with fluoxetine. Psychiatry Res 198:407–411. doi:10.1016/j.psychres.2012.01.029
Pomara N, Willoughby LM, Sidtis JJ, et al. (2005) Cortisol response to diazepam: its relationship to age, dose, duration of treatment, and presence of generalized anxiety disorder. Psychopharmacology 178:1–8. doi:10.1007/s00213-004-1974-8
Reichenberg A, Yirmiya R, Schuld A, et al. (2001) Cytokine-associated emotional and cognitive disturbances in humans. Arch Gen Psychiatry 58:445–452
Santarelli L, Saxe M, Gross C, et al. (2003) Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 301:805–809. doi:10.1126/science.1083328
Sapolsky RM (1982) The endocrine stress-response and social status in the wild baboon. Horm Behav 16:279–292
Sapolsky RM, Krey LC, McEwen BS (1984) Stress down-regulates corticosterone receptors in a site-specific manner in the brain. Endocrinology 114:287–292. doi:10.1210/endo-114-1-287
Schaefer IC, Siebel AM, Piato AL, et al. (2015) The side-by-side exploratory test: a simple automated protocol for the evaluation of adult zebrafish behavior simultaneously with social interaction. Behav Pharmacol 26:691–696. doi:10.1097/FBP.0000000000000145
Schiepers OJG, Wichers MC, Maes M (2005) Cytokines and major depression. Prog Neuro-Psychopharmacol Biol Psychiatry 29:201–217. doi:10.1016/j.pnpbp.2004.11.003
Selye H (1936) A syndrome produced by diverse nocuous agents. Nature 138:32
Song C, Horrobin DF, Leonard BE (2006) The comparison of changes in behavior, neurochemistry, endocrine, and immune functions after different routes, doses and durations of administrations of IL-1beta in rats. Pharmacopsychiatry 39:88–99. doi:10.1055/s-2006-941557
Tang R, Dodd A, Lai D, et al. (2007) Validation of zebrafish (Danio rerio) reference genes for quantitative real-time RT-PCR normalization. Acta Biochim Biophys Sin 39:384–390
Tovote P, Fadok JP, Lüthi A (2015) Neuronal circuits for fear and anxiety. Nat Rev Neurosci 16:317–331. doi:10.1038/nrn3945
Troib A, Azab AN (2015) Effects of psychotropic drugs on nuclear factor kappa B. Eur Rev Med Pharmacol Sci 19:1198–1208
Varela M, Dios S, Novoa B, Figueras A (2012) Characterisation, expression and ontogeny of interleukin-6 and its receptors in zebrafish (Danio rerio. Dev Comp Immunol 37:97–106. doi:10.1016/j.dci.2011.11.004
Wang J-M, Yang L-H, Zhang Y-Y, et al. (2015) BDNF and COX-2 participate in anti-depressive mechanisms of catalpol in rats undergoing chronic unpredictable mild stress. Physiol Behav 151:360–368. doi:10.1016/j.physbeh.2015.08.008
Willner P (1997) Validity, reliability and utility of the chronic mild stress model of depression: a 10-year review and evaluation. Psychopharmacology 134:319–329
Willner P (2005) Chronic mild stress (CMS) revisited: consistency and behavioural-neurobiological concordance in the effects of CMS. Neuropsychobiology 52:90–110. doi:10.1159/000087097
Willner P, Muscat R, Papp M (1992) Chronic mild stress-induced anhedonia: a realistic animal model of depression. Neurosci Biobehav Rev 16:525–534
Yalcin I, Belzung C, Surget A (2008) Mouse strain differences in the unpredictable chronic mild stress: a four-antidepressant survey. Behav Brain Res 193:140–143. doi:10.1016/j.bbr.2008.04.021
Zhao Y, Wang Z, Dai J, et al. (2012) Beneficial effects of benzodiazepine diazepam on chronic stress-induced impairment of hippocampal structural plasticity and depression-like behavior in mice. Behav Brain Res 228:339–350. doi:10.1016/j.bbr.2011.12.013
Zimmermann FF, Altenhofen S, Kist LW, et al. (2015) Unpredictable chronic stress alters adenosine metabolism in zebrafish brain. Mol Neurobiol. doi:10.1007/s12035-015-9270-7
Acknowledgments
This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico-Brazil (CNPq, Proc. 472715/2012-7). L.W.K. is the recipient of fellowship CAPES/PNPD Program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Matheus Marcon and Ana P. Herrmann contributed equally for this article.
Rights and permissions
About this article
Cite this article
Marcon, M., Herrmann, A.P., Mocelin, R. et al. Prevention of unpredictable chronic stress-related phenomena in zebrafish exposed to bromazepam, fluoxetine and nortriptyline. Psychopharmacology 233, 3815–3824 (2016). https://doi.org/10.1007/s00213-016-4408-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4408-5