Abstract
Rationale
Upregulation of α4β2* nicotinic acetylcholine receptors (nAChRs) is one of the most well-established effects of chronic cigarette smoking on the brain. Prior research by our group gave a preliminary indication that cigarette smokers with concomitant use of caffeine or marijuana have altered nAChR availability.
Objective
We sought to determine if smokers with heavy caffeine or marijuana use have different levels of α4β2* nAChRs than smokers without these drug usages.
Methods
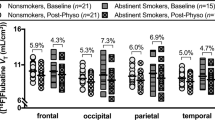
One hundred and one positron emission tomography (PET) scans, using the radiotracer 2-FA (a ligand for β2*-containing nAChRs), were obtained from four groups of males: non-smokers without heavy caffeine or marijuana use, smokers without heavy caffeine or marijuana use, smokers with heavy caffeine use (mean four coffee cups per day), and smokers with heavy marijuana use (mean 22 days of use per month). Total distribution volume (Vt/fp) was determined for the brainstem, prefrontal cortex, and thalamus, as a measure of nAChR availability.
Results
A significant between-group effect was found, resulting from the heavy caffeine and marijuana groups having the highest Vt/fp values (especially for the brainstem and prefrontal cortex), followed by smokers without such use, followed by non-smokers. Direct between-group comparisons revealed significant differences for Vt/fp values between the smoker groups with and without heavy caffeine or marijuana use.
Conclusions
Smokers with heavy caffeine or marijuana use have higher α4β2* nAChR availability than smokers without these drug usages. These findings are likely due to increased nicotine exposure but could also be due to an interaction on a cellular/molecular level.


Similar content being viewed by others
References
Agrawal A, Budney AJ, Lynskey MT (2012) The co-occurring use and misuse of cannabis and tobacco: a review. Addiction 107:1221–33
Badiani A, Boden JM, De Pirro S, Fergusson DM, Horwood LJ, Harold GT (2015) Tobacco smoking and cannabis use in a longitudinal birth cohort: evidence of reciprocal causal relationships. Drug Alcohol Depend 150:69–76
Bartal M (2001) Health effects of tobacco use and exposure. Monaldi ArchChest Dis 56:545–554
Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J (1961) An inventory for measuring depression. Arch Gen Psychiatry 4:561–571
Benwell ME, Balfour DJK, Anderson JM (1988) Evidence that tobacco smoking increases the density of (−)-[3 H]nicotine binding sites in human brain. J Neurochem 50:1243–1247
Bowes L, Chollet A, Fombonne E, Melchior M (2015) Psychological, social and familial factors associated with tobacco cessation among young adults. Eur Addict Res 21:153–9
Breese CR, Marks MJ, Logel J, Adams CE, Sullivan B, Collins AC, Leonard S (1997) Effect of smoking history on [3H]nicotine binding in human postmortem brain. J Pharmacol Exp Ther 282:7–13
Brody AL, Mandelkern MA, London ED, Olmstead RE, Farahi J, Scheibal D, Jou J, Allen V, Tiongson E, Chefer SI, Koren AO, Mukhin AG (2006) Cigarette smoking saturates brain alpha4beta2 nicotinic acetylcholine receptors. Arch Gen Psychiatry 63:907–915
Brody AL, Mandelkern MA, Costello MR, Abrams AL, Scheibal D, Farahi J, London ED, Olmstead RE, Rose JE, Mukhin AG (2009) Brain nicotinic acetylcholine receptor occupancy: effect of smoking a denicotinized cigarette. Int J Neuropsychopharmacol 12:305–316
Brody AL, Mandelkern MA, London ED, Khan A, Kozman D, Costello MR, Vellios EE, Archie MM, Bascom R, Mukhin AG (2011) Effect of secondhand smoke on occupancy of nicotinic acetylcholine receptors in brain. Arch Gen Psychiatry 68:953–60
Brody AL, Mukhin AG, La Charite J, Ta K, Farahi J, Sugar CA, Mamoun MS, Vellios E, Archie M, Kozman M, Phuong J, Arlorio F, Mandelkern MA (2013) Up-regulation of nicotinic acetylcholine receptors in menthol cigarette smokers. Int J Neuropsychopharmacol 16:957–66
Brody AL, Mukhin AG, Mamoun MS, Luu T, Neary M, Liang L, Shieh J, Sugar CA, Rose JE, Mandelkern MA (2014) Brain nicotinic acetylcholine receptor availability and response to smoking cessation treatment: a randomized trial. JAMA Psychiatry 71:797–805
Brown DW (2009) Smoking Prevalence among US Veterans. J Gen Intern Med 25:147–9
Cdc (2008) Cigarette smoking among adults--United States, 2007. MMWR 57:1221–1226
Cosgrove KP, Batis J, Bois F, Maciejewski PK, Esterlis I, Kloczynski T, Stiklus S, Krishnan-Sarin S, O’Malley S, Perry E, Tamagnan G, Seibyl JP, Staley JK (2009) beta2-Nicotinic acetylcholine receptor availability during acute and prolonged abstinence from tobacco smoking. Arch Gen Psychiatry 66:666–76
Dani JA, Harris RA (2005) Nicotine addiction and comorbidity with alcohol abuse and mental illness. Nat Neurosci 8:1465–70
Dolle F, Valette H, Bottlaender M, Hinnen F, Vaufrey F, Guenther I, Crouzel C (1998) Synthesis of 2-[F-18]fluoro-3-[2(S)-2-azetidinylmethoxy]pyridine, a highly potent radioligand for in vivo imaging central nicotinic acetylcholine receptors. J Label Compd Radiopharm 41:451–463
El-Mas MM, El-Gowilly SM, Fouda MA, Saad EI (2011) Role of adenosine A2A receptor signaling in the nicotine-evoked attenuation of reflex cardiac sympathetic control. Toxicol Appl Pharmacol 254:229–37
Esterlis I, Cosgrove KP, Batis JC, Bois F, Stiklus SM, Perkins E, Seibyl JP, Carson RE, Staley JK (2010) Quantification of smoking-induced occupancy of beta2-nicotinic acetylcholine receptors: estimation of nondisplaceable binding. J Nucl Med 51:1226–33
Ezekiel F, Bosma R, Morton JB (2013) Dimensional change card sort performance associated with age-related differences in functional connectivity of lateral prefrontal cortex. Dev Cogn Neurosci 5:40–50
Fagerstrom KO (1978) Measuring the degree of physical dependence to tobacco smoking with reference to individualization of treatment. Addict Behav 3:235–241
Ford DE, Vu HT, Anthony JC (2002) Marijuana use and cessation of tobacco smoking in adults from a community sample. Drug Alcohol Depend 67:243–8
Gil E, Chen B, Kleerup E, Webber M, Tashkin DP (1995) Acute and chronic effects of marijuana smoking on pulmonary alveolar permeability. Life Sci 56:2193–9
Goren A, Annunziata K, Schnoll RA, Suaya JA (2014) Smoking cessation and attempted cessation among adults in the United States. PLoS One 9, e93014
Goya-Maldonado R, Weber K, Trost S, Diekhof E, Keil M, Dechent P, Gruber O (2015) Dissociating pathomechanisms of depression with fMRI: bottom-up or top-down dysfunctions of the reward system. Eur Arch Psychiatry Clin Neurosci 265:57–66
Hamilton M (1967) Development of a rating scale for primary depressive illness. Br J Soc Psychol 6:278–296
Hamilton M (1969) Diagnosis and rating of anxiety. Br J Psychiatry 3:76–79
Heatherton TF, Kozlowski LT, Frecker RC, Fagerström KO (1991) The Fagerström Test for Nicotine Dependence: a revision of the Fagerström Tolerance Questionnaire. Br J Addict 86:1119–1127
Innis RB, Cunningham VJ, Delforge J, Fujita M, Gjedde A, Gunn RN, Holden J, Houle S, Huang SC, Ichise M, Iida H, Ito H, Kimura Y, Koeppe RA, Knudsen GM, Knuuti J, Lammertsma AA, Laruelle M, Logan J, Maguire RP, Mintun MA, Morris ED, Parsey R, Price JC, Slifstein M, Sossi V, Suhara T, Votaw JR, Wong DF, Carson RE (2007) Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab 27:1533–1539
Jacob P 3rd, Yu L, Wilson M, Benowitz NL (1991) Selected ion monitoring method for determination of nicotine, cotinine and deuterium-labeled analogs: absence of an isotope effect in the clearance of (S)-nicotine-3′,3′-d2 in humans. Biol Mass Spectrom 20:247–52
Justinova Z, Ferre S, Barnes C, Wertheim CE, Pappas LA, Goldberg SR, Le Foll B (2009) Effects of chronic caffeine exposure on adenosinergic modulation of the discriminative-stimulus effects of nicotine, methamphetamine, and cocaine in rats. Psychopharmacology (Berl) 203:355–67
Kimes AS, Chefer SI, Matochik JA, Contoreggi CS, Vaupel DB, Stein EA, Mukhin AG (2008) Quantification of nicotinic acetylcholine receptors in the human brain with PET: bolus plus infusion administration of 2-[18F]F-A85380. Neuroimage 39:717–727
Koob GF, Volkow ND (2010) Neurocircuitry of addiction. Neuropsychopharmacology 35:217–38
Kordosky-Herrera K, Grow WA (2009) Caffeine and nicotine decrease acetylcholine receptor clustering in C2C12 myotube culture. Cell Tissue Res 335:341–8
Koren AO, Horti AG, Mukhin AG, Gundisch D, Kimes AS, Dannals RF, London ED (1998) 2-, 5-, and 6-halo-3-(2(S)-azetidinylmethoxy)pyridines: Synthesis, affinity for nicotinic acetylcholine receptors, and molecular modeling. J Med Chem 41:3690–3698
Lasser K, Boyd JW, Woolhandler S, Himmelstein DU, McCormick D, Bor DH (2000) Smoking and mental illness - A population-based prevalence study. JAMA 284:2606–2610
Leistikow BN (2000) The human and financial costs of smoking. ClinChest Med 21: 189-1xi.
Leistikow BN, Miller TR (1998) The health care costs of smoking. N Engl J Med 338:471
Leistikow BN, Martin DC, Milano CE (2000) Fire injuries, disasters, and costs from cigarettes and cigarette lights: a global overview. Prev Med 31:91–99
Liu X, Jernigan C (2012) Effects of caffeine on persistence and reinstatement of nicotine-seeking behavior in rats: interaction with nicotine-associated cues. Psychopharmacology (Berl) 220:541–50
Mahgoub M, Keun-Hang SY, Sydorenko V, Ashoor A, Kabbani N, Al Kury L, Sadek B, Howarth CF, Isaev D, Galadari S, Oz M (2013) Effects of cannabidiol on the function of alpha7-nicotinic acetylcholine receptors. Eur J Pharmacol 720:310–9
Mamede M, Ishizu K, Ueda M, Mukai T, Iida Y, Kawashima H, Fukuyama H, Togashi K, Saji H (2007) Temporal change in human nicotinic acetylcholine receptor after smoking cessation: 5IA SPECT study. J Nucl Med 48:1829–1835
Mokdad AH, Marks JS, Stroup DF, Gerberding JL (2004) Actual causes of death in the United States, 2000. JAMA 291:1238–1245
Mukhin AG, Kimes AS, Chefer SI, Matochik JA, Contoreggi CS, Horti AG, Vaupel DB, Pavlova O, Stein EA (2008) Greater nicotinic acetylcholine receptor density in smokers than in nonsmokers: a PET study with 2-18F-FA-85380. J Nucl Med 49:1628–35
Pauly JR, Stitzel JA, Marks MJ, Collins AC (1989) An autoradiographic analysis of cholinergic receptors in mouse brain. Brain ResBull 22:453–459
Pauly JR, Marks MJ, Robinson SF, van de Kamp JL, Collins AC (1996) Chronic nicotine and mecamylamine treatment increase brain nicotinic receptor binding without changing alpha 4 or beta 2 mRNA levels. J Pharmacol Exp Ther 278:361–369
Pecina M, Martinez-Jauand M, Love T, Heffernan J, Montoya P, Hodgkinson C, Stohler CS, Goldman D, Zubieta JK (2014) Valence-specific effects of BDNF Val66Met polymorphism on dopaminergic stress and reward processing in humans. J Neurosci 34:5874–81
Pistillo F, Fasoli F, Moretti M, McClure-Begley T, Zoli M, Marks MJ, Gotti C (2016) Chronic nicotine and withdrawal affect glutamatergic but not nicotinic receptor expression in the mesocorticolimbic pathway in a region-specific manner. Pharmacol Res 103:167–76
Rabin RA, George TP (2015) A review of co-morbid tobacco and cannabis use disorders: possible mechanisms to explain high rates of co-use. Am J Addict 24:105–16
Research RD (2008) Survey of 1,095 U.S. Adults
Rezvani AH, Sexton HG, Johnson J, Wells C, Gordon K, Levin ED (2013) Effects of caffeine on alcohol consumption and nicotine self-administration in rats. Alcohol Clin Exp Res 37:1609–17
Shoaib M, Schindler CW, Goldberg SR, Pauly JR (1997) Behavioural and biochemical adaptations to nicotine in rats: influence of MK801. Psychopharm (Berl) 134:121–130
Shumway DA, Pavlova OA, Mukhin AG (2007) A simplified method for the measurement of nonmetabolized 2-[18F]F-A-85380 in blood plasma using solid-phase extraction. Nucl Med Biol 34:221–8
Singh S, Singh K, Patel S, Patel DK, Singh C, Nath C, Singh MP (2008) Nicotine and caffeine-mediated modulation in the expression of toxicant responsive genes and vesicular monoamine transporter-2 in 1-methyl 4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson’s disease phenotype in mouse. Brain Res 1207:193–206
Sorger D, Becker GA, Patt M, Schildan A, Grossmann U, Schliebs R, Seese A, Kendziorra K, Kluge M, Brust P, Mukhin AG, Sabri O (2007) Measurement of the alpha4beta2* nicotinic acetylcholine receptor ligand 2-[(18)F]Fluoro-A-85380 and its metabolites in human blood during PET investigation: a methodological study. Nucl Med Biol 34:331–42
Staley JK, Krishnan-Sarin S, Cosgrove KP, Krantzler E, Frohlich E, Perry E, Dubin JA, Estok K, Brenner E, Baldwin RM, Tamagnan GD, Seibyl JP, Jatlow P, Picciotto MR, London ED, O’Malley S, van Dyck CH (2006) Human tobacco smokers in early abstinence have higher levels of beta2* nicotinic acetylcholine receptors than nonsmokers. J Neurosci 26:8707–8714
Subramaniyan M, Dani JA (2015) Dopaminergic and cholinergic learning mechanisms in nicotine addiction. Ann N Y Acad Sci 1349:46–63
Tanji J, Hoshi E (2008) Role of the lateral prefrontal cortex in executive behavioral control. Physiol Rev 88:37–57
Thany SH, Courjaret R, Lapied B (2008) Effect of calcium on nicotine-induced current expressed by an atypical alpha-bungarotoxin-insensitive nAChR2. Neurosci Lett 438:317–21
Wager TD, Spicer J, Insler R, Smith EE (2014) The neural bases of distracter-resistant working memory. Cogn Affect Behav Neurosci 14:90–105
Wallace TL, Bertrand D (2013) Importance of the nicotinic acetylcholine receptor system in the prefrontal cortex. Biochem Pharmacol 85:1713–20
Westmaas JL, Langsam K (2005) Unaided smoking cessation and predictors of failure to quit in a community sample: effects of gender. Addict Behav 30:1405–24
Wullner U, Gundisch D, Herzog H, Minnerop M, Joe A, Warnecke M, Jessen F, Schutz C, Reinhardt M, Eschner W, Klockgether T, Schmaljohann J (2008) Smoking upregulates alpha4beta2* nicotinic acetylcholine receptors in the human brain. Neurosci Lett 430:34–7
Yates SL, Bencherif M, Fluhler EN, Lippiello PM (1995) Up-regulation of nicotinic acetylcholine receptors following chronic exposure of rats to mainstream cigarette smoke or alpha 4 beta 2 receptors to nicotine. Biochem Pharmacol 50:2001–2008
Zhang X, Tian JY, Svensson AL, Gong ZH, Meyerson B, Nordberg A (2002) Chronic treatments with tacrine and (−)-nicotine induce different changes of nicotinic and muscarinic acetylcholine receptors in the brain of aged rat. J Neural Transm 109:377–392
Acknowledgments
The authors thank the PET and MRI technicians at the VA Greater Los Angeles Healthcare System for assistance in acquiring brain images for this study. The authors also thank the laboratory of Payton Jacob III at the University of California at San Francisco for determining plasma nicotine levels from study samples.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Participants gave written informed consent, using a form approved by the Institutional Review Board at the VA Greater Los Angeles Healthcare System.
Funding
This work was supported by the National Institute on Drug Abuse (R01 DA20872), the Department of Veterans Affairs, Office of Research and Development (CSR&D Merit Review Award I01 CX000412), and the Tobacco-Related Disease Research Program (#23XT-0002) to ALB. The authors report no biomedical financial interests or potential conflicts of interest.
Rights and permissions
About this article
Cite this article
Brody, A.L., Hubert, R., Mamoun, M.S. et al. Nicotinic acetylcholine receptor availability in cigarette smokers: effect of heavy caffeine or marijuana use. Psychopharmacology 233, 3249–3257 (2016). https://doi.org/10.1007/s00213-016-4367-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4367-x