Abstract
Rationale
We previously reported that prenatal treatment with phencyclidine (PCP) induces glutamatergic dysfunction in the prefrontal cortex (PFC), leading to schizophrenia-like behavioral deficits in adult mice. However, little is known about the prenatal effect of PCP treatment on other types of neurons.
Objectives
We focused on γ-aminobutyric acid (GABA)-ergic interneurons and evaluated the effect of prenatal PCP exposure on the neurodevelopment of GABAergic interneurons in the PFC.
Methods
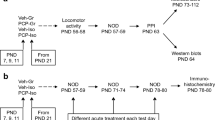
PCP was administered at the dose of 10 mg/kg/day to pregnant dams from embryonic day 6.5 to 18.5. After the pups were reared to adult, we analyzed their GABAergic system in the PFC using immunohistological, biochemical, and behavioral analyses in adulthood.
Results
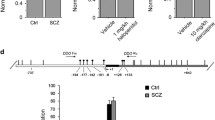
The prenatal PCP treatment decreased the density of parvalbumin-positive cells and reduced the expression level of glutamic acid decarboxylase 67 (GAD67) and GABA content of the PFC in adults. Additionally, prenatal PCP treatment induced behavioral deficits in adult mice, such as hypersensitivity to PCP and prepulse inhibition (PPI) deficits. These behavioral deficits were ameliorated by pretreatment with the GABAB receptor agonist baclofen. Furthermore, the density of c-Fos-positive cells was decreased after the PPI test in the PFC of mice treated with PCP prenatally, and this effect was ameliorated by pretreatment with baclofen.
Conclusions
These findings suggest that prenatal treatment with PCP induced GABAergic dysfunction in the PFC, which caused behavioral deficits.



Similar content being viewed by others
References
Abekawa T, Ito K, Nakagawa S, Koyama T (2007) Prenatal exposure to an NMDA receptor antagonist, MK-801 reduces density of parvalbumin-immunoreactive GABAergic neurons in the medial prefrontal cortex and enhances phencyclidine-induced hyperlocomotion but not behavioral sensitization to methamphetamine in postpubertal rats. Psychopharmacology 192:303–316. doi:10.1007/s00213-007-0729-8
Akbarian S, Kim JJ, Potkin SG et al (1995) Gene expression for glutamic acid decarboxylase is reduced without loss of neurons in prefrontal cortex of schizophrenics. Arch Gen Psychiatry 52:258–266. doi:10.1001/archpsyc.1995.03950160008002
Allison LA, Mayer GS, Shoup RE (1984) o-Phthalaldehyde derivatives of amines for high-speed liquid chromatography/electrochemistry. Anal Chem 56:1089–1096. doi:10.1021/ac00271a010
Andersen JD, Pouzet B (2004) Spatial memory deficits induced by perinatal treatment of rats with PCP and reversal effect of D-serine. Neuropsychopharmacology 29:1080–1090. doi:10.1038/sj.npp.1300394
Aoyama Y, Mouri A, Toriumi K et al (2014) Clozapine ameliorates epigenetic and behavioral abnormalities induced by phencyclidine through activation of dopamine D1 receptor. Int J Neuropsychopharmacol 17:723–737. doi:10.1017/S1461145713001466
Arai S, Takuma K, Mizoguchi H et al (2008) Involvement of pallidotegmental neurons in methamphetamine- and MK-801-induced impairment of prepulse inhibition of the acoustic startle reflex in mice: reversal by GABAB receptor agonist baclofen. Neuropsychopharmacology 33:3164–3175. doi:10.1038/npp.2008.41
Arai S, Takuma K, Mizoguchi H et al (2009) GABAB receptor agonist baclofen improves methamphetamine-induced cognitive deficit in mice. Eur J Pharmacol 602:101–104. doi:10.1016/j.ejphar.2008.10.065
Benes FM, Berretta S (2001) GABAergic interneurons: implications for understanding schizophrenia and bipolar disorder. Neuropsychopharmacology 25:1–27. doi:10.1016/S0893-133X(01)00225-1
Benes FM, Vincent SL, Marie A, Khan Y (1996) Up-regulation of GABAA receptor binding on neurons of the prefrontal cortex in schizophrenic subjects. Neuroscience 75:1021–1031. doi:10.1016/0306-4522(96)00328-4
Brown KN, Chen S, Han Z et al (2011) Clonal production and organization of inhibitory interneurons in the neocortex. Science 334:480–486. doi:10.1126/science.1208884
Carlson GC, Talbot K, Halene TB et al (2011) Dysbindin-1 mutant mice implicate reduced fast-phasic inhibition as a final common disease mechanism in schizophrenia. Proc Natl Acad Sci U S A 108:E962–E970. doi:10.1073/pnas.1109625108
Daskalakis ZJ, Christensen BK, Chen R et al (2002) Evidence for impaired cortical inhibition in schizophrenia using transcranial magnetic stimulation. Arch Gen Psychiatry 59:347–354. doi:10.1001/archpsyc.59.4.347
Fazzari P, Paternain AV, Valiente M et al (2010) Control of cortical GABA circuitry development by Nrg1 and ErbB4 signalling. Nature 464:1376–1380. doi:10.1038/nature08928
Fejgin K, Pålsson E, Wass C et al (2009) Prefrontal GABAB receptor activation attenuates phencyclidine-induced impairments of prepulse inhibition: involvement of nitric oxide. Neuropsychopharmacology 34:1673–1684. doi:10.1038/npp.2008.225
Ferrarelli F, Sarasso S, Guller Y et al (2012) Reduced natural oscillatory frequency of frontal thalamocortical circuits in schizophrenia. Arch Gen Psychiatry 69:766–774. doi:10.1001/archgenpsychiatry.2012.147
Fisahn A, Neddens J, Yan L, Buonanno A (2009) Neuregulin-1 modulates hippocampal gamma oscillations: implications for schizophrenia. Cereb Cortex 19:612–618. doi:10.1093/cercor/bhn107
Flames N, Long JE, Garratt AN et al (2004) Short- and long-range attraction of cortical GABAergic interneurons by neuregulin-1. Neuron 44:251–261. doi:10.1016/j.neuron.2004.09.028
Gandal MJ, Sisti J, Klook K et al (2012) GABAB-mediated rescue of altered excitatory–inhibitory balance, gamma synchrony and behavioral deficits following constitutive NMDAR-hypofunction. Transl Psychiatry 2, e142. doi:10.1038/tp.2012.69
Geyer MA, Swerdlow NR (2001) Measurement of startle response, prepulse inhibition, and habituation. Curr Protoc Neurosci Chapter 8:Unit 8.7. doi: 10.1002/0471142301.ns0807s03
Glickstein SB, Moore H, Slowinska B et al (2007) Selective cortical interneuron and GABA deficits in cyclin D2-null mice. Development 134:4083–4093. doi:10.1242/dev.008524
Harich S, Gross G, Bespalov A (2007) Stimulation of the metabotropic glutamate 2/3 receptor attenuates social novelty discrimination deficits induced by neonatal phencyclidine treatment. Psychopharmacology 192:511–519. doi:10.1007/s00213-007-0742-y
Hashimoto T, Volk DW, Eggan SM et al (2003) Gene expression deficits in a subclass of GABA neurons in the prefrontal cortex of subjects with schizophrenia. J Neurosci 23:6315–6326. doi:10.1016/S0920-9964(03)80597-2
Herdegen T, Leah JD (1998) Inducible and constitutive transcription factors in the mammalian nervous system: control of gene expression by Jun, Fos and Krox, and CREB/ATF proteins. Brain Res Brain Res Rev 28:370–490. doi:10.1016/S0165-0173(98)00018-6
Hikida T, Jaaro-Peled H, Seshadri S et al (2007) Dominant-negative DISC1 transgenic mice display schizophrenia-associated phenotypes detected by measures translatable to humans. Proc Natl Acad Sci U S A 104:14501–14506. doi:10.1073/pnas.0704774104
Ikonomidou C, Bosch F, Miksa M et al (1999) Blockade of NMDA receptors and apoptotic neurodegeneration in the developing brain. Science 283:70–74. doi:10.1126/science.283.5398.70
Ishikawa M, Mizukami K, Iwakiri M, Asada T (2005) Immunohistochemical and immunoblot analysis of gamma-aminobutyric acid B receptor in the prefrontal cortex of subjects with schizophrenia and bipolar disorder. Neurosci Lett 383:272–277. doi:10.1016/j.neulet.2005.04.025
Javitt DC, Zukin SR (1991) Recent advances in the phencyclidine model of schizophrenia. Am J Psychiatry 148:1301–1308. doi:10.1176/ajp.148.10.1301
Kimoto T, Tsurugizawa T, Ohta Y et al (2001) Neurosteroid synthesis by cytochrome p450-containing systems localized in the rat brain hippocampal neurons: N-methyl-D-aspartate and calcium-dependent synthesis. Endocrinology 142:3578–3589. doi:10.1210/endo.142.8.8327
Lewis DA, Hashimoto T, Volk DW (2005) Cortical inhibitory neurons and schizophrenia. Nat Rev Neurosci 6:312–324. doi:10.1038/nrn1648
Lu L, Mamiya T, Lu P et al (2010) Prenatal exposure to phencyclidine produces abnormal behaviour and NMDA receptor expression in postpubertal mice. Int J Neuropsychopharmacol 13:877–889. doi:10.1017/S1461145709990757
Lu L, Mamiya T, Lu P et al (2011) Prenatal exposure to PCP produces behavioral deficits accompanied by the overexpression of GLAST in the prefrontal cortex of postpubertal mice. Behav Brain Res 220:132–139. doi:10.1016/j.bbr.2011.01.035
Luk KC, Kennedy TE, Sadikot AF (2003) Glutamate promotes proliferation of striatal neuronal progenitors by an NMDA receptor-mediated mechanism. J Neurosci 23:2239–2250
Maddox VH, Godefroi EF, Parcell RF (1965) The synthesis of phencyclidine and other 1-arylcyclohexylamines. J Med Chem 8:230–235. doi:10.1021/jm00326a019
Mao Y, Ge X, Frank CL et al (2009) Disrupted in schizophrenia 1 regulates neuronal progenitor proliferation via modulation of GSK3beta/beta-catenin signaling. Cell 136:1017–1031. doi:10.1016/j.cell.2008.12.044
Marx CE, Bradford DW, Hamer RM et al (2011) Pregnenolone as a novel therapeutic candidate in schizophrenia: emerging preclinical and clinical evidence. Neuroscience 191:78–90. doi:10.1016/j.neuroscience.2011.06.076
Miwa H, Kakizaki T, Kaneko R et al (2015) Glutamate decarboxylase 67 deficiency in a subset of GABAergic neurons induces schizophrenia-related phenotypes. Neuropsychopharmacology 40:2475–2486. doi:10.1038/npp.2015.117
Mouri A, Noda Y, Enomoto T, Nabeshima T (2007a) Phencyclidine animal models of schizophrenia: approaches from abnormality of glutamatergic neurotransmission and neurodevelopment. Neurochem Int 51:173–184. doi:10.1016/j.neuint.2007.06.019
Mouri A, Noda Y, Noda A et al (2007b) Involvement of a dysfunctional dopamine-D1/N-methyl-d-aspartate-NR1 and Ca2+/calmodulin-dependent protein kinase II pathway in the impairment of latent learning in a model of schizophrenia induced by phencyclidine. Mol Pharmacol 71:1598–1609. doi:10.1124/mol.106.032961
Mouri A, Sasaki A, Watanabe K et al (2012) MAGE-D1 regulates expression of depression-like behavior through serotonin transporter ubiquitylation. J Neurosci 32:4562–4580. doi:10.1523/JNEUROSCI.6458-11.2012
National Research Council Committee for the Update of the Guide for the Care and Use of Laboratory Animals (2011) Guide for the Care and Use of Laboratory Animals, 8th edn. National Academies Press, Washington
Nakatani-Pawlak A, Yamaguchi K, Tatsumi Y et al (2009) Neonatal phencyclidine treatment in mice induces behavioral, histological and neurochemical abnormalities in adulthood. Biol Pharm Bull 32:1576–1583
Niwa M, Nitta A, Mizoguchi H et al (2007) A novel molecule “shati” is involved in methamphetamine-induced hyperlocomotion, sensitization, and conditioned place preference. J Neurosci 27:7604–7615. doi:10.1523/JNEUROSCI.1575-07.2007
Niwa M, Kamiya A, Murai R et al (2010) Knockdown of DISC1 by in utero gene transfer disturbs postnatal dopaminergic maturation in the frontal cortex and leads to adult behavioral deficits. Neuron 65:480–489. doi:10.1016/j.neuron.2010.01.019
Ritsner MS (2011) The clinical and therapeutic potentials of dehydroepiandrosterone and pregnenolone in schizophrenia. Neuroscience 191:91–100. doi:10.1016/j.neuroscience.2011.04.017
Sircar R (2003) Postnatal phencyclidine-induced deficit in adult water maze performance is associated with N-methyl-D-aspartate receptor upregulation. Int J Dev Neurosci 21:159–167. doi:10.1016/S0736-5748(03)00026-1
Takahashi M, Kakita A, Futamura T et al (2006) Sustained brain-derived neurotrophic factor up-regulation and sensorimotor gating abnormality induced by postnatal exposure to phencyclidine: comparison with adult treatment. J Neurochem 99:770–780. doi:10.1111/j.1471-4159.2006.04106.x
Ting AK, Chen Y, Wen L et al (2011) Neuregulin 1 promotes excitatory synapse development and function in GABAergic interneurons. J Neurosci 31:15–25. doi:10.1523/JNEUROSCI.2538-10.2011
Toriumi K, Mouri A, Narusawa S et al (2012) Prenatal NMDA receptor antagonism impaired proliferation of neuronal progenitor, leading to fewer glutamatergic neurons in the prefrontal cortex. Neuropsychopharmacology 37:1387–1396. doi:10.1038/npp.2011.324
Toriumi K, Kondo M, Nagai T et al (2014) Deletion of SHATI/NAT8L increases dopamine D1 receptor on the cell surface in the nucleus accumbens, accelerating methamphetamine dependence. Int J Neuropsychopharmacol 17:443–453. doi:10.1017/S1461145713001302
Wang C, McInnis J, Ross-Sanchez M et al (2001) Long-term behavioral and neurodegenerative effects of perinatal phencyclidine administration: implications for schizophrenia. Neuroscience 107:535–550. doi:10.1016/S0306-4522(01)00384-0
Wang C, McInnis J, West JB et al (2003) Blockade of phencyclidine-induced cortical apoptosis and deficits in prepulse inhibition by M40403, a superoxide dismutase mimetic. J Pharmacol Exp Ther 304:266–271. doi:10.1124/jpet.102.041798
Wang CZ, Yang SF, Xia Y, Johnson KM (2008) Postnatal phencyclidine administration selectively reduces adult cortical parvalbumin-containing interneurons. Neuropsychopharmacology 33:2442–2455. doi:10.1038/sj.npp.1301647
Weinberger DR, Berman KF, Zec RF (1986) Physiologic dysfunction of dorsolateral prefrontal cortex in schizophrenia. I. Regional cerebral blood flow evidence. Arch Gen Psychiatry 43:114–124. doi:10.1001/archpsyc.1986.01800020020004
Wiley JL, Bühler KG, Lavecchia KL, Johnson KM (2003) Pharmacological challenge reveals long-term effects of perinatal phencyclidine on delayed spatial alternation in rats. Prog Neuropsychopharmacol Biol Psychiatry 27:867–873. doi:10.1016/S0278-5846(03)00146-5
Williams SM, Goldman-Rakic PS, Leranth C (1992) The synaptology of parvalbumin-immunoreactive neurons in the primate prefrontal cortex. J Comp Neurol 320:353–369. doi:10.1002/cne.903200307
Wonders CP, Anderson SA (2006) The origin and specification of cortical interneurons. Nat Rev Neurosci 7:687–696. doi:10.1038/nrn1954
Acknowledgments
This study was supported by the following: Grants-in-Aid for Scientific Research (C) (2646024 and 15K08218); Grants-in-Aid for Young Scientists (B) (26870878); Joint Research Project under the Japan-Korea Basic Scientific Cooperation Program; Program for Advancing Strategic International Networks to Accelerate the Circulation of Talented Researchers (S2603) from the Japan Society for the Promotion of Science (JSPS); Research Grants from the Ministry of Health, Labor, and Welfare of Japan; and Research Grant from the Smoking Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experimental procedures were approved by the Animal Experiment Committee of Meijo University (YakuJitsu-Nos. 9, 12, and 25). Procedures involving animals and their care were conducted according to international guidelines (National Research Council Committee 2011).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Toriumi, K., Oki, M., Muto, E. et al. Prenatal phencyclidine treatment induces behavioral deficits through impairment of GABAergic interneurons in the prefrontal cortex. Psychopharmacology 233, 2373–2381 (2016). https://doi.org/10.1007/s00213-016-4288-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4288-8