Abstract
Rationale
Rats emit 50-kHz ultrasonic vocalizations (USVs) in response to pleasurable stimuli, and these USVs are considered a tool for investigating reward and motivation.
Objectives
This study aimed to clarify how activity of adenosine A2A receptors, which modulate reward and motivation, influences 50-kHz USV emission in rats.
Methods
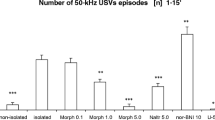
Rats received one of the following treatments in a test cage: (1) acute administration of the A2A receptor agonist CGS 21680 (0.05–0.2 mg/kg, i.p.) during social interactions; (2) long-term amphetamine (1 or 2 mg/kg, i.p.) or morphine (7.5 mg/kg, s.c.) administration on alternate days, alone or with CGS 21680, followed after 7 days of discontinuation by test cage re-exposure, to assess drug-conditioning effects, and thereafter drug challenge; (3) acute administration of the D1/D2 receptor agonist apomorphine (4 mg/kg, i.p.), alone or with CGS 21680; and (4) long-term administration of the non-selective A1/A2A receptor antagonist caffeine (15 mg/kg, i.p.), on alternate days. USVs and locomotor activity were evaluated throughout the treatments.
Results
CGS 21680 attenuated 50-kHz USV emission stimulated by social interactions, amphetamine, apomorphine, and morphine, and rats administered CGS 21680 with amphetamine or morphine emitted fewer conditioned 50-kHz USVs upon test cage re-exposure, compared with rats administered amphetamine or morphine alone. Moreover, CGS 21680 administration prevented long-term changes in locomotor activity in amphetamine- and morphine-treated rats. Finally, caffeine had no effect on 50-kHz USVs.
Conclusions
These results indicate that activation of A2A receptors attenuates 50-kHz USV emission in rats and further elucidate how these receptors modulate the motivational properties of natural and pharmacological stimuli.







Similar content being viewed by others
References
Ahrens AM, Ma ST, Maier EY, Duvauchelle CL, Schallert T (2009) Repeated intravenous amphetamine exposure: rapid and persistent sensitization of 50-kHz ultrasonic trill calls in rats. Behav Brain Res 197:205–209. doi:10.1016/j.bbr.2008.08.037
Bardo MT, Valone JM, Bevins RA (1999) Locomotion and conditioned place preference produced by acute intravenous amphetamine: role of dopamine receptors and individual differences in amphetamine self-administration. Psychopharmacology 143:39–46. doi:10.1007/s002130050917
Barker DJ, Bercovicz D, Servilio LC, Simmons SJ, Ma S, Root DH, Pawlak AP, West MO (2014) Rat ultrasonic vocalizations demonstrate that the motivation to contextually reinstate cocaine-seeking behavior does not necessarily involve a hedonic response. Addict Biol 19:781–790. doi:10.1111/adb.12044
Barker DJ, Simmons SJ, West MO (2015) Ultrasonic vocalizations as a measure of affect in preclinical models of drug abuse: a review of current findings. Curr Neuropharmacol 13:193–201. doi:10.2174/1570159X13999150318113642
Barraco RA, Helfman CC, Anderson GF (1996) Augmented release of serotonin by adenosine A2a receptor activation and desensitization by CGS 21680 in the rat nucleus tractus solitarius. Brain Res 16:155–161. doi:10.1016/0006-8993(96)00279-X
Belardinelli L, Shryock JC, Snowdy S, Zhang Y, Monopoli A, Lozza G, Ongini E, Olsson RA, Dennis DM (1998) The A2A adenosine receptor mediates coronary vasodilation. J Pharmacol Exp Ther 284:1066–1073
Boison D (2012) Adenosine dysfunction in epilepsy. Glia 60:1234–1243. doi:10.1002/glia.22285
Bonci A, Bernardi G, Grillner P, Mercuri NB (2003) The dopamine-containing neuron: maestro or simple musician in the orchestra of addiction? Trends Pharmacol Sci 24:172–177. doi:10.1016/S0165-6147(03)00068-3
Brudzynski SM (2013) Ethotransmission: communication of emotional states through ultrasonic vocalization in rats. Curr Opin Neurobiol 23:310–317. doi:10.1016/j.conb.2013.01.014
Brudzynski SM (2015) Pharmacology of ultrasonic vocalizations in adult rats: significance, call classification and neural substrate. Curr Neuropharmacol 13:180–192. doi:10.2174/1570159X13999150210141444
Buck CL, Malavar JC, George O, Koob GF, Vendruscolo LF (2014) Anticipatory 50 kHz ultrasonic vocalizations are associated with escalated alcohol intake in dependent rats. Behav Brain Res 271:171–176. doi:10.1016/j.bbr.2014.06.003
Burgdorf J, Knutson B, Panksepp J, Ikemoto S (2001) Nucleus accumbens amphetamine microinjections unconditionally elicit 50-kHz ultrasonic vocalizations in rats. Behav Neurosci 115:940–944. doi:10.1037/0735-7044.115.4.940
Burgdorf J, Kroes RA, Moskal JR, Pfaus JG, Brudzynski SM, Panksepp J (2008) Ultrasonic vocalizations of rats (Rattus norvegicus) during mating, play, and aggression: Behavioral concomitants, relationship to reward, and self-administration of playback. J Comp Psychol 122:357–367. doi:10.1037/a0012889
Burgdorf J, Panksepp J, Brudzynski SM, Beinfeld MC, Cromwell HC, Kroes RA, Moskal JR (2009) The effects of selective breeding for differential rates of 50-kHz ultrasonic vocalizations on emotional behavior in rats. Dev Psychobiol 51:34–46. doi:10.1002/dev.20343
Burgdorf J, Panksepp J, Moskal JR (2011) Frequency-modulated 50 kHz ultrasonic vocalizations: a tool for uncovering the molecular substrates of positive affect. Neurosci Biobehav Rev 35:1831–1836. doi:10.1016/j.neubiorev.2010.11.011
Castañé A, Wells L, Soria G, Hourani S, Ledent C, Kitchen I, Opacka-Juffry J, Maldonado R, Valverde O (2008) Behavioural and biochemical responses to morphine associated with its motivational properties are altered in adenosine A2A receptor knockout mice. Br J Pharmacol 155:757–766. doi:10.1038/bjp.2008.299
Corsi C, Melani A, Bianchi L, Pepeu G, Pedata F (1999) Striatal A2A adenosine receptors differentially regulate spontaneous and K+-evoked glutamate release in vivo in young and aged rats. Neuroreport 10:687–691
Costa G, Morelli M, Simola N (2015) Involvement of glutamate NMDA receptors in the acute, long-term, and conditioned effects of amphetamine on rat 50 kHz ultrasonic vocalizations. Int J Neuropsychopharmacol 18(11):1–12. doi: 10.1093/ijnp/pyv057
Cristalli G, Müller CE, Volpini R (2009) Recent developments in adenosine A2A receptor ligands. In: Wilson CN, Mustafa SJ (eds) Handbook of experimental pharmacology. Springer, Berlin, pp 59–98. doi:10.1007/978-3-540-89615-9_3
Di Chiara G, Bassareo V, Fenu S, De Luca MA, Spina L, Cadoni C, Acquas E, Carboni E, Valentini V, Lecca D (2004) Dopamine and drug addiction: the nucleus accumbens shell connection. Neuropharmacology 47:227–241. doi:10.1016/j.neuropharm.2004.06.032
Doyle SE, Breslin FJ, Rieger JM, Beauglehole A, Lynch WJ (2012) Time and sex-dependent effects of an adenosine A2A/A1 receptor antagonist on motivation to self-administer cocaine in rats. Pharmacol Biochem Behav 102:257–263. doi:10.1016/j.pbb.2012.05.001
Farrar AM, Pereira M, Velasco F, Hockemeyer J, Müller CE, Salamone JD (2007) Adenosine A2A receptor antagonism reverses the effects of dopamine receptor antagonism on instrumental output and effort-related choice in the rat: implications for studies of psychomotor slowing. Psychopharmacology 191:579–586. doi:10.1007/s00213-006-0554-5
Fenu S, Spina L, Rivas E, Longoni R, Di Chiara G (2006) Morphine-conditioned single-trial place preference: role of nucleus accumbens shell dopamine receptors in acquisition, but not expression. Psychopharmacology 187:143–153. doi:10.1007/s00213-006-0415-2
Ferré S, Fredholm BB, Morelli M, Popoli P, Fuxe K (1997) Adenosine-dopamine receptor-receptor interactions as an integrative mechanism in the basal ganglia. Trends Neurosci 20:482–487. doi:10.1016/S0166-2236(97)01096-5
Font L, Mingote S, Farrar AM, Pereira M, Worden L, Stopper C, Port RG, Salamone JD (2008) Intra-accumbens injections of the adenosine A2A agonist CGS 21680 affect effort-related choice behavior in rats. Psychopharmacology 199:515–526. doi:10.1007/s00213-008-1174-z
Gołembiowska K, Zylewska A (1997) Adenosine receptors—the role in modulation of dopamine and glutamate release in the rat striatum. Pol J Pharmacol 49:317–322
Gomes CV, Kaster MP, Tomé AR, Agostinho PM, Cunha RA (2011) Adenosine receptors and brain diseases: neuroprotection and neurodegeneration. Biochim Biophys Acta 1808:1380–1399. doi:10.1016/j.bbamem.2010.12.001
Hamed A, Taracha E, Szyndler J, Krząścik P, Lehner M, Maciejak P, Skórzewska A, Płaźnik A (2012) The effects of morphine and morphine conditioned context on 50 kHz ultrasonic vocalisation in rats. Behav Brain Res 229:447–450. doi:10.1016/j.bbr.2012.01.053
Harper LK, Beckett SR, Marsden CA, McCreary AC, Alexander SP (2006) Effects of the A2A adenosine receptor antagonist KW6002 in the nucleus accumbens in vitro and in vivo. Pharmacol Biochem Behav 83:114–121. doi:10.1016/j.pbb.2005.12.014
Houchi H, Persyn W, Legastelois R, Naassila M (2013) The adenosine A2A receptor agonist CGS 21680 decreases ethanol self-administration in both non-dependent and dependent animals. Addict Biol 18:812–825. doi:10.1111/adb.12032
Huang ZL, Urade Y, Hayaishi O (2011) The role of adenosine in the regulation of sleep. Curr Top Med Chem 11:1047–1057. doi:10.2174/156802611795347654
Hutchison AJ, Webb RL, Oei HH, Ghai GR, Zimmerman MB, Williams M (1989) CGS 21680C, an A2 selective adenosine receptor agonist with preferential hypotensive activity. J Pharmacol Exp Ther 251:47–55
Khroyan TV, Fuchs RA, Beck AM, Groff RS, Neisewander JL (1999) Behavioral interactions produced by co-administration of 7-OH-DPAT with cocaine or apomorphine in the rat. Psychopharmacology 142:383–392. doi:10.1007/s002130050903
Kirkup AJ, Eastwood C, Grundy D, Chessell IP, Humphrey PP (1998) Characterization of adenosine receptors evoking excitation of mesenteric afferents in the rat. Br J Pharmacol 125:1352–1360. doi:10.1038/sj.bjp.0702202
Klotz KN, Hessling J, Hegler J, Owman C, Kull B, Fredholm BB, Lohse MJ (1998) Comparative pharmacology of human adenosine receptor subtypes—characterization of stably transfected receptors in CHO cells. Naunyn Schmiedebergs Arch Pharmacol 357:1–9. doi:10.1007/PL00005131
Knutson B, Burgdorf J, Panksepp J (1999) High-frequency ultrasonic vocalizations index conditioned pharmacological reward in rats. Physiol Behav 66:639–643. doi:10.1016/S0031-9384(98)00337-0
Listos J, Talarek S, Poleszak E, Wróbel A, Fidecka S (2011) Attenuating effect of adenosine receptor agonists on the development of behavioral sensitization induced by sporadic treatment with morphine. Pharmacol Biochem Behav 98:356–361. doi:10.1016/j.pbb.2011.01.019
Maier EY, Abdalla M, Ahrens AM, Schallert T, Duvauchelle CL (2012) The missing variable: ultrasonic vocalizations reveal hidden sensitization and tolerance-like effects during long-term cocaine administration. Psychopharmacology 219:1141–1152. doi:10.1007/s00213-011-2445-7
Mingote S, Pereira M, Farrar AM, McLaughlin PJ, Salamone JD (2008) Systemic administration of the adenosine A2A agonist CGS 21680 induces sedation at doses that suppress lever pressing and food intake. Pharmacol Biochem Behav 89:345–351. doi:10.1016/j.pbb.2008.01.006
Morelli M, Simola N (2011) Methylxanthines and drug dependence: a focus on interactions with substances of abuse. In: Fredholm BB (ed) Handbook of Experimental Pharmacology. Springer, Berlin, pp 483–507. doi:10.1007/978-3-642-13443-2_20
Mott AM, Nunes EJ, Collins LE, Port RG, Sink KS, Hockemeyer J, Müller CE, Salamone JD (2009) The adenosine A2A antagonist MSX-3 reverses the effects of the dopamine antagonist haloperidol on effort-related decision making in a T-maze cost/benefit procedure. Psychopharmacology 204:103–112. doi:10.1007/s00213-008-1441-z
Mu P, Fuchs T, Saal DB, Sorg BA, Dong Y, Panksepp J (2009) Repeated cocaine exposure induces sensitization of ultrasonic vocalization in rats. Neurosci Lett 453:31–35. doi:10.1016/j.neulet.2009.02.007
Nunes EJ, Randall PA, Podurgiel S, Correa M, Salamone JD (2013) Nucleus accumbens neurotransmission and effort-related choice behavior in food motivation: effects of drugs acting on dopamine, adenosine, and muscarinic acetylcholine receptors. Neurosci Biobehav Rev 37:2015–2025. doi:10.1016/j.neubiorev.2013.04.002
Okada M, Kawata Y, Murakami T, Wada K, Mizuno K, Kondo T, Kaneko S (1999) Differential effects of adenosine receptor subtypes on release and reuptake of hippocampal serotonin. Eur J Neurosci 11:1–9. doi:10.1046/j.1460-9568.1999.00415.x
Panksepp J (2005) Beyond a joke: from animal laughter to human joy? Science 308:62–63. doi:10.1126/science.1112066
Pinna A, Morelli M (2014) A critical evaluation of behavioral rodent models of motor impairment used for screening of antiparkinsonian activity: the case of adenosine A2A receptor antagonists. Neurotox Res 25:392–401. doi:10.1007/s12640-013-9446-8
Poleszak E, Malec D (2003) Effects of adenosine receptor agonists and antagonists in amphetamine-induced conditioned place preference test in rats. Pol J Pharmacol 55:319–326
Rimondini R, Ferré S, Ogren SO, Fuxe K (1997) Adenosine A2A agonists: a potential new type of atypical antipsychotic. Neuropsychopharmacology 17:82–91. doi:10.1016/S0893-133X(97)00033-X
Rippberger H, van Gaalen MM, Schwarting RKW, Wöhr M (2015) Environmental and pharmacological modulation of amphetamine-induced 50-kHz ultrasonic vocalizations in rats. Curr Neuropharmacol 13:220–232. doi:10.2174/1570159X1302150525124408
Robinson TE, Berridge KC (2001) Incentive-sensitization and addiction. Addiction 96:103–114. doi:10.1046/j.1360-0443.2001.9611038.x
Ruiz-Medina J, Ledent C, Carretón O, Valverde O (2011) The A2a adenosine receptor modulates the reinforcement efficacy and neurotoxicity of MDMA. J Psychopharmacol 25:550–564. doi:10.1177/0269881110389210
Rump LC, Jabbari-T J, von Kügelgen I, Oberhauser V (1999) Adenosine mediates nitric-oxide-independent renal vasodilation by activation of A2A receptors. J Hypertens 17:1987–1993
Sahraei H, Motamedi F, Khoshbaten A, Zarrindast MR (1999) Adenosine A2 receptors inhibit morphine self-administration in rats. Eur J Pharmacol 383:107–113. doi:10.1016/S0014-2999(99)00613-5
Sahraei H, Barzegari AA, Shams J, Zarrindast MR, Haeri-Rohani A, Ghoshooni H, Sepehri H, Salimi SH (2006) Theophylline inhibits tolerance and sensitization induced by morphine: a conditioned place preference paradigm study in female mice. Behav Pharmacol 17:621–628. doi:10.1097/01.fbp.0000236274.18042.54
Sato A, Terata K, Miura H, Toyama K, Loberiza FR Jr, Hatoum OA, Saito T, Sakuma I, Gutterman DD (2005) Mechanism of vasodilation to adenosine in coronary arterioles from patients with heart disease. Am J Physiol Heart Circ Physiol 288:H1633–H1640
Schiffmann SN, Fisone G, Moresco R, Cunha RA, Ferré S (2007) Adenosine A2A receptors and basal ganglia physiology. Prog Neurobiol 83:277–292. doi:10.1016/j.pneurobio.2007.05.001
Schindler CW, Karcz-Kubicha M, Thorndike EB, Müller CE, Tella SR, Ferré S, Goldberg SR (2005) Role of central and peripheral adenosine receptors in the cardiovascular responses to intraperitoneal injections of adenosine A1 and A2A subtype receptor agonists. Br J Pharmacol 144:642–650. doi:10.1038/sj.bjp.0706043
Schwarting RK, Jegan N, Wöhr M (2007) Situational factors, conditions and individual variables which can determine ultrasonic vocalizations in male adult Wistar rats. Behav Brain Res 182:208–222. doi:10.1016/j.bbr.2007.01.029
Simola N (2015) Rat ultrasonic vocalizations and behavioral neuropharmacology: from the screening of drugs to the study of disease. Curr Neuropharmacol 13:164–179. doi:10.2174/1570159X13999150318113800
Simola N, Morelli M (2015) Repeated amphetamine administration and long-term effects on 50-kHz ultrasonic vocalizations: possible relevance to the motivational and dopamine-stimulating properties of the drug. Eur Neuropsychopharmacol 25:343–355. doi:10.1016/j.euroneuro.2015.01.010
Simola N, Cauli O, Morelli M (2006) Sensitization to caffeine and cross-sensitization to amphetamine: influence of individual response to caffeine. Behav Brain Res 172:72–79. doi:10.1016/j.bbr.2006.04.019
Simola N, Morelli M, Seeman P (2008) Increase of dopamine D2High receptors in the striatum of rats sensitized to caffeine motor effects. Synapse 62:394–397. doi:10.1002/syn.20507
Simola N, Ma ST, Schallert T (2010) Influence of acute caffeine on 50-kHz ultrasonic vocalizations in male adult rats and relevance to caffeine-mediated psychopharmacological effects. Int J Neuropsychopharmacol 13:123–132. doi:10.1017/S1461145709990113
Simola N, Fenu S, Costa G, Pinna A, Plumitallo A, Morelli M (2012) Pharmacological characterization of 50-kHz ultrasonic vocalizations in rats: comparison of the effects of different psychoactive drugs and relevance in drug-induced reward. Neuropharmacology 63:224–234. doi:10.1016/j.neuropharm.2012.03.013
Simola N, Frau L, Plumitallo A, Morelli M (2014) Direct and long-lasting effects elicited by repeated drug administration on 50-kHz ultrasonic vocalizations are regulated differently: implications for the study of the affective properties of drugs of abuse. Int J Neuropsychopharmacol 17:429–441. doi:10.1017/S1461145713001235
Taracha E, Kaniuga E, Charpusta SJ, Maciejak P, Sliwa L, Hamed A, Krząścik P (2014) Diverging frequency-modulated 50-kHz vocalization, locomotor activity and conditioned place preference effects in rats given repeated amphetamine treatment. Neuropharmacology 83:128–136. doi:10.1016/j.neuropharm.2014.04.008
Thompson B, Leonard KC, Brudzynski SM (2006) Amphetamine-induced 50 kHz calls from rat nucleus accumbens: a quantitative mapping study and acoustic analysis. Behav Brain Res 168:64–73. doi:10.1016/j.bbr.2005.10.012
Tuazon DB, Suzuki T, Misawa M, Watanabe S (1992) Methylxanthines (caffeine and theophylline) blocked methamphetamine-induced conditioned place preference in mice but enhanced that induced by cocaine. Ann N Y Acad Sci 654:531–533. doi:10.1111/j.1749-6632.1992.tb26022.x
Willuhn I, Tose A, Wanat MJ, Hart AS, Hollon NG, Phillips PE, Schwarting RK, Wöhr M (2014) Phasic dopamine release in the nucleus accumbens in response to pro-social 50 kHz ultrasonic vocalizations in rats. J Neurosci 34:10616–10623. doi:10.1523/JNEUROSCI.1060-14.2014
Wintink AJ, Brudzynski SM (2001) The related roles of dopamine and glutamate in the initiation of 50-kHz ultrasonic calls in adult rats. Pharmacol Biochem Behav 70:317–323. doi:10.1016/S0091-3057(01)00615-3
Wöhr M, Rippberger H, Schwarting RK, van Gaalen MM (2015) Critical involvement of 5-HT2C receptor function in amphetamine-induced 50-kHz ultrasonic vocalizations in rats. Psychopharmacology 232:1817–1829. doi:10.1007/s00213-014-3814-9
Wright JM, Deng L, Clarke PB (2012) Failure of rewarding and locomotor stimulant doses of morphine to promote adult rat 50-kHz ultrasonic vocalizations. Psychopharmacology 224:477–487. doi:10.1007/s00213-012-2776-z
Wydra K, Gołembiowska K, Suder A, Kamińska K, Fuxe K, Filip M (2015) On the role of adenosine (A)2A receptors in cocaine-induced reward: a pharmacological and neurochemical analysis in rats. Psychopharmacology 232:421–435. doi:10.1007/s00213-014-3675-2
Acknowledgments
The authors are grateful to Ms. Flora Corrias and Mr. Riccardo Orrù for their help with the recordings of vocalizations and locomotor activity.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experiments were conducted in accordance with the guidelines for animal experimentation of the EU directives (2010/63/EU; L.276; 22 September 2010) and with the guidelines approved by the Ethical Committee of the University of Cagliari.
Funding
This work was supported by funds from Fondazione Banco di Sardegna (project number 2014.0395) and by funds from the Regione Autonoma della Sardegna (Legge Regionale 7 Agosto 2007, N.7, annualità 2010). Dr. Nicola Simola gratefully acknowledges the Sardinian Regional Government for financial support (P.O.R. Sardegna F.S.E. Operational Programme of the Autonomous Region of Sardinia, European Social Fund 2007–2013—Axis IV Human Resources, Objective l.3, Line of Activity l.3.1 “Avviso di chiamata per il finanziamento di Assegni di Ricerca”).
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
Summary of the results of ANOVA followed by Tukey’s post hoc test obtained in day 1 of repeated treatment from rats administered amphetamine (1 or 2 mg/kg, i.p.) or morphine (7.5 mg/kg, s.c.). Each drug was administered alone or with the A2A receptor agonist CGS 21680 (0.05–0.2 mg/kg, i.p., 15 min before). Differences between the experimental groups are presented by comparing the group in each line with the group in each corresponding column. A = amphetamine, M = morphine, C = CGS 21680. ↑ = significantly higher compared with the group in the corresponding column; ↓ = significantly lower compared with the group in the corresponding column; n.s. = no significant difference compared with the group in the corresponding column; barred cells indicate that no statistical comparison between the two groups was performed. (DOC 89 kb)
Supplementary Table 2
Summary of the results of ANOVA followed by Tukey’s post hoc test obtained in day 5 of repeated treatment from rats administered amphetamine (1 or 2 mg/kg, i.p.) or morphine (7.5 mg/kg, s.c.). Each drug was administered alone or with the A2A receptor agonist CGS 21680 (0.05–0.2 mg/kg, i.p., 15 min before). Differences between the experimental groups are presented by comparing the group in each line with the group in each corresponding column. A = amphetamine, M = morphine, C = CGS 21680. ↑ = significantly higher compared with the group in the corresponding column; ↓ = significantly lower compared with the group in the corresponding column; n.s. = no significant difference compared with the group in the corresponding column; barred cells indicate that no statistical comparison between the two groups was performed. (DOC 91 kb)
Supplementary Table 3
Summary of the results of ANOVA followed by Tukey’s post hoc test obtained in day 9 of repeated treatment from rats administered amphetamine (1 or 2 mg/kg, i.p.) or morphine (7.5 mg/kg, s.c.). Each drug was administered alone or with the A2A receptor agonist CGS 21680 (0.05–0.2 mg/kg, i.p.). Differences between the experimental groups are presented by comparing the group in each line with the group in each corresponding column. A = amphetamine, M = morphine, C = CGS 21680. ↑ = significantly higher compared with the group in the corresponding column; ↓ = significantly lower compared with the group in the corresponding column; n.s. = no significant difference compared with the group in the corresponding column; barred cells indicate that no statistical comparison between the two groups was performed. (DOC 91 kb)
Supplementary Table 4
Summary of the results of ANOVA followed by Tukey’s post hoc test obtained after drug challenge (day 16) with amphetamine (1 or 2 mg/kg, i.p.) or morphine (7.5 mg/kg, s.c.) in rats repeatedly treated with either drug. Each drug was administered alone or with the A2A receptor agonist CGS 21680 (0.05–0.2 mg/kg, i.p.) during repeated treatment (days 1–9), whereas no CGS 21680 was administered in day 16. Differences between the experimental groups are presented by comparing the group in each line with the group in each corresponding column. A = amphetamine, M = morphine, C = CGS 21680. ↑ = significantly higher compared with the group in the corresponding column; ↓ = significantly lower compared with the group in the corresponding column; n.s. = no significant difference compared with the group in the corresponding column; barred cells indicate that no statistical comparison between the two groups was performed. (DOC 257 kb)
Rights and permissions
About this article
Cite this article
Simola, N., Costa, G. & Morelli, M. Activation of adenosine A2A receptors suppresses the emission of pro-social and drug-stimulated 50-kHz ultrasonic vocalizations in rats: possible relevance to reward and motivation. Psychopharmacology 233, 507–519 (2016). https://doi.org/10.1007/s00213-015-4130-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-015-4130-8