Abstract
Rationale
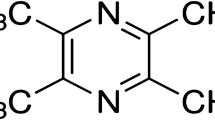
Evidences indicate that methylglyoxal, a highly reactive metabolite of hyperglycemia, can enhance protein glycation, oxidative stress, or inflammation. Mangiferin, a polyphenol compound of C-glucoside, has many beneficial biological activities, including anti-inflammation, anti-oxidation, neuroprotection, cognitive enhancement, etc. Whether mangiferin alleviates diabetes-associated cognitive impairment is still unclear.
Objectives
The present study was designed to investigate the effects of mangiferin on the behavioral deficits of diabetic rats induced by streptozotocin; the mechanisms associated with methylglyoxal toxicity are especially investigated.
Methods
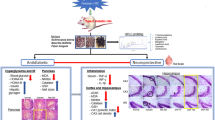
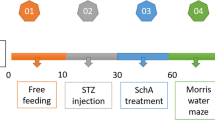
Diabetic rats were treated with mangiferin (15, 30, and 60 mg/kg, p.o.) for 9 weeks. Cognitive performances were evaluated with the Morris water maze. Hippocampus and blood were obtained for evaluation of the effects of mangiferin on protein glycation, oxidative stress, and inflammation in diabetic state.
Results
Mangiferin significantly improved the behavioral performances of diabetic rats, evidenced by a decrease in escape latency as well as increases in numbers of crossing the platform and percentage of time spent in the target quadrant, which were accompanied by decreases in the levels of advanced glycation end-products and their receptor (RAGE), interleukin-1β, TNF-α, and malondialdehyde and increases in the activity and expression of glyoxalase 1 as well as glutathione level in the hippocampus of diabetic rats. Furthermore, mangiferin produced a significant decrease in malondialdehyde level and increased glutathione level and superoxide dismutase activity in the serum of diabetic rats.
Conclusions
This study demonstrates that mangiferin can markedly ameliorate diabetes-associated cognitive decline in rats, which is done likely through suppressing methylglyoxal hyperactivity (promoting protein glycation, oxidative stress, and inflammation) mediated noxious effects.






Similar content being viewed by others
References
Beeri MS, Moshier E, Schmeidler J et al (2011) Serum concentration of an inflammatory glycotoxin, methylglyoxal, is associated with increased cognitive decline in elderly individuals. Mech Ageing Dev 132:583–587
Biessels GJ, Deary IJ, Ryan CM (2008) Cognition and diabetes: a lifespan perspective. Lancet Neurol 7:184–190
Brouwers O, Niessen PM, Ferreira I et al (2011) Overexpression of glyoxalase-I reduces hyperglycemia-induced levels of advanced glycation end products and oxidative stress in diabetic rats. J Biol Chem 286:1374–1380
Brownlee M (2005) The pathobiology of diabetic complications: a unifying mechanism. Diabetes 54:1615–1625
Calcutt NA, Cooper ME, Kern TS, Schmidt AM (2009) Therapies for hyperglycaemia-induced diabetic complications: from animal models to clinical trials. Nat Rev Drug Discov 8:417–429
Ceriello A (1999) Hyperglycaemia: the bridge between non-enzymatic glycation and oxidative stress in the pathogenesis of diabetic complications. Diabetes Nutr Metab 12:42–46
Das S, Nageshwar Rao B, Satish Rao BS (2011) Mangiferin attenuates methylmercury induced cytotoxicity against IMR-32, human neuroblastoma cells by the inhibition of oxidative stress and free radical scavenging potential. Chem Biol Interact 93:129–140
Di Loreto S, Caracciolo V, Colafarina S et al (2004) Methylglyoxal induces oxidative stress-dependent cell injury and up-regulation of interleukin-1beta and nerve growth factor in cultured hippocampal neuronal cells. Brain Res 1006:157–167
Di Loreto S, Zimmitti V, Sebastiani P et al (2008) Methylglyoxal causes strong weakening of detoxifying capacity and apoptotic cell death in rat hippocampal neurons. Int J Biochem Cell Biol 40:245–257
Fosmark DS, Berg JP, Jensen AB et al (2009) Increased retinopathy occurrence in type 1 diabetes patients with increased serum levels of the advanced glycation endproduct hydroimidazolone. Acta Ophthalmol 87:498–500
Garrido G, Delgado R, Lemus Y et al (2004) Protection against septic shock and suppression of tumor necrosis factor alpha and nitric oxide production on macrophages and microglia by a standard aqueous extract of Mangifera indica L. (VIMANG). Role of mangiferin isolated from the extract. Pharmacol Res 50:165–172
Gottlieb M, Leal-Campanario R, Campos-Esparza MR et al (2006) Neuroprotection by two polyphenols following excitotoxicity and experimental ischemia. Neurobiol Dis 23:374–386
Guo ZH, Xi RG, Wang XB, Wu LJ, Gao HY (2009) A new trincallane derivative from Salacia hainanensis Chun et How. Yao Xue Xue Bao 44:1123–1126 (in Chinese)
Hou Y, Fan S, Zhang H et al (2010) Pharmacokinetic study of mangiferin in rat plasma and retina using high-performance liquid chromatography. Mol Vis 16:1659–1668
Jung K, Lee B, Han SJ, Ryu JH, Kim DH (2009) Mangiferin ameliorates scopolamine-induced learning deficits in mice. Biol Pharm Bull 32:242–246
Kuhad A, Chopra K (2007) Curcumin attenuates diabetic encephalopathy in rats: behavioral and biochemical evidences. Eur J Pharmacol 576:34–42
Kuhad A, Bishnoi M, Tiwari V, Chopra K (2009) Suppression of NF-kappabeta signaling pathway by tocotrienol can prevent diabetes associated cognitive deficits. Pharmacol Biochem Behav 92:251–259
Leiro J, Arranz JA, Yanez M et al (2004) Expression profiles of genes involved in the mouse nuclear factor-kappa B signal transduction pathway are modulated by mangiferin. Int Immunopharmacol 4:763–778
Li X, Cui X, Sun X et al (2010) Mangiferin prevents diabetic nephropathy progression in streptozotocin-induced diabetic rats. Phytother Res 24:893–899
Liu YC, Liu HY, Yang HW et al (2007) Impaired expression and function of breast cancer resistance protein (Bcrp) in brain cortex of streptozocin-induced diabetic rats. Biochem Pharmacol 74:1766–1772
Liu YW, Zhu X, Lu Q et al (2012a) Total saponins from Rhizoma Anemarrhenae ameliorate diabetes-associated cognitive decline in rats: involvement of amyloid-beta decrease in brain. J Ethnopharmacol 139:194–200
Liu YW, Zhu X, Li W et al (2012b) Ginsenoside Re attenuates diabetes-associated cognitive deficits in rats. Pharmacol Biochem Behav 101:93–98
Lu J, Randell E, Han Y et al (2011) Increased plasma methylglyoxal level, inflammation, and vascular endothelial dysfunction in diabetic nephropathy. Clin Biochem 44:307–311
Maher P, Dargusch R, Ehren JL et al (2011) Fisetin lowers methylglyoxal dependent protein glycation and limits the complications of diabetes. PLoS One 6:e21226
Marquez L, Garcia-Bueno B, Madrigal JL, Leza JC (2012) Mangiferin decreases inflammation and oxidative damage in rat brain after stress. Eur J Nutr 51:729–739
Martinez Sanchez G, Candelario-Jalil E, Giuliani A et al (2001) Mangifera indica L. extract (QF808) reduces ischaemia-induced neuronal loss and oxidative damage in the gerbil brain. Free Radic Res 35:465–473
Miura T, Ichiki H, Iwamoto N et al (2001) Antidiabetic activity of the rhizoma of Anemarrhena asphodeloides and active components, mangiferin and its glucoside. Biol Pharm Bull 24:1009–1011
Muruganandan S, Gupta S, Kataria M, Lal J, Gupta PK (2002) Mangiferin protects the streptozotocin-induced oxidative damage to cardiac and renal tissues in rats. Toxicology 176:165–173
Muruganandan S, Srinivasan K, Gupta S, Gupta PK, Lal J (2005) Effect of mangiferin on hyperglycemia and atherogenicity in streptozotocin diabetic rats. J Ethnopharmacol 97:497–501
Pardo Andreu GL, Maurmann N, Reolon GK et al (2010) Mangiferin, a naturally occurring glucoxilxanthone improves long-term object recognition memory in rats. Eur J Pharmacol 635:124–128
Prabhu S, Jainu M, Sabitha KE, Devi CS (2006) Role of mangiferin on biochemical alterations and antioxidant status in isoproterenol-induced myocardial infarction in rats. J Ethnopharmacol 107:126–133
Rabbani N, Thornalley PJ (2008) Dicarbonyls linked to damage in the powerhouse: glycation of mitochondrial proteins and oxidative stress. Biochem Soc Trans 36:1045–1050
Ryan CM, Geckle MO, Orchard TJ (2003) Cognitive efficiency declines over time in adults with type 1 diabetes: effects of micro- and macrovascular complications. Diabetologia 46:940–948
Saxena G, Singh SP, Pal R, Singh S, Pratap R, Nath C (2007) Gugulipid, an extract of Commiphora whighitii with lipid-lowering properties, has protective effects against streptozotocin-induced memory deficits in mice. Pharmacol Biochem Behav 86:797–805
Sena CM, Matafome P, Crisostomo J et al (2012) Methylglyoxal promotes oxidative stress and endothelial dysfunction. Pharmacol Res 65:497–506
Shinohara M, Thornalley PJ, Giardino I et al (1998) Overexpression of glyoxalase-I in bovine endothelial cells inhibits intracellular advanced glycation endproduct formation and prevents hyperglycemia-induced increases in macromolecular endocytosis. J Clin Invest 101:1142–1147
Sima AA (2010) Encephalopathies: the emerging diabetic complications. Acta Diabetol 47:279–293
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34:497–500
Thornalley PJ (2005) Dicarbonyl intermediates in the Maillard reaction. Ann N Y Acad Sci 1043:111–117
Thornalley PJ (2007) Endogenous alpha-oxoaldehydes and formation of protein and nucleotide advanced glycation endproducts in tissue damage. Novartis Found Symp 285:229–243, discussion 243–226
van Deutekom AW, Niessen HW, Schalkwijk CG, Heine RJ, Simsek S (2008) Increased Nepsilon-(carboxymethyl)-lysine levels in cerebral blood vessels of diabetic patients and in a (streptozotocin-treated) rat model of diabetes mellitus. Eur J Endocrinol 158:655–660
Wang SH, Sun ZL, Guo YJ, Yuan Y, Yang BQ (2009) Diabetes impairs hippocampal function via advanced glycation end product mediated new neuron generation in animals with diabetes-related depression. Toxicol Sci 111:72–79
Wrighten SA, Piroli GG, Grillo CA, Reagan LP (2009) A look inside the diabetic brain: contributors to diabetes-induced brain aging. Biochim Biophys Acta 1792:444–453
Yamawaki H, Saito K, Okada M, Hara Y (2008) Methylglyoxal mediates vascular inflammation via JNK and p38 in human endothelial cells. Am J Physiol Cell Physiol 295:C1510–C1517
Yao D, Brownlee M (2010) Hyperglycemia-induced reactive oxygen species increase expression of the receptor for advanced glycation end products (RAGE) and RAGE ligands. Diabetes 59:249–255
Acknowledgment
The work was supported by the Natural Science Foundation of Jiangsu Province (BK2011208), China, China Postdoctoral Science Foundation (201150M1576), and a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), China. The experiments comply with the current laws of the People’s Republic of China.
Conflict of interest
The authors have declared that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yao-Wu Liu and Xia Zhu contributed equally to this project.
Rights and permissions
About this article
Cite this article
Liu, YW., Zhu, X., Yang, QQ. et al. Suppression of methylglyoxal hyperactivity by mangiferin can prevent diabetes-associated cognitive decline in rats. Psychopharmacology 228, 585–594 (2013). https://doi.org/10.1007/s00213-013-3061-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-013-3061-5