Abstract
Rationale
Protracted use of methamphetamine (mAMPH) can result in long-term impairments in cognitive function in humans. A previous study reported reversal-specific learning impairments in rats after a binge administration of mAMPH. Several studies show that extended exposure to mAMPH may confer protection against cognitive impairments and the insult to monoamine systems typically observed after larger binge doses.
Objectives
To explore this issue, we compared the effects of escalating and single doses of mAMPH (and saline, SAL) on retention, reversal learning, and post-mortem analysis of dopamine and serotonin transporters, DAT and SERT.
Methods
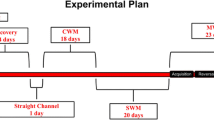
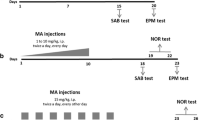
Rats learned to discriminate equiluminant stimuli and then were treated with either: (1) 4 weeks of mAMPH increasing by 0.3 mg/kg, culminating in 6 mg/kg (mAMPHescal); (2) 4 weeks of SAL with a single dose of 6 mg/kg on the last day of treatment (mAMPHsingle); or (3) 4 weeks of SAL. Following treatment, rats were tested on retention and reversal learning, with subsequent analysis of DAT and SERT binding across subregions of the striatum and frontoparietal cortex, respectively.
Results
Retention of the pretreatment discrimination was not significantly impaired in either mAMPH treatment group. A significant decrease in ventrolateral striatal DAT binding was observed only in the mAMPHsingle group and frontoparietal SERT was unaffected by either mAMPH treatment. Both treatment groups demonstrated attenuated reversal learning, particularly on measures of accuracy and effort.
Conclusions
These results show that extended and single-dose pretreatment with mAMPH similarly and selectively affect reversal learning, even in the absence of significant DAT or SERT changes.




Similar content being viewed by others
References
Belcher AM, O’Dell SJ, Marshall JF (2006) A sensitizing regimen of methamphetamine causes impairments in a novelty preference task of object recognition. Behav Brain Res 170:167–172
Belcher AM, Feinstein EM, O’Dell SJ, Marshall JF (2008) Methamphetamine influences on recognition memory: comparison of escalating and single-day dosing regimens. Neuropsychopharmacology 33:1453–1463
Boja JW, Mitchell WM, Patel A, Kopajtic TA, Carroll FI, Lewin AH, Abraham P, Kuhar MJ (1992) High-affinity binding of [125I]RTI-55 to dopamine and serotonin transporters in rat brain. Synapse (N Y) 12:27–36
Cadet JL, Krasnova IN, Ladenheim B, Cai N-S, McCoy MT, Atianjoh FE (2009) Methamphetamine preconditioning: differential protective effects on monoaminergic systems in the rat brain. Neurotox Res 15:252–259
Cass WA, Manning MW (1999) Recovery of presynaptic dopaminergic functioning in rats treated with neurotoxic doses of methamphetamine. J Neurosci 19:7653–7660
Cheng R-K, Etchegaray M, Meck WH (2007) Impairments in timing, temporal memory, and reversal learning linked to neurotoxic regimens of methamphetamine intoxication. Brain Res 1186:255–266
Clark RE, Kuczenski R, Segal DS (2007) Escalating dose, multiple binge methamphetamine regimen does not impair recognition memory in rats. Synapse (N Y) 61:515–522
Cools R, Frank MJ, Gibbs SE, Miyakawa A, Jagust W, D’Esposito M (2009) Striatal dopamine predicts outcome-specific reversal learning and its sensitivity to dopaminergic drug administration. J Neurosci 29:1538–1543
Daberkow DP, Riedy MD, Kesner RP, Keefe KA (2008) Effect of methamphetamine neurotoxicity on learning-induced Arc mRNA expression in identified striatal efferent neurons. Neurotox Res 14:307–315
Featherstone RE, Rizos Z, Kapur S, Fletcher PJ (2008) A sensitizing regimen of amphetamine that disrupts attentional set-shifting does not disrupt working or long-term memory. Behav Brain Res 189:170–179
Fleckenstein AE, Volz TJ, Hanson GR (2009) Psychostimulant-induced alterations in vesicular monoamine transporter-2 function: neurotoxic and therapeutic implications. Neuropharmacology 56:133–138
Fletcher PJ, Tenn CC, Sinyard J, Rizos Z, Kapur S (2007) A sensitizing regimen of amphetamine impairs visual attention in the 5-choice serial reaction time test: reversal by a D1 receptor agonist injected into the medial prefrontal cortex. Neuropsychopharmacology 32:1122–1132
Friedman SD, Castañeda E, Hodge GK (1998) Long-term monoamine depletion, differential recovery, and subtle behavioral impairment following methamphetamine-induced neurotoxicity. Pharmacol Biochem Behav 61:35–44
Fukumura M, Cappon GD, Pu C, Broening HW, Vorhees CV (1998) A single dose model of methamphetamine-induced neurotoxicity in rats: effects on neostriatal monoamines and glial fibrillary acidic protein. Brain Res 806:1–7
Graham DL, Noailles P-AH, Cadet JL (2008) Differential neurochemical consequences of an escalating dose-binge regimen followed by single-day multiple-dose methamphetamine challenges. J Neurochem 105:1873–1885
Groman SM, Lee B, Seu E, James AS, Feiler K, Mandelkern MA, London ED, Jentsch JD (2012) Dysregulation of D2-mediated dopamine transmission in monkeys after chronic escalating methamphetamine exposure. J Neurosci 32:5843–5852
Hoffman WF, Moore M, Templin R, McFarland B, Hitzemann RJ, Mitchell SH (2006) Neuropsychological function and delay discounting in methamphetamine-dependent individuals. Psychopharmacology 188:162–170
Izquierdo A, Jentsch JD (2012) Reversal learning as a measure of impulsive and compulsive behavior in addictions. Psychopharmacology 219:607–620
Izquierdo A, Wiedholz LM, Millstein RA, Yang RJ, Bussey TJ, Saksida LM, Holmes A (2006) Genetic and dopaminergic modulation of reversal learning in a touchscreen-based operant procedure for mice. Behav Brain Res 171:181–188
Izquierdo A, Belcher AM, Scott L, Cazares VA, Chen J, O’Dell SJ, Malvaez M, Wu T, Marshall JF (2010) Reversal-specific learning impairments after a binge regimen of methamphetamine in rats: possible involvement of striatal dopamine. Neuropsychopharmacology 35:505–514
Jocham G, Klein TA, Neumann J, von Cramon Y, Reuter M, Ullsperger M (2009) Dopamine DRD2 polymorphism alters reversal learning and associated neural activity. J Neurosci 29:3695–3704
Jones B, Miskin M (1972) Limbic lesions and the problem of stimulus-reinforcement associations. Exp Neurol 36:362–377
Kesner RP, Churchwell JC (2011) An analysis of rat prefrontal cortex in mediating executive function. Neurobiol Learn Mem 96:417–431
Kosheleff AR, Grimes M, O’Dell SJ, Marshall JF, Izquierdo A (2011) Work aversion and associated changes in dopamine and serotonin transporter after methamphetamine exposure in rats. Psychopharmacology 219:411–420
Lucantonio F, Stalnaker TA, Shaham Y, Niv Y, Schoenbaum G (2012) The impact of orbitofrontal dysfunction on cocaine addiction. Nat Neurosci 15:358–366
Madden LJ, Flynn CT, Zandonatti MA, May M, Parsons LH, Katner SN, Henriksen SJ, Fox HS (2005) Modeling human methamphetamine exposure in nonhuman primates: chronic dosing in the rhesus macaque leads to behavioral and physiological abnormalities. Neuropsychopharmacology 30:350–359
Marshall JF, Belcher AM, Feinstein EM, O’Dell SJ (2007) Methamphetamine-induced neural and cognitive changes in rodents. Addict 102:61–69
Melega WP, Raleigh MJ, Stout DB, Lacan G, Huang SC, Phelps ME (1997) Recovery of striatal dopamine function after acute amphetamine- and methamphetamine-induced neurotoxicity in the vervet monkey. Brain Res 766:113–120
Meredith CW, Jaffe C, Ang-Lee K, Saxon AJ (2005) Implications of chronic methamphetamine use: a literature review. Harv Rev Psychiatr 13:141–154
National Research Council of the National Academies (2011) Guide for the care and use of laboratory animals, 8th edn. The National Academies Press, Washington
Nordahl TE, Salo R, Leamon M (2003) Neuropsychological effects of chronic methamphetamine use on neurotransmitters and cognition: a review. J Neuropsychiatry Clin Neurosci 15:317–325
O’Dell SJ, Galvez BA, Ball AJ, Marshall JF (2012) Running wheel exercise ameliorates methamphetamine-induced damage to dopamine and serotonin terminals. Synapse (N Y) 66:71–80
Parsegian A, Glen WB, Lavin A, See RE (2011) Methamphetamine self-administration produces attentional set-shifting deficits and alters prefrontal cortical neurophysiology in rats. Biol Psychiatry 69:253–259
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates. Elsevier, London
Perry JL, Carroll ME (2008) The role of impulsive behavior in drug abuse. Psychopharmacology 200:1–26
Ragozzino ME (2002) The effects of dopamine D(1) receptor blockade in the prelimbic-infralimbic areas on behavioral flexibility. Learn Mem 9:18–28
Ragozzino ME, Ragozzino KE, Mizumori SJY, Kesner RP (2002) Role of the dorsomedial striatum in behavioral flexibility for response and visual cue discrimination learning. Behav Neurosci 116:105–115
Reichel CM, Schwendt M, McGinty JF, Olive MF, See RE (2011) Loss of object recognition memory produced by extended access to methamphetamine self-administration is reversed by positive allosteric modulation of metabotropic glutamate receptor 5. Neuropsychopharmacology 36:782–792
Ricaurte GA, Schuster CR, Seiden LS (1980) Long-term effects of repeated methylamphetamine administration on dopamine and serotonin neurons in the rat brain: a regional study. Brain Res 193:153–163
Robinson TE, Berridge KC (2003) Addiction. Annu Rev Psychol 54:25–53
Rogers JL, De Santis S, See RE (2008) Extended methamphetamine self-administration enhances reinstatement of drug seeking and impairs novel object recognition in rats. Psychopharmacology 199:615–624
Salo R, Nordahl TE, Possinc K, Leamon M, Gibsond DR, Galloway GP, Flynnd NM, Henikf A, Pfefferbaum A, Sullivang EV (2002) Preliminary evidence of reduced cognitive inhibition in methamphetamine-dependent individuals. Psychiatry Res 111:65–74
Schmidt CJ, Ritter JK, Sonsalla PK, Hanson GR, Gibb JW (1985) Role of dopamine in the neurotoxic effects of methamphetamine. J Pharmacol Exp Ther 233:539–544
Schröder N, O’Dell SJ, Marshall JF (2003) Neurotoxic methamphetamine regimen severely impairs recognition memory in rats. Synapse (N Y) 49:89–96
Segal DS, Kuczenski R (1997) Repeated binge exposures to amphetamine and methamphetamine: behavioral and neurochemical characterization. J Pharmacol Exp Ther 282:561–573
Segal DS, Kuczenski R, O’Neil ML, Melega WP, Cho AK (2003) Escalating dose methamphetamine pretreatment alters the behavioral and neurochemical profiles associated with exposure to a high-dose methamphetamine binge. Neuropsychopharmacology 28:1730–1740
Shepard JD, Bossert JM, Liu SY, Shaham Y (2004) The anxiogenic drug yohimbine reinstates methamphetamine seeking in a rat model of drug relapse. Biol Psychiatry 55:1082–1089
Simon SL, Richardson K, Dacey J, Glynn S, Domier CP, Rawson RA, Ling W (2002) A comparison of patterns of methamphetamine and cocaine use. J Addict Dis 21:35–44
Verdejo-Garcia A, Bechara A, Recknor EC, Perez-Garcia M (2006) Executive dysfunction in substance dependent individuals during drug use and abstinence: an examination of the behavioral, cognitive and emotional correlates of addiction. J Int Neuropsychol Soc 12:405–415
Wagner GC, Ricaurte GA, Seiden LS, Schuster CR, Miller RJ, Westley J (1980) Long-lasting depletions of striatal dopamine and loss of dopamine uptake sites following repeated administration of methamphetamine. Brain Res 181:151–160
White IM, Minamoto T, Odell JR, Mayhorn J, White W (2009) Brief exposure to methamphetamine (METH) and phencyclidine (PCP) during late development leads to long-term learning deficits in rats. Brain Res 1266:72–86
Yamamoto BK, Moszczynska A, Gudelsky GA (2010) Amphetamine toxicities. Ann N Y Acad Sci 1187:101–121
Acknowledgments
This work was supported by 1SC2MH087974 (Izquierdo) and 1RO1 DA012204 (Marshall). We thank the CSULA Animal Care staff for their excellent support. We are grateful to Millie Grimes, Vivian Zeng, Chelsi Darling, and Aviva Sterns for their help with behavioral testing and data collection.
Conflict of interest
There is nothing to disclose nor are there any conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kosheleff, A.R., Rodriguez, D., O’Dell, S.J. et al. Comparison of single-dose and extended methamphetamine administration on reversal learning in rats. Psychopharmacology 224, 459–467 (2012). https://doi.org/10.1007/s00213-012-2774-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-012-2774-1