Abstract
Rationale
Individuals learn associations between alcohol’s sensory properties and intoxication, with such conditioned stimuli (CS) becoming involved in craving and relapse. However, these CS also carry idiosyncratic associations.
Objectives
This study aimed to test brain responses to novel CS conditioned with alcohol intoxication.
Methods
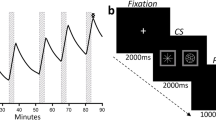
Fourteen heavy drinkers (age 24.9 ± 3.2) performed a reaction time task with embedded novel geometric CS and were told only that the task was to measure alcohol’s effect on speed. Rapid intravenous alcohol infusion (the unconditioned stimulus; UCS) began with the appearance of a CS+, using pharmacokinetic modeling to increment breath alcohol by ~18 mg% in 200 s per each of six CS–UCS pairings. Placebo–saline infusion with CS− used the same infusion parameters in same-day randomized/counterbalanced sessions. The next morning subjects, connected to inactive intravenous pumps, underwent functional magnetic resonance imaging (fMRI) of the same task with mixed brief presentations of CS+, CS−, and irrelevant CS and were told that alcohol could be infused at any time during imaging.
Results
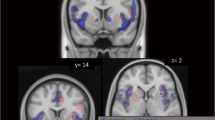
CS− responses were significantly greater than those of CS+ in medial frontal cortex. Notably, CS+ responses were negative, suggesting reduced neural activity. Negative activity was most pronounced in early scans, extinguishing with time. As subjects were told that alcohol could be administered in fMRI, a CS+ without alcohol is similar to a negative prediction error, with associated reduced frontal activity during withheld reward.
Conclusions
Novel stimuli relatively free of demand characteristics can be classically conditioned to intermittent brain exposure of even low alcohol concentrations, permitting imaging studies of conditioned alcohol expectancies.





Similar content being viewed by others
References
Abler B, Walter H, Erk S, Kammerer H, Spitzer M (2006) Prediction error as a linear function of reward probability is coded in human nucleus accumbens. NeuroImage 31:790–795
Bragulat V, Dzemidzic M, Talavage T, Davidson D, O’Connor SJ, Kareken DA (2008) Alcohol sensitizes cerebral responses to the odors of alcoholic drinks: an fMRI study. Alcohol Clin Exp Res 32:1124–1134
Bray S, O’Doherty J (2007) Neural coding of reward-prediction error signals during classical conditioning with attractive faces. J Neurophysiol 97:3036–3045
Brett M, Anton J-L, Valabregue R, Poline J-B (2002) Region of interest analysis using an SPM toolbox. NeuroImage 16:2 [Abstract]
Carter BL, Tiffany ST (1999) Meta-analysis of cue-reactivity in addiction research. Addiction 94:327–340
Chiba T, Kayahara T, Nakano K (2001) Efferent projections of infralimbic and prelimbic areas of the medial prefrontal cortex in the Japanese monkey, Macaca fuscata. Brain Res 888:83–101
Cooney NL, Litt MD, Morse PA, Bauer LO (1997) Alcohol cue reactivity, negative-mood reactivity, and relapse in treated alcoholic men. J Abnorm Psychol 106:243–250
Cox WM, Hogan LM, Kristian MR, Race JH (2002) Alcohol attentional bias as a predictor of alcohol abusers’ treatment outcome. Drug Alcohol Depend 68:237–243
Field M, Duka T (2002) Cues paired with a low dose of alcohol acquire conditioned incentive properties in social drinkers. Psychopharmacology 159:325–334
Field M, Mogg K, Zetteler J, Bradley BP (2004) Attentional biases for alcohol cues in heavy and light social drinkers: the roles of initial orienting and maintained attention. Psychopharmacology 176:88–93
Filbey FM, Ray L, Smolen A, Claus ED, Audette A, Hutchison KE (2008a) Differential neural response to alcohol priming and alcohol taste cues is associated with DRD4 VNTR and OPRM1 genotypes. Alcohol Clin Exp Res 32:1113–1123
Filbey FM, Claus E, Audette AR, Niculescu M, Banich MT, Tanabe J, Du YP, Hutchison KE (2008b) Exposure to the taste of alcohol elicits activation of the mesocorticolimbic neurocircuitry. Neuropsychopharmacology 33:1391–1401
Fiorillo CD, Tobler PN, Schultz W (2003) Discrete coding of reward probability and uncertainty by dopamine neurons. Science 299:1898–1902
Freeman N, Friedman RS, Bartholow BD, Wulfert E (2010) Effects of alcohol priming on social disinhibition. Exp Clin Psychopharmacol 18:135–144
Friedman RS, McCarthy DM, Bartholow BD, Hicks JA (2007) Interactive effects of alcohol outcome expectancies and alcohol cues on nonconsumptive behavior. Exp Clin Psychopharmacol 15:102–114
Glautier S, Drummond C, Remington B (1994) Alcohol as an unconditioned stimulus in human classical conditioning. Psychopharmacology 116:360–368
Glautier S, Bankart J, Williams A (2000) Flavour conditioning and alcohol: a multilevel model of individual differences. Biol Psychol 52:17–36
Goldman MS (2002) Expectancy and risk for alcoholism: the unfortunate exploitation of a fundamental characteristic of neurobehavioral adaptation. Alcohol Clin Exp Res 26:737–746
Haber SN, Kim KS, Mailly P, Calzavara R (2006) Reward-related cortical inputs define a large striatal region in primates that interface with associative cortical connections, providing a substrate for incentive-based learning. J Neurosci 26:8368–8376
Hare TA, O’Doherty J, Camerer CF, Schultz W, Rangel A (2008) Dissociating the role of the orbitofrontal cortex and the striatum in the computation of goal values and prediction errors. J Neurosci 28:5623–5630
Hare TA, Camerer CF, Rangel A (2009) Self-control in decision-making involves modulation of the vmPFC valuation system. Science 324:646–648
Hollerman JR, Schultz W (1998) Dopamine neurons report an error in the temporal prediction of reward during learning. Nat Neurosci 1:304–309
Kable JW, Glimcher PW (2007) The neural correlates of subjective value during intertemporal choice. Nat Neurosci 10:1625–1633
Kareken DA, Bragulat V, Dzemidzic M, Cox C, Talavage T, Davidson D, O’Connor SJ (2010a) Family history of alcoholism mediates the frontal response to alcoholic drink odors and alcohol in at-risk drinkers. NeuroImage 50:267–276
Kareken DA, Liang T, Wetherill L, Dzemidzic M, Bragulat V, Cox C, Talavage T, O’Connor SJ, Foroud T (2010b) A polymorphism in GABRA2 is associated with the medial frontal response to alcohol cues in an fMRI study. Alcohol Clin Exp Res 34:2169–2178
Kim H, Shimojo S, O’Doherty JP (2006) Is avoiding an aversive outcome rewarding? Neural substrates of avoidance learning in the human brain. PLoS Biol 4:e233
Knutson B, Fong GW, Adams CM, Varner JL, Hommer D (2001) Dissociation of reward anticipation and outcome with event-related fMRI. NeuroReport 12:3683–3687
Knutson B, Fong GW, Bennett SM, Adams CM, Hommer D (2003) A region of mesial prefrontal cortex tracks monetarily rewarding outcomes: characterization with rapid event-related fMRI. NeuroImage 18:263–272
Kramer DA, Goldman MS (2003) Using a modified Stroop task to implicitly discern the cognitive organization of alcohol expectancies. J Abnorm Psychol 112:171–175
Lapish CC, Seamans JK, Judson CL (2006) Glutamate-dopamine cotransmission and reward processing in addiction. Alcohol Clin Exp Res 30:1451–1465
Litt MD, Cooney NL, Morse P (2000) Reactivity to alcohol-related stimuli in the laboratory and in the field: predictors of craving in treated alcoholics. Addiction 95:889–900
McCaul ME, Turkkan JS, Stitzer ML (1989) Conditioned opponent responses: effects of placebo challenge in alcoholic subjects. Alcohol Clin Exp Res 13:631–635
McClure SM, Berns GS, Montague PR (2003) Temporal prediction errors in a passive learning task activate human striatum. Neuron 38:339–346
McClure SM, Ericson KM, Laibson DI, Loewenstein G, Cohen JD (2007) Time discounting for primary rewards. J Neurosci 27:5796–5804
Mirenowicz J, Schultz W (1996) Preferential activation of midbrain dopamine neurons by appetitive rather than aversive stimuli. Nature 379:449–451
Myrick H, Anton RF, Li X, Henderson S, Randall PK, Voronin K (2008) Effect of naltrexone and ondansetron on alcohol cue-induced activation of the ventral striatum in alcohol-dependent people. Arch Gen Psychiatry 65:466–475
O’Connor S, Morzorati S, Christian J, Li TK (1998) Clamping breath alcohol concentration reduces experimental variance: application to the study of acute tolerance to alcohol and alcohol elimination rate. Alcohol Clin Exp Res 22:202–210
O’Doherty JP, Dayan P, Friston K, Critchley H, Dolan RJ (2003) Temporal difference models and reward-related learning in the human brain. Neuron 38:329–337
Pagnoni G, Zink CF, Montague PR, Berns GS (2002) Activity in human ventral striatum locked to errors of reward prediction. Nat Neurosci 5:97–98
Ramchandani VA, Bolane J, Li TK, O’Connor S (1999) A physiologically-based pharmacokinetic (PBPK) model for alcohol facilitates rapid BrAC clamping. Alcohol Clin Exp Res 23:617–623
Ramchandani VA, O’Connor S, Neumark Y, Zimmermann US, Morzorati SL, Wit H (2006) The alcohol clamp: applications, challenges, and new directions—an RSA 2004 symposium summary. Alcohol Clin Exp Res 30:155–164
Ramnani N, Elliott R, Athwal BS, Passingham RE (2004) Prediction error for free monetary reward in the human prefrontal cortex. NeuroImage 23:777–786
Redish AD, Jensen S, Johnson A, Kurth-Nelson Z (2007) Reconciling reinforcement learning models with behavioral extinction and renewal: implications for addiction, relapse, and problem gambling. Psychol Rev 114:784–805
Rescorla RA, Wagner AR (1972) A theory of Pavlovian conditioning: variations in the effectiveness of reinforcement and nonreinforcement. In: Black AH, Prokasy WF (eds) Classical conditioning II: current research and theory. Appleton, New York, pp 64–99
Rodriguez PF, Aron AR, Poldrack RA (2006) Ventral striatal/nucleus accumbens sensitivity to prediction errors during classification learning. Hum Brain Mapp 27:306–313
Roehrich L, Goldman MS (1995) Implicit priming of alcohol expectancy memory processes and subsequent drinking behavior. Exp Clin Psychopharmacol 3:402–410
Saunders JB, Aasland OG, Babor TF, de la Fuente JR, Grant M (1993) Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption—II. Addiction 88:791–804
Schott BH, Minuzzi L, Krebs RM, Elmenhorst D, Lang M, Winz OH, Seidenbecher CI, Coenen HH, Heinze HJ, Zilles K, Duzel E, Bauer A (2008) Mesolimbic functional magnetic resonance imaging activations during reward anticipation correlate with reward-related ventral striatal dopamine release. J Neurosci 28:14311–14319
Schultz W, Dayan P, Montague PR (1997) A neural substrate of prediction and reward. Science 275:1593–1599
Shmuel A, Yacoub E, Pfeuffer J, Van de Moortele P-F, Adriany G, Hu X, Ugurbil K (2002) Sustained negative BOLD, blood flow and oxygen consumption response and its coupling to the positive response in the human brain. Neuron 36:1195–1210
Sobell MB, Sobell LC, Klajner F, Pavan D, Basian E (1986) The reliability of a timeline method for assessing normal drinker college students’ recent drinking history: utility for alcohol research. Addict Behav 11:149–161
Stacy AW (1997) Memory activation and expectancy as prospective predictors of alcohol and marijuana use. J Abnorm Psychol 106:61–73
Staiger PK, White JM (1988) Conditioned alcohol-like and alcohol-opposite responses in humans. Psychopharmacology 95:87–91
Tapert SF, Cheung EH, Brown GG, Frank LR, Paulus MP, Schweinsburg AD, Meloy MJ, Brown SA (2003) Neural response to alcohol stimuli in adolescents with alcohol use disorder. Arch Gen Psychiatry 60:727–735
Thesen S, Heid O, Mueller E, Schad LR (2000) Prospective acquisition correction for head motion with image-based tracking for real-time fMRI. Magn Reson Med 44:457–465
Townshend JM, Duka T (2001) Attentional bias associated with alcohol cues: differences between heavy and occasional social drinkers. Psychopharmacology 157:67–74
Williams SM, Goldman-Rakic PS (1998) Widespread origin of the primate mesofrontal dopamine system. Cerebral Cortex 8:321–345
Yoder KK, Morris ED, Constantinescu CC, Cheng TE, Normandin MD, O’Connor SJ, Kareken DA (2009) When what you see isn’t what you get: alcohol cues, alcohol administration, prediction error, and human striatal dopamine. Alcohol Clin Exp Res 33:139–149
Acknowledgments
This study was supported by R21 AA018020 (DAK), R01 AA017661 (DAK), the Indiana Alcohol Research Center P60 AA007611, T32 AA007462, and the Indiana University Clinical Research Center (RR 025761). We gratefully acknowledge Kyle Windisch (Department of Psychiatry) for paradigm programming, Michele Beal and Courtney Robbins for assistance with imaging (Department of Radiology), and Kieren Mather, M.D. (Department of Medicine) for assistance with medical supervision.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kareken, D.A., Grahame, N., Dzemidzic, M. et al. fMRI of the brain’s response to stimuli experimentally paired with alcohol intoxication. Psychopharmacology 220, 787–797 (2012). https://doi.org/10.1007/s00213-011-2526-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-011-2526-7