Abstract
Background
Corticotropin-releasing factor receptor type 1 (CRF1) antagonists have been proposed as therapeutic agents in the treatment of mood and anxiety disorders although clinical evidence supporting their development and understanding of a dose–response relationship has been lacking.
Methods
We tested two doses of the CRF1 antagonist R317573 for effects on regional cerebral glucose metabolism (rCMglu) using [18F] fluoro-2-deoxy-d-glucose (FDG) positron emission tomography (PET) following single-dose challenges in a double-blind, placebo-controlled, cross-over design, in 12 healthy male volunteers.
Results
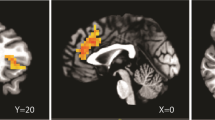
Single 30- and 200-mg doses of R317573 resulted in dose-related changes in rCMglu. Relative increases in rCMglu were observed in frontal cortical regions while relative decreases occurred in the putamen and right amygdala after both doses. Relative decreases occurred in cerebellum and right parahippocampal gyrus following the higher dose.
Conclusions
R317573 appears to produce acute dose-dependent changes in rCMglu. Effects occurred in regions that may be behaviorally relevant to mood and anxiety disorders. In some regions, these effects may be related to the receptor (target) density. Measuring acute effects on rCMglu with FDG-PET may offer a method for defining pharmacologically active doses for central nervous system targets for which selective radiotracers are lacking.



Similar content being viewed by others
References
Binneman B, Feltner D, Kolluri S, Shi Y, Qiu R, Stiger T (2008) A 6-week randomized, placebo-controlled trial of CP-316, 311 (a selective CRH1 antagonist) in the treatment of major depression. Am J Psychiatry 165(5):617–620
Cha CI, Foote SL (1988) Corticotropin-releasing factor in olivocerebellar climbing-fiber system of monkey (Saimiri sciureus and Macaca fascicularis): parasagittal and regional organization visualized by immunohistochemistry. J Neurosci 8(11):4121–4137
Charney DS, Drevets WC (2002) The neurobiological basis of anxiety disorders. Chapter 63. In: Davis K, Charney DS, Coyle J, Nemeroff CB (eds) Psychopharmacology: the fifth generation of progress. Williams and Wilkins, New York
Cook EH, Metz J, Leventhal BL, Lebovitz M, Nathan M, Semerdjian SA et al (1994) Fluoxetine effects on cerebral glucose metabolism. NeuroReport 5:1745–1748
Davis M (2005) Neural circuitry of anxiety and stress disorders. Chapter 64. In: Davis K, Charney DS, Coyle J, Nemeroff CB (eds) Psychopharmacology: the fifth generation of progress. Williams and Wilkins, New York
de Leon MJ, McRae T, Rusinek H, Convit A, De Santi S, Tarshish C et al (1997) Cortisol reduces hippocampal glucose metabolism in normal elderly, but not in Alzheimer’s disease. J Clin Endocrinol Metab 82(10):3251–3259
Dunn AJ, Berridge CW (1990) Physiological and behavioral responses to corticotropin-releasing factor administration: is CRF a mediator of anxiety or stress responses? Brain Res Brain Res Rev 15(2):71–100
Faul F, Erdfelder E, Lang AG, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39(2):175–191
Funk D, Li Z, Shaham Y, Lê AD (2003) Effect of blockade of corticotropin-releasing factor receptors in the median raphe nucleus on stress-induced c-fos mRNA in the rat brain. Neuroscience 122(1):1–4
Gallopin T, Geoffroy H, Rossier J, Lambolez B (2006) Cortical sources of CRF, NKB, and CCK and their effects on pyramidal cells in the neocortex. Cerebral Cortex 16:1440–1452
Heiss WD, Holghoff V, Pawlik G, Neveling M (1990) Effect of nimodipine on regional cerebral glucose metabolism in patients with acute ischemic stroke as measured by positron emission tomography. J Cereb Blood Flow Metab 1:127–132
Hunter GJ, Hamberg LM, Alpert NM, Choi NC, Fischman AJ (1996) Simplified measurement of deoxyglucose utilization rate. J Nucl Med 37(6):950–955
Ising M, Zimmermann US, Künzel HE, Uhr M, Foster AC, Learned-Coughlin SM et al (2007) High-affinity CRF1 receptor antagonist NBI-34041: preclinical and clinical data suggest safety and efficacy in attenuating elevated stress response. Neuropsychopharmacology 32(9):1941–1949
Jochman KA, Newman SM, Kalin NH, Bakshi VP (2005) Corticotropin-releasing factor-1 receptors in the basolateral amygdala mediate stress-induced anorexia. Behav Neurosci 119(6):1448–1458
Kumar JS, Majo VJ, Sullivan GM, Prabhakaran J, Simpson NR, Van Heertum RL et al (2006) Synthesis and in vivo evaluation of [11C]SN003 as a PET ligand for CRF1 receptors. Bioorg Med Chem 14(12):4029–4034
Millan MA, Samra AB, Wynn PC, Catt KJ, Aguilera G (1987) Receptors and actions of corticotropin-releasing hormone in the primate pituitary gland. J Clin Endocrinol Metab 64(5):1036–1041
Palamarchouk VS, Swiergiel AH, Dunn AJ (2002) Hippocampal noradrenergic responses to CRF injected into the locus coeruleus of unanesthetized rats. Brain Res 950(1–2):31–38
Powers RE, De Souza EB, Walker LC, Price DL, Vale WW, Young WS III (1987) Corticotropin-releasing factor as a transmitter in the human olivocerebellar pathway. Brain Res 415:347–352
Price ML, Lucki I (2001) Regulation of serotonin release in the lateral septum and striatum by corticotropin-releasing factor. J Neurosci 21(8):2833–2841
Sánchez MM, Young LJ, Plotsky PM, Insel TR (1999) Autoradiographic and in situ hybridization localization of corticotropin-releasing factor 1 and 2 receptors in nonhuman primate brain. J Comp Neurol 408(3):365–3677
Schmidt ME, Matochik JA, Goldstein DS, Schouten JL, Zametkin AJ, Potter WZ (1997) Gender differences in brain metabolic and plasma catecholamine responses to alpha 2 adrenoceptor blockade. Neuropsychopharmacology 16:298–310
Schmidt ME, Oshinsky RJ, Kim H-G, Schouten JL, Folley BS, Potter WZ (1999) Cerebral glucose metabolic and plasma catecholamine responses to the alpha-2 adrenoceptor antagonist ethoxyidazoxan given to healthy volunteers. Psychopharmacology 146:119–127
Sharkey J, Appel NM, De Souza EB (1989) Alterations in local cerebral glucose utilization following central administration of corticotropin-releasing factor in rats. Synapse 4(1):80–87
Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, Hergueta T, Baker R, Dunbar GC (1998) The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 59(Suppl 20):22–33
Smagin GN, Zhou J, Harris RB, Ryan DH (1997) CRF receptor antagonist attenuates immobilization stress-induced norepinephrine release in the prefrontal cortex in rats. Brain Res Bull 42(6):431–434
Steckler T (2008) Peptide receptor ligands to treat anxiety disorders. In: Blanchard RJ, Blanchard DC, Griebel G, Nutt D (eds) Handbook of anxiety and fear, vol 17. Academic, Amsterdam, pp 81–99
Steckler T, Dautzenberg F (2006) Corticotropin-releasing factor antagonists in affective disorders and drug dependence—an update. CNS Neurol Disorders Drug Targets 5(2):147–165
Steckler T, Nakazato A, Kennis L, Mackie C, Nakamura M, Vinken P et al (2006) CRF1 antagonists—therapeutic implications for affective and mood disorders (Antagonistes des récepturs CRF1 antidépresseurs et anxiolytiques). Actualités de Chimie Thérapeutique, Société de Chimie Thérapeutique 32:1–19
Strome EM, Wheler GH, Higley JD, Loriaux DL, Suomi SJ, Doudet DJ (2002) Intracerebroventricular corticotropin-releasing factor increases limbic glucose metabolism and has social context-dependent behavioral effects in nonhuman primates. Proc Natl Acad Sci 99(24):15749–15754
Tuvnes FA, Steffenach HA, Murison R, Moser MB, Moser EI (2003) Selective hippocampal lesions do not increase adrenocortical activity. J Neurosci 23(10):4345–4354
Wald FDM, Mellenbergh GJ (1990) De verkorte versie van de Nederlandse vertaling van de Profile of Mood States (POMS). Ned Tijdschr Psychol 45:86–90
Warnock G, Moechars D, Langlois ST (2009) In vivo evidence for ligand-specific receptor activation in the central CRF system, as measured by local cerebral glucose utilization. Peptides 30:947–954
Zobel AW, Nickel T, Kuenzel HE, Ackl N, Sonntag A, Ising M et al (2000) Effects of the high-affinity corticotropin-releasing hormone receptor 1 antagonist R121919 in major depression: the first 20 patients treated. J Psychiatric Res 34:171–181
Acknowledgments
We would like to thank Taisho Pharmaceutical Co., Ltd. for their efforts in the identification and characterization of R317573 and their collaboration in clinical studies with the compound. We would like to thank Stijn Dirix, Michel Koole, Hendrikje Jeandarme, Dominique Vanderghinste, Tjibbe de Groot, and Marva Bex (University Hospital and K.U. Leuven) and Anne Van Hecken and Jo Van Effen (Center for Clinical Pharmacology, University Hospital Gasthuisberg, K.U. Leuven) for their skillful help during tracer production, project management, and data acquisition. This study was sponsored by Johnson & Johnson Pharmaceutical Research and Development (J&JPRD), Beerse, Belgium. Study number: R317573DEP1005.
Disclosure/Conflict of interest
MES, PvdA, EM, and TS are employees of J&JPRD. RA, TB, JdH, and KVL have provided professional services to J&JPRD based on service provider contracts.
Author information
Authors and Affiliations
Corresponding author
Additional information
Clinical trial identification: Study number: R317573DEP1005; title: A randomized, double-blind, placebo-controlled, crossover study in healthy young male subjects to explore acute effects of R317573 on regional brain glucose metabolism using [18F] fluoro-deoxyglucose (FDG) positron emission tomography (PET); Eudract: 2006-002220-42.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 7 kb)
Rights and permissions
About this article
Cite this article
Schmidt, M.E., Andrews, R.D., van der Ark, P. et al. Dose-dependent effects of the CRF1 receptor antagonist R317573 on regional brain activity in healthy male subjects. Psychopharmacology 208, 109–119 (2010). https://doi.org/10.1007/s00213-009-1714-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-009-1714-1