Abstract
Rationale
Cannabis is the most widely consumed drug associated with 3,4-methylenedioxymethamphetamine (MDMA) use.
Objectives
This study examines whether low doses of MDMA and delta-9-tetrahydrocannabinol (THC) produce synergistic rewarding/reinforcing effects in mice using the conditioned place preference (CPP) and operant self-administration paradigms. Changes in dopamine (DA) outflow were monitored in the nucleus accumbens (NAC) after single or combined administration of these compounds.
Results
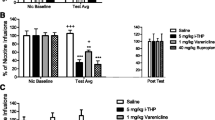
MDMA induced a significant CPP at the dose of 10 mg/kg but not at the dose of 3 mg/kg. THC (0.3 mg/kg) by itself was also ineffective in this paradigm. The combined administration of the low dose of MDMA (3 mg/kg) and THC (0.3 mg/kg) produced CPP, whereas the combination of MDMA (10 mg/kg) and THC (0.3 mg/kg) significantly decreased CPP. Animals treated with THC self-administered a sub-threshold dose of MDMA (0.06 mg/kg per infusion), while animals receiving vehicle did not. However, THC did not modify the self-administration of an effective dose of MDMA (0.125 mg/kg per infusion). In microdialysis studies, a low dose of THC significantly increased DA outflow in the NAC, while a low dose of MDMA did not. When MDMA was administered before THC, DA levels decreased with respect to THC. However, when THC was administered before MDMA, DA levels were not significantly modified with respect to THC.
Conclusions
These results demonstrate that a low dose of THC modifies in different ways (increases and decreases) the sensitivity of animals to the behavioural effects of MDMA and that THC and MDMA converge at a common mechanism modulating DA outflow in the NAC of mice.





Similar content being viewed by others
References
Beardsley PM, Balster RL, Harris LS (1986) Self-administration of methylenedioxy-methamphetamine (MDMA) by rhesus monkeys. Drug Alcohol Depend 18:149–57
Berrendero F, Maldonado R (2002) Involvement of the opioid system in the anxiolytic-like effects induced by delta(9)-tetrahydrocannabinol. Psychopharmacology 163:111–117
Bilsky EJ, Reid LD (1991) MDL72222, a serotonin 5-HT3 receptor antagonist, blocks MDMA’s ability to establish a conditioned place preference. Pharmacol Biochem Behav 39:509–512
Bilsky EJ, Hui Y, Hubbell CL, Reid LD (1990) Methylenedioxymethamphetamine’s capacity to establish place preferences and modify intake of an alcoholic beverage. Pharmacol Biochem Behav 37:633–638
Braida D, Sala M (2002) Role of the endocannabinoid system in MDMA intracerebral self-administration in rats. Br J Pharmacol 136:1089–1092
Braida D, Iosue S, Pegorini S, Sala M (2005) 3,4 Methylenedioxymethamphetamine-induced conditioned place preference (CPP) is mediated by endocannabinoid system. Pharmacol Res 51:177–182
Carboni E, Spielewoy C, Vacca C, Nosten-Bertrand M, Giros B, Di Chiara G (2001) Cocaine and amphetamine increase extracellular dopamine in the nucleus accumbens of mice lacking the dopamine transporter gene. J Neurosci 21(RC141):1–4
Chen JP, Paredes W, Li J, Smith D, Lowinson J, Gardner EL (1990) Delta 9-tetrahydrocannabinol produces naloxone-blockable enhancement of presynaptic basal dopamine efflux in nucleus accumbens of conscious, freely-moving rats as measured by intracerebral microdialysis. Psychopharmacology 102:156–162
Fantegrossi WE, Ullrich T, Rice KC, Woods JH, Winger G (2002) 3,4-Methylenedioxymethamphetamine (MDMA, “ecstasy”) and its stereoisomers as reinforcers in rhesus monkeys: serotoninergic involvement. Psychopharmacology 161:356–364
Gouzoulis-Mayfrank E, Daumann J (2006) The confounding problem of polydrug use in recreational ecstasy/MDMA users: a brief overview. J Psychopharmacol 20:188–193
Justinova Z, Tanda G, Redhi GH, Goldberg SR (2003) Self-administration of delta9-tetrahydrocannabinol (THC) by drug naive squirrel monkeys. Psychopharmacology (Berl) 169:135–140
Kankaanpää A, Meririnne E, Lillsunde P, Seppala T (1998) The acute effects of amphetamine derivatives on extracellular serotonin and dopamine levels in rat nucleus accumbens. Pharmacol Biochem Behav 59:1003–1009
Lamb RJ, Griffiths RR (1987) Self-injection of d,1–3,4-methylenedioxymethamphetamine (MDMA) in the baboon. Psychopharmacology 91:268–272
Lepore M, Vorel SR Lowinson J, Gardner EL (1995) Conditioned place preference induced by delta 9-tetrahydrocannabinol: comparison with cocaine, morphine, and food reward. Life Sci 56:2073–2080
Malone DT, Taylor DA (1999) Modulation by fluoxetine of striatal dopamine release following Delta9-tetrahydrocannabinol: a microdialysis study in conscious rats. Br J Pharmacol 128:21–26
Marona-Lewicka D, Rhee GS, Sprague JE, Nichols DE (1996) Reinforcing effects of certain serotonin-releasing amphetamine derivatives. Pharmacol Biochem Behav 53:99–105
Morley KC, Li KM, Hunt GE, Mallet PE, McGregor IS (2004) Cannabinoids prevent the acute hyperthermia and partially protect against the 5-HT depleting effects of MDMA (“Ecstasy”) in rats. Neuropharmacology 46:954–965
Paxinos G, Franklin KBJ (1997) The mouse brain in stereotaxic coordinates. Academic, San Diego
Ratzenboeck E, Saria A, Kriechbaum N, Zernig G (2001) Reinforcing effects of MDMA (“ecstasy”) in drug-naive and cocaine-trained rats. Pharmacology 62:138–144
Robledo P, Balerio G, Berrendero F, Maldonado R (2004a) Study of the behavioural responses related to the potential addictive properties of MDMA in mice. Naunyn Schmiedebergs Arch Pharmacol 369:338–349
Robledo P, Mendizabal V, Ortuno J, de la Torre R, Kieffer BL, Maldonado R (2004b) The rewarding properties of MDMA are preserved in mice lacking mu-opioid receptors. Eur J Neurosci 20:853–858
Salzmann J, Marie-Claire C, Le Guen S, Roques BP, Noble F (2003) Importance of ERK activation in behavioural and biochemical effects induced by MDMA in mice. Br J Pharmacol 140:831–838
Schenk S, Gittings D, Johnstone M, Daniela E (2003) Development, maintenance and temporal pattern of self-administration maintained by ecstasy (MDMA) in rats. Psychopharmacology 169:21–27
Soria G, Mendizabal V, Tourino C, Robledo P, Ledent C, Parmentier M, Maldonado R, Valverde O (2005) Lack of CB1 cannabinoid receptor impairs cocaine self-administration. Neuropsychopharmacology 30:1670–1680
Tanda G, Pontieri FE, Di Chiara G (1997) Cannabinoid and heroin activation of mesolimbic dopamine transmission by a common mu1 opioid receptor mechanism. Science 276:2048–2050
Tossmann P, Boldt S, Tensil MD (2001) The use of drugs within the techno party scene in European metropolitan cities. Eur Addict Res 7:2–23
Trigo JM, Panayi F, Soria G, Maldonado R, Robledo P (2006) A reliable model of intravenous MDMA self-administration in naive mice. Psychopharmacology 184:212–220
Trigo JM, Renoir T, Lanfumey L, Hamon M, Lesch KP, Robledo P, Maldonado R (2007) MDMA self-administration is abolished in serotonin transporter knock-out mice. Biological Psychiatry (in press)
Valjent E, Maldonado R (2000) A behavioural model to reveal place preference to delta 9-tetrahydrocannabinol in mice. Psychopharmacology 147:436–438
Valjent E, Mitchell JM, Besson MJ, Caboche J, Maldonado R (2002) Behavioural and biochemical evidence for interactions between Delta 9-tetrahydrocannabinol and nicotine. Br J Pharmacol 135:564–578
White SR, Obradovic T, Imel KM, Wheaton MJ (1996) The effects of methylenedioxymethamphetamine (MDMA, “Ecstasy”) on monoaminergic neurotransmission in the central nervous system. Prog Neurobiol 49:455–479
Yamamoto BK, Spanos LJ (1988) The acute effects of methylenedioxymethamphetamine on dopamine release in the awake-behaving rat. Eur J Pharmacol 148:195–203
Zimmermann P, Wittchen HU, Waszak F, Nocon A, Hofler M, Lieb R (2005) Pathways into ecstasy use: the role of prior cannabis use and ecstasy availability. Drug Alcohol Depend 79:331–341
Acknowledgements
The authors would like to thank Ms. Dulce Real Muñoz for her expert help in the microdialysis experiments, and Mr. Jordi Ortuño for his assistance with the HPLC methodology. This work was supported by FIS grant number 03/0305, Plan Nacional Sobre Drogas 2005, NIH-NIDA (USA), Extra-mural research project (#5 R01 DA016768), I.S. CARLOS III Redes de grupos ISCIII (# RTA G03/005), Ministerio de Ciencia y Tecnología (# BFU2004–00920/BFI and # GEN2003–20651), Generalitat de Catalunya 2005SGR00131 and GENADDICT LSHM-CT-2004–05166.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Robledo, P., Trigo, J.M., Panayi, F. et al. Behavioural and neurochemical effects of combined MDMA and THC administration in mice. Psychopharmacology 195, 255–264 (2007). https://doi.org/10.1007/s00213-007-0879-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-007-0879-8