Abstract
Rationale
Several studies have suggested that nicotine treatment can modulate the behavioral and neurochemical responses to other psychostimulants, such as cocaine.
Objectives
The current study examined the hypothesis that nicotinic acetylcholine receptor (nAChR) blockade influences the ability of cocaine to elicit increases in extracellular dopamine levels.
Materials and methods
Pharmacological studies using nicotinic antagonists as well as genetic inactivation of β2* nAChRs were used to determine the effect of nAChR blockade on dopamine levels in ventral striatum elicited by acute or repeated administrations of cocaine in mice.
Results
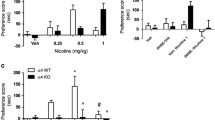
Administration of mecamylamine (a general nicotinic antagonist that is not highly selective for individual nAChR subtypes) or co-administration of methyllycaconitine (a more selective antagonist of α7* nAChRs) with dihydro-β-erythroidine (a more selective antagonist of β2* nAChRs and other heteromeric nAChR subtypes) prevented sensitization of cocaine-elicited increases in extracellular DA levels in the ventral striatum in wild-type mice. In contrast, neither of the more specific antagonists alone was effective in preventing sensitization. Finally, methyllycaconitine administration prevents sensitization in β2−/− mice but not in β2+/+ or wild-type mice.
Conclusions
These data indicate that inhibition of both α7* and β2* nAChRs is necessary to prevent development of sensitization of cocaine-elicited increases in extracellular dopamine levels in the ventral striatum of mice.






Similar content being viewed by others
References
Balfour DJ (2002) Neuroplasticity within the mesoaccumbens dopamine system and its role in tobacco dependence. Curr Drug Target CNS Neurol Disord 1:413–421
Balfour DJ, Benwell ME, Birrell CE, Kelly RJ, Al-Aloul M (1998) Sensitization of the mesoaccumbens dopamine response to nicotine. Pharmacol Biochem Behav 59:1021–1030
Bechtholt AJ, Mark GP (2002) Enhancement of cocaine-seeking behavior by repeated nicotine exposure in rats. Psychopharmacology (Berl) 162:178–185
Blokhina EA, Kashkin VA, Zvartau EE, Danysz W, Bespalov AY (2005) Effects of nicotinic and NMDA receptor channel blockers on intravenous cocaine and nicotine self-administration in mice. Eur Neuropsychopharmacol 15:219–225
Budney AJ, Higgins ST, Hughes JR, Bickel WK (1993) Nicotine and caffeine use in cocaine-dependent individuals. J Subst Abuse 5: 117–130
Champtiaux N, Gotti C, Cordero-Erausquin M, David D-J, Przybylski C, Lena C, Clementi F, Moretti M, Rossi FM, Le Novère N, McIntosh JM, Gardier AM, Changeux JP (2003) Subunit composition of functional nicotinic receptors in dopaminergic neurons investigated with knock-out mice. J Neurosci 23:7820–7829
Damaj MI, Glassco W, Dukat M, Martin BR (1999) Pharmacological characterization of nicotine-induced seizures in mice. J Pharmacol Exp Ther 291:1284–1291
Dani JA, Heinemann S (1996) Molecular and cellular aspects of nicotine abuse. Neuron 16:905–908
Day JC, Piazza PV, Le Moal M, Maccari S (1997) Cocaine- induced increase in cortical acetylcholine release: interaction with the hypothalamo–pituitary–adrenal axis. Eur J Neurosci 9:1130–1136
de Rover M, Mansvelder HD, Lodder JC, Wardeh G, Schoffelmeer AN, Brussaard AB (2004) Long-lasting nicotinic modulation of GABAergic synaptic transmission in the rat nucleus accumbens associated with behavioral sensitization to amphetamine. Eur J Neurosci 19:2859–2870
Dewey SL, Chaurasia CS, Chen CE, Volkow ND, Clarkson FA, Porter SP, Straughter-Moore RM, Alexoff DL, Tedeschi D, Russo NB, Fowler JS, Brodie JD (1997) GABAergic attenuation of cocaine-induced dopamine release and locomotor activity. Synapse 25:393–398
Di Chiara G, Bassareo V, Fenu S, De Luca MA, Spina L, Cadoni C, Acquas E, Carboni E, Valentini V, Lecca D (2004) Dopamine and drug addiction: the nucleus accumbens shell connection. Neuropharmacology 47(Suppl 1):227–241
Erhardt S, Schwieler L, Engberg G (2002) Excitatory and inhibitory responses of dopamine neurons in the ventral tegmental area to nicotine. Synapse 43:227–237
Fadda P, Scherma M, Fresu A, Collu M, Fratta W (2003) Baclofen antagonizes nicotine-, cocaine-, and morphine-induced dopamine release in the nucleus accumbens of rat. Synapse 50:1–6
Gommans J, Stolerman IP, Shoaib M (2000) Antagonism of the discriminative and aversive stimulus properties of nicotine in C57BL/6J mice. Neuropharmacology 39:2840–2847
Grabus SD, Martin BR, Brown SE, Damaj MI (2006) Nicotine place preference in the mouse: influences of prior handling, dose and strain and attenuation by nicotinic receptor antagonists. Psychopharmacology (Berl) 184:456–463
Harvey SC, Maddox FN, Luetje CW (1996) Multiple determinants of dihydro-beta-erythroidine sensitivity on rat neuronal nicotinic receptor alpha subunits. J Neurochem 67:1953–1959
Horger BA, Giles MK, Schenk S (1992) Preexposure to amphetamine and nicotine predisposes rats to self-administer a low dose of cocaine. Psychopharmacology 107:271–276
Imperato A, Obinu MC, Gessa GL (1993) Effects of cocaine and amphetamine on acetylcholine release in the hippocampus and caudate nucleus. Eur J Pharmacol 238:377–381
Klink R, de Kerchove-d’Exaerde A, Zoli M, Changeux JP (2001) Molecular and physiological diversity of nicotinic acetylcholine receptors in the midbrain dopaminergic nuclei. J Neurosci 21:1452–1463
Koob GF, Nestler EJ (1997) The neurobiology of drug addiction. J Neuropsychiatry Clin Neurosci 9:482–497
Levin ED, Mead T, Rezvani AH, Rose JE, Gallivan C, Gross R (2000) The nicotinic antagonist mecamylamine preferentially inhibits cocaine vs. food self-administration in rats. Physiol Behav 71:565–570
Lindsay GB, Rainey J (1997) Psychosocial and pharmacologic explanations of nicotine’s “gateway drug” function. J Sch Health 67:123–126
Mansvelder HD, Keath-R, McGehee DS (2002) Synaptic mechanisms underlie nicotine-induced excitability of brain reward areas. Neuron 33:905–919
Mansvelder HD, de Rover M, McGehee DS, Brussaard AB (2003) Cholinergic modulation of dopaminergic reward areas: upstream and downstream targets of nicotine addiction. Eur J Pharmacol 480:117–123
Mark GP, Hajnal A, Kinney AE, Keys AS (1999) Self-administration of cocaine increases the release of acetylcholine to a greater extent than response- independent cocaine in the nucleus accumbens of rats. Psychopharmacology (Berl) 143:47–53
Marubio LM, Paylor R (2004) Impaired passive avoidance learning in mice lacking central neuronal nicotinic acetylcholine receptors. Neuroscience 129:575–582
Mogg AJ, Whiteaker P, McIntosh JM, Marks M, Collins AC, Wonnacott S (2002) Methyllycaconitine is a potent antagonist of alpha-conotoxin-MII-sensitive presynaptic nicotinic acetylcholine receptors in rat striatum. J Pharmacol Exp Ther 302:197–204
Nestler EJ (2001) Molecular basis of of long-term plasticity underlying addiction. Nat Rev Neurosci 2:119–128
Orr-Urtreger A, Goldner FM, Saeki M, Lorenzo I, Goldberg L, De-Biasi M, Dani JA, Patrick JW, Beaudet AL (1997) Mice deficient in the α7 neuronal nicotinic acetylcholine receptor lack α-bungarotoxin binding sites and hippocampal fast nicotinic currents. J Neurosci 17:9165–9171
Picciotto MR, Zoli M, Rimondini R, Léna C, Marubio LM, Merlo Pich E, Fuxe K, Changeux JP (1998) Acetylcholine receptors containing the beta-2 subunit are involved in the reinforcing properties of nicotine. Nature 391:173–177
Reid MS, Mickalian JD, Delucchi KL, Hall SM, Berger SP (1998) An acute dose of nicotine enhances cue-induced cocaine craving. Drug Alcohol Depend 49:95–104
Reid MS, Mickalian JD, Delucchi KL, Berger SP (1999) A nicotine antagonist, mecamylamine, reduces cue-induced cocaine craving in cocaine- dependent subjects. Neuropsychopharmacology 20:297–307
Schoffelmeer AN, De Vries TJ, Wardeh G, van de Ven HW, Vanderschuren LJ (2002) Psychostimulant-induced behavioral sensitization depends on nicotinic receptor activation. J Neurosci 22:3269–3276
Smith JE, Vaughn TC, Co C (2004) Acetylcholine turnover rates in rat brain regions during cocaine self-administration. J Neurochem 88:502–512
Stolerman IP, Naylor C, Elmer GI, Goldberg SR (1999) Discrimination and self-administration of nicotine by inbred strains of mice. Psychopharmacology (Berl) 141:297–306
Vanderschuren LJ, Kalivas PW (2000) Alterations in dopaminergic and glutamatergic transmission in the induction and expression of behavioral sensitization: a critical review of preclinical studies. Psychopharmacology (Berl) 151:99–120
Whiteaker P, Marks M-J, Grady S-R, Lu Y, Picciotto M-R, Changeux J-P, Collins AC (2000) Pharmacological and null mutation approaches reveal nicotinic receptor diversity. Eur J Pharmacol 393:123–135
Zachariou V, Caldarone BJ, Weathers-Lowin A, George TP, Elsworth JD, Roth RH, Changeux J-P, Picciotto MR (2001) Nicotine receptor inactivation decreases sensitivity to cocaine. Neuropsychopharmacology 24:576–589
Zocchi A, Pert A (1994) Alterations in striatal acetylcholine overflow by cocaine, morphine, and MK-801: relationship to locomotor output. Psychopharmacology (Berl) 115:297–304
Zoli M, Léna C, Picciotto MR, Changeux J-P (1998) Identification of four classes of brain nicotinic receptors using b2-mutant mice. J Neurosci 18:4461–4472
Zoli M, Moretti M, Zanardi A, McIntosh J-M, Clementi F, Gotti C (2002) Identification of the nicotinic receptor subtypes expressed on dopaminergic terminals in the rat striatum. J Neurosci 22:8785–8789
Acknowledgements
We thank Marco Savigni and Delphine Houtteman for skillful technical assistance. This work was supported by grants from the Italian MIUR (MM05152538) (M.Z.) and grants #DA00436, DA14241 and AA15632 from the National Institutes of Health (M. R. P.). AdeKD’E is Research Associate from Fonds National de la Recherche Scientifique (Belgium). The Authors have no financial interest in or financial conflict with the subject matter or the materials discussed in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zanetti, L., de Kerchove D’Exaerde, A., Zanardi, A. et al. Inhibition of both α7* and β2* nicotinic acetylcholine receptors is necessary to prevent development of sensitization to cocaine-elicited increases in extracellular dopamine levels in the ventral striatum. Psychopharmacology 187, 181–188 (2006). https://doi.org/10.1007/s00213-006-0419-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0419-y