Abstract
Rationale
Identifying the long-term neurocognitive sequelae of drug addiction may have important implications for understanding the compulsive, chronically relapsing nature of this brain disorder.
Objectives
Our aim was to investigate the consequences of chronic intravenous self-administration of cocaine or heroin on visual attentional processes in rats.
Methods
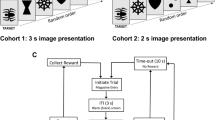
Adult male rats were pretrained on a five-choice serial reaction time task (5-CSRTT) of sustained visual attention and impulsivity and later trained to self-administer cocaine or heroin intravenously during multiple ‘long-access’ self-administration cycles. Control rats had identical training and surgical experience, but received passive infusions of saline during self-administration sessions. Executive cognitive processes of selection and inhibitory response control were evaluated 24 h after drug discontinuation and for a further 6 days prior to the next cycle of self-administration.
Results
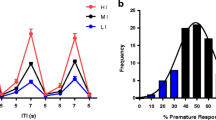
Findings indicate similar behavioural disturbances on the five-choice task in cocaine- and heroin-withdrawn rats with significantly impaired attentional accuracy, increased omissions and slower latencies to respond correctly during the early, but not late, withdrawal period. The self-administration of either drug was not associated with significant alterations in impulsive actions, and there was no evidence of persistent alterations in visual attentional performance. However, unlike rats self-administering cocaine, the motivation to collect food reward on the 5-CSRTT was significantly reduced in heroin-withdrawn animals for a period of at least 6 weeks.
Conclusions
These data, together with recent findings of attentional dysfunction during the withdrawal of intravenous self-administration of amphetamine, suggest that generically different drugs of abuse produce similar disturbances in visual attentional performance during the early withdrawal period.






Similar content being viewed by others
References
Ahmed SH, Koob GF (1999) Long-lasting increase in the set point for cocaine self-administration after escalation in rats. Psychopharmacology 146:303–312
Ahmed SH, Walker JR, Koob GF (2000) Persistent increase in the motivation to take heroin in rats with a history of drug escalation. Neuropsychopharmacology 22:413–421
Ahmed SH, Kenny PJ, Koob GF, Markou A (2002) Neurobiological evidence for hedonic allostasis associated with escalating cocaine use. Nat Neurosci 5:625–626
Beatty WW, Katzung VM, Moreland VJ, Nixon SJ (1995) Neuropsychological performance of recently abstinent alcoholics and cocaine abusers. Drug Alcohol Depend 37:247–253
Ben-Shahar O, Ahmed SH, Koob GF, Ettenberg A (2004) The transition from controlled to compulsive drug use is associated with a loss of sensitization. Brain Res 995:46–54
Berridge KC (1996) Food reward: brain substrates of wanting and liking. Neurosci Biobehav Rev 20:1–25
Berry J, van Gorp W, Herzberg DS, Hinkin C, Boone K, Steinman L, Wilkins JN (1993) Neuropsychological deficits in abstinent cocaine abusers: preliminary findings after two weeks of abstinence. Drug Alcohol Depend 32:231–237
Bolla KI, Rothman R, Cadet JL (1999) Dose-related neurobehavioral effects of chronic cocaine use. J Neuropsychiatry Clin Neurosci 11:361–369
Caine SB, Lintz R, Koob GF (1992) Intravenous drug self-administration techniques in animals. In: Sahgal A (ed) Behavioral neuroscience, a practical approach, vol I. Oxford University Press, Oxford, pp 117–143
Cardinal RN, Aitken MRF (2001) Whisker, version 2.2, computer software. http://www.whisker.control.com
Dalley JW, Theobald DE, Eagle DM, Passetti F, Robbins TW (2002) Deficits in impulse control associated with tonically-elevated serotonergic function in rat prefrontal cortex. Neuropsychopharmacology 26:716–728
Dalley JW, Theobald DEH, Berry D, Milstein J, Laane K, Everitt BJ, Robbins TW (2005) Cognitive sequelae of intravenous amphetamine self-administration in rats: evidence for selective effects on attentional performance. Neuropsychopharmacology 30:525–537
Dawe S, Loxton NJ (2004) The role of impulsivity in the development of substance use and eating disorders. Neurosci Biobehav Rev 28:343–351
Ettenberg A, Petit HO, Bloom FE, Koob GF (1982) Heroin and cocaine intravenous self-administration in rats: mediation by separate neural systems. Psychopharmacology 78:204–209
Evenden JL (1999) Impulsivity: a discussion of clinical and experimental findings. J Psychopharmacol 13(2):180–192
Fillmore MT, Rush CR, Hays L (2002) Acute effects of oral cocaine on inhibitory control of behavior in humans. Drug Alcohol Depend 67:157–167
Gawin FH, Kleber HD (1986) Abstinence symptomatology and psychiatric diagnosis in cocaine abusers. Arch Gen Psychiatry 43:107–113
Goldberg SR, Schuster CR (1967) Conditioned suppression by a stimulus associated with nalorphine in morphine-dependent monkeys. J Exp Anal Behav 10:235–242
Grant I, Adams KM, Carlin AS, Rennick PM, Judd LL, Schoof K (1978) Collaborative neuropsychological study of polydrug users. Arch Gen Psychiatry 35:1063–1074
Harris GC, Aston-Jones G (2003) Altered motivation and learning following opiate withdrawal: evidence for prolonged dysregulation of reward processing. Neuropsychopharmacology 28:865–871
Hester R, Garavan H (2004) Executive dysfunction in cocaine addiction: evidence for discordant frontal, cingulate, and cerebellar activity. J Neurosci 24:11017–11022
Jentsch JD, Taylor JR (1999) Impulsivity resulting from frontostriatal dysfunction in drug abuse: implications for the control of behavior by reward-related stimuli. Psychopharmacology 146:373–390
Kelley AE, Bakshi VP, Haber SN, Steininger TL, Will MJ, Zhang M (2002) Opioid modulation of taste hedonics within the ventral striatum. Physiol Behav 76:365–377
Keppel G (1991) Design and analysis. Prentice Hall, Englewood Cliffs, NJ
Khantzian EJ (1985) The self-medication hypothesis of addictive disorders: focus on heroin and cocaine dependence. Am J Psychiatry 142:1259–1264
Koob GF, Le Moal M (1997) Drug abuse: hedonic homeostatic dysregulation. Science 278:52–58
Kuhar MJ, Pilotte NS (1996) Neurochemical changes in cocaine withdrawal. Trends Pharmacol Sci 17:260–264
Levin FR, Kleber HD (1995) Attention-deficit hyperactivity disorder and substance abuse: relationships and implications for treatment. Harv Rev Psychiatry 2:246–258
Lyvers M (2000) ‘Loss of control’ in alcoholism and drug addiction: a neuroscientific interpretation. Exp Clin Psychopharmacol 8:225–249
Maisonneuve IM, Ho A, Kreek MJ (1995) Chronic administration of a cocaine ‘binge’ alters basal extracellular levels in male rats: an in-vivo microdialysis study. J Pharmacol Exp Ther 272:652–657
Markou A, Koob GF (1991) Postcocaine anhedonia. An animal model of cocaine withdrawal. Neuropsychopharmacology 4:17–26
O'Malley S, Adamse M, Heaton RK, Gawin FH (1992) Neuropsychological impairments in chronic cocaine abusers. Am J Drug Alcohol Abuse 18:131–144
Ornstein TJ, Iddon JL, Baldacchino AM, Sahakian BJ, London M, Everitt BJ, Robbins TW (2000) Profiles of cognitive dysfunction in chronic amphetamine and heroin abusers. Neuropsychopharmacology 23:113–126
Parsons LH, Smith AD, Justice JB (1991) Basal extracellular dopamine is decreased in the rat nucleus accumbens during abstinence from chronic cocaine. Synapse 9:60–65
Petry NM, Bickel WK, Arnett M (1998) Shortened time horizons and insensitivity to future consequences in heroin addicts. Addiction 93:729–738
Pettit HO, Ettenberg A, Bloom FE, Koob GF (1984) Destruction of dopamine in the nucleus accumbens selectively attenuates cocaine but not heroin self-administration in rats. Psychopharmacology 84:167–173
Pothos E, Rada P, Mark GP, Hoebel BG (1991) Dopamine microdialysis in the nucleus accumbens during acute and chronic morphine, naloxone-precipitated withdrawal and clonidine treatment. Brain Res 566:348–350
Robbins TW (2002) The 5-choice serial reaction time task: behavioral pharmacology and functional neurochemistry. Psychopharmacology 163:363–380
Rogers RD, Robbins TW (2001) Investigating the neurocognitive deficits associated with chronic drug misuse. Curr Opin Neurobiol 11:250–257
Rogers RD, Everitt BJ, Baldacchino A, Blackshaw AJ, Swainson R, Wynne K, Baker NM, Hunter J, Carthy T, Booker E, London M, Deakin JFW, Sahakian BJ, Robbins TW (1999) Dissociable deficits in the decision-making cognition of chronic amphetamine abusers, opiate abusers, patients with frontal damage to prefrontal cortex, and tryptophan-depleted normal volunteers: evidence for monoaminergic mechanisms. Neuropsychopharmacology 20:322–339
Rosselli M, Ardila A (1996) Cognitive effects of cocaine and polydrug abuse. J Clin Exp Neuropsychol 18:122–135
Rossetti ZL, Hmaidan Y, Gessa GL (1992) Marked inhibition of mesolimbic dopamine release: a common feature of ethanol, morphine, cocaine and amphetamine abstinence in rats. Eur J Pharmacol 221:227–234
Shoaib M, Bizarro L (2005) Deficits in a sustained attention task following nicotine withdrawal in rats. Psychopharmacology 178:211–222
Sim T, Simon SL, Domier CP, Richardson K, Rawson RA, Ling W (2002) Cognitive deficits among methamphetamine users with attention deficit hyperactivity disorder symptomatology. J Addict Dis 21:75–89
Volkow ND, Fowler JS, Wang G-J, Hitzemann R, Logan J, Schlyer DJ, Dewey SL, Wolf AP (1993) Decreased dopamine D2 receptor availability is associated with reduced frontal metabolisms in cocaine abusers. Synapse 14:169–177
Zacny JP (1995) A review of the effects of opioids on psychomotor and cognitive functioning in humans. Exp Clin Psychopharmacol 3:432–466
Acknowledgements
This study was supported by the Wellcome Trust within the Cambridge Medical Research Council Centre for Behavioural and Clinical Neuroscience.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dalley, J.W., Lääne, K., Pena, Y. et al. Attentional and motivational deficits in rats withdrawn from intravenous self-administration of cocaine or heroin. Psychopharmacology 182, 579–587 (2005). https://doi.org/10.1007/s00213-005-0107-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-005-0107-3