Abstract
Rationale
Although nicotine dependence and tolerance develop in rats, few studies have examined these processes in the mouse. Establishing such mouse models would eventually allow for an examination of the role of specific nicotinic receptor subtypes in mediating these processes (i.e. through the use of receptor knockouts).
Objectives
The goals of the present study were to establish mouse models of nicotine dependence and tolerance.
Methods
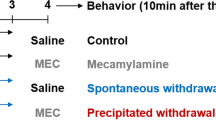
Mice were chronically exposed to nicotine (0–200 μg/ml) in their drinking solution and assayed for plasma nicotine and cotinine levels, withdrawal signs following nicotine cessation (spontaneous withdrawal) or nicotinic antagonist administration (precipitated withdrawal), or nicotine tolerance. Dependence assays included somatic sign observations (paw tremors, backing and head shakes), tail-flick, plantar stimulation, elevated plus-maze and spontaneous activity. Tolerance was assayed using tail-flick, hot-plate and body temperature tests.
Results
Plasma nicotine and cotinine levels were elevated during oral nicotine exposure (15.85 ng/ml and 538.00 ng/ml, respectively) and quickly declined following nicotine cessation (<1 ng/ml and <2 ng/ml, respectively), providing evidence that the oral route was pharmacologically relevant. Nicotine withdrawal increased numbers of somatic signs (spontaneous and mecamylamine-precipitated withdrawal) and/or hyperalgesia (spontaneous withdrawal only). Chronic nicotine exposure also produced tolerance, as indicated by reduced responsivity to acute nicotine in assays of analgesia and hypothermia.
Conclusions
These results indicate that chronic oral nicotine produces dependence and tolerance in the mouse. Further, nicotine dependence may be mediated by multiple nicotinic receptor subtypes, since specific nicotinic receptor antagonists failed to precipitate withdrawal.




Similar content being viewed by others
References
Atwell L, Jacobson AE (1978) The search for less harmful analgesics. Lab Anim 7:42–47
Carstens E, Anderson KA, Simons CT, Carstens MI, Jinks S (2001) Analgesia induced by chronic nicotine infusion in rats: differences by gender and pain test. Psychopharmacology, 157:40–405
D’Amour FE, Smith DL (1941) A method for determining loss of pain sensation. J Pharmacol Exp Ther 107:385–939
Damaj MI, Martin BR (1996) Tolerance to the antinociceptive effect of epibatidine after acute and chronic administration in mice. Eur J Pharmacol 300:51–57
Damaj MI, Welch SP, Martin BR (1995) In vivo pharmacological effects of dihydro-beta-erythroidine, a nicotinic antagonist, in mice. Psychopharmacology 117:67–73
Damaj MI, Welch SP, Martin BR (1996) Characterization and modulation of acute tolerance to nicotine in mice. J Pharmacol Exp Ther 277:454–461
Damaj MI, Kao W, Martin BR (2003) Characterization of spontaneous and precipitated nicotine withdrawal in the mouse. J Pharmacol Exp Ther 307:526–534
Dewey WL, Harris LS, Howes JS, Nuite JA (1970) The effect of various neurohormonal modulations on the activity of morphine and the narcotic antagonists in tail-flick and phenylquinone test. J Pharmacol Exp Ther 175:435–442
Eddy NB, Leimbach D (1953) Synthetic analgesics II: dithienybutenyl- and benzomorphans. J Pharmacol Exp Ther 107:385–939
Epping-Jordan MP, Watkins SS, Koob GF, Markou A (1998) Dramatic decreases in brain reward function during nicotine withdrawal. Nature 393:76–79
Gäddnäs H, Pietilä K, Ahtee L (2000) Effects of chronic oral nicotine treatment and its withdrawal on locomotor activity and brain monoamines in mice. Behav Brain Res 113:65–72
Hariharan M, VanNoord T, Greden JF (1988) A high-performance liquid chromatographic method for routine simultaneous determination of nicotine and cotinine in plasma. Clin Chem 34:724–729
Hildebrand BE, Panagis G, Svensson TH, Nomikos GG (1999) Behavioral and biochemical manifestations of mecamylamine-precipitated nicotine withdrawal in the rat: role of nicotinic receptors in the ventral tegmental area. Neuropsychopharmacology 21:560–574
Isola R, Vogelsberg V, Wemlinger TA, Neff NH, Hadjiconstantinou M (1999) Nicotine abstinence in the mouse. Brain Res 850:189–196
Malin DH (2001) Nicotine dependence: studies with a laboratory model. Pharmacol Biochem Behav 70:551–559
Malin DH, Lake JR, Newlin-Maultsby P, Roberts LK, Lanier JG, Carter VA, Cunningham JS, Wilson OB (1992) Rodent model of nicotine abstinence syndrome. Pharmacol Biochem Behav 43:779–784
Malin DH, Lake JR, Carter VA, Cunningham JS, Hebert KM, Conrad DL, Wilson OB (1994) The nicotinic antagonist mecamylamine precipitates nicotine abstinence syndrome in the rat. Psychopharmacology 115:180–184
Malin DH, Lake JR, Schopen CK, Kirk JW, Sailer EE, Lawless BA, Upchurch TP, Shenoi M, Rajan N (1997) Nicotine abstinence syndrome precipitated by central but not peripheral hexamethonium. Pharmacol Biochem Behav 58:695–699
Malin DH, Lake JR, Upchurch TP, Shenoi M, Rajan N, Schweinle WE (1998) Nicotine abstinence syndrome precipitated by the competitive antagonist dihydro-β-erythroidine. Pharmacol Biochem Behav 60:609–613
Markou A, Paterson NE (2001) The nicotinic antagonist methyllycaconitine has differential effects on nicotine self-administration and nicotine withdrawal in the rat. Nicotine Tob Res 3:361–373
Marks MJ, Stitzel JA, Collins AC (1985) Time course study of the effects of chronic nicotine infusion on drug response and brain receptors. J Pharmacol Exp Ther 235:619–628
Mattila MJ, Ahtee L, Saarnivaara L (1968) The analgesic and sedative effects of nicotine in white mice, rabbits and golden hamsters. Ann Med Exp Fenn 46:78–84
McCallum SE, Caggiula AR, Epstein LH, Saylor S, Ploskina T, Sved AF (1999) Mecamylamine blocks the development of tolerance to nicotine in rats: implications for the mechanisms of tolerance. Psychopharmacology 141:332–338
Messina ES, Tyndale RF, Sellers EM (1997) A major role for CYP2A6 in nicotine C-oxidation by human liver microsomes. J Pharmacol Exp Ther 282:1608–1614
Okamoto M, Kita T, Okuda H, Nakashima T (1992) Tolerance to the convulsions induced by daily nicotine treatment in rats. Jpn J Pharmacol 594:449–455
Pietilä K, Ahtee L (2000) Chronic nicotine administration in the drinking water affects the striatal dopamine in mice. Pharmacol Biochem Behav 66:95–103
Pietilä K, Laakso I, Ahtee L (1995) Chronic oral nicotine administration affects the circadian rhythm of dopamine and 5-hydroxytryptamine metabolism in the striata of mice. Naunyn Schmiedeberg’s Arch Pharmacol 353:110–115
Pietilä K, Lähde T, Attila M, Ahtee L, Nordberg A (1998) Regulation of nicotinic receptors in the brain of mice withdrawn from chronic oral nicotine treatment. Naunyn Schmiedeberg’s Arch Pharmacol 357:176–182
Robinson SF, Grun EU, Pauly JR, Collins AC (1996) Changes in sensitivity to nicotine and brain nicotinic receptors following chronic nicotine and corticosterone treatments in mice. Pharmacol Biochem Behav 54:587–593
Salminen O, Ahtee L (2000) The effects of acute nicotine on the body temperature and striatal dopamine metabolism of mice during chronic nicotine infusion. Neurosci Lett 284:37–40
Sparks JA, Pauly JR (1999) Effects of continuous oral nicotine administration on brain nicotinic receptors and responsiveness to nicotine in C57Bl/6 mice. Psychopharmacology 141:145–153
Szyndler J, Sienkiewicz-Jarosz H, Maciejak P, Siemiatkowski M, Rokicki D, Czlonkowska A, Plaznik A (2001) The anxiolytic-like effect of nicotine undergoes rapid tolerance in a model of contextual fear conditioning in rats. Pharmacol Biochem Behav 69:511–518
Yang CY, Wu WH, Zbuzek VK (1992) Antinociceptive effect of chronic nicotine and nociceptive effect of its withdrawal measured by hot-plate and tail-flick in rats. Psychopharmacology 106:417–420
Zarrindast M-R, Khoshayand M-R, Shafaghi B (1999) The development of cross-tolerance between morphine and nicotine in mice. Eur Neuropsychopharmacol 9:227–233
Acknowledgements
The authors thank Tie Shan-Han and Alicia Stevans for their technical support on this project. Research was supported by NIDA DA-05274, NIH DA-07027 and CIHR MOP 14173. Research complied with the Guide for the Care and Use of Laboratory Animals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grabus, S.D., Martin, B.R., Batman, A.M. et al. Nicotine physical dependence and tolerance in the mouse following chronic oral administration. Psychopharmacology 178, 183–192 (2005). https://doi.org/10.1007/s00213-004-2007-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-004-2007-3