Abstract
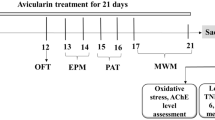
Alzheimer’s disease (AD) is the leading neurodegenerative disorder with extracellular senile plaques and neurofibrillary tangles as the major hallmarks. The objective was to evaluate the effect of phloretin in a chronic model of sporadic AD by injecting aggregated form of Aβ25-35 peptide sequence intracerebroventricularly (icv) in Wistar rats. To achieve this, male Wistar rats were injected with aggregated Aβ25-35 peptide icv, followed by 21 days phloretin (2.5 mg/kg, 5 mg/kg) administration after recovery period. Barnes maze and elevated plus maze along with the biochemical estimation of antioxidant enzymes activities were conducted. The hippocampus region of the rat brains were stained with Congo red and Nissl stain. TNF-α was estimated in the brain homogenates using the ELISA assay. In this study, phloretin improved the spatial memory formation and retention in Barnes maze test. Additionally, phloretin alleviated the antioxidant defense biomarkers and thereby reduced oxidative stress, decreased TNF-α-mediated neuroinflammation. Furthermore, phloretin treatment showed decreased amyloid beta accumulation in the CA1 region and less number of pyknotic nuclei in the dentate gyrus of the Aβ25-35-injected rat brains. The above experimental findings evinced the promising role of phloretin in Aβ25-35-injected rats and which further envisage its potential to be explored in the treatment of AD.










Similar content being viewed by others
Abbreviations
- 4-HNE:
-
4-Hydroxynonenal
- AD:
-
Alzheimer’s disease
- Aβ:
-
Amyloid beta
- BACE:
-
Beta site APP cleaving enzyme 1
- CAT:
-
Catalase
- DON:
-
Donepezil
- ELT:
-
Escape latency time
- EPM:
-
Elevated plus maze
- ETS:
-
Electron transport system
- GSH:
-
Reduced glutathione
- GSK3 β:
-
Glycogen synthase kinase 3β
- icv:
-
Intracerebroventricular
- MDA:
-
Malondialdehyde
- PBS:
-
Phosphate buffer saline
- PHL:
-
Phloretin
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- TNFα:
-
Tumor necrosis factor alpha
References
Armstrong RA, Myers D, Smith CU, Cairns N, Luthert PJ (1991) Alzheimer's disease: the relationship between the density of senile plaques, neurofibrillary tangles and A4 protein in human patients. Neurosci Lett 123(2):141–143
Bancroft JD, Gamble M (2002) Theory and practice of histological techniques. Churchill Livingstone, Edeinburgh, London, New York
Barnes CA (1979) Memory deficits associated with senescence: a neurophysiological and behavioral study in the rat. J Comp Physiol Psychol 93:74–104
Barreca D, Currò M, Bellocco E, Ficarra S, Laganà G, Tellone E, Laura Giunta M, Visalli G, Caccamo D, Galtieri A, Ientile R (2017) Neuroprotective effects of phloretin and its glycosylated derivative on rotenone-induced toxicity in human SH-SY5Y neuronal-like cells. BioFactors 43(4):549–557. https://doi.org/10.1002/biof.1358
Bhullar KS, Rupa singhe HP (2013) Polyphenols: multipotent therapeutic agents in neurodegenerative diseases. Oxidative Med Cell Longev 891748. https://doi.org/10.1155/2013/891748
Boonen RA, van Tijn P, Zivkovic D (2009) Wnt signaling in Alzheimer’s disease: up or down, that is the question. Ageing Res Rev 8(2):71–82. https://doi.org/10.1016/j.arr.2008.11.003
Cetin F, Dincer S (2007) The effect of intrahippocampal beta amyloid (1-42) peptide injection on oxidant and antioxidant status in rat brain. Ann N Y Acad Sci 1100(1):510–517. https://doi.org/10.1196/annals.1395.056
Chang WT, Huang WC, Liou CJ (2012) Evaluation of the anti-inflammatory effects of phloretin and phlorizin in lipopolysaccharide-stimulated mouse macrophages. Food Chem 134(2):972–979. https://doi.org/10.1016/j.foodchem.2012.03.002
Cohen RM, Rezai-Zadeh K, Weitz TM, Rentsendorj A, Gate D, Spivak I, Bholat Y, Vasilevko V, Glabe CG, Breunig JJ, Rakic P (2013) A transgenic Alzheimer rat with plaques, tau pathology, behavioral impairment, oligomeric aβ, and frank neuronal loss. J Neurosci 33(15):6245–6256. https://doi.org/10.1523/JNEUROSCI.3672-12.2013
Delobette S, Privat A, Maurice T (1997) In vitro aggregation facilitates β-amyloid peptide-(25–35)-induced amnesia in the rat. Eur J Pharmacol 319(1):1–4
File SE, Gonzalez LE (1996) Anxiolytic effects in the plus-maze of 5-HT 1A-receptor ligands in dorsal raphe and ventral hippocampus. Pharmacol Biochem Behav 54(1):123–128
Fong CW (2015) Permeability of the blood-brain barrier: molecular mechanism of transport of drugs and physiologically important compounds. J Membr Biol 248(4):651–669. https://doi.org/10.1007/s00232-015-9778-9
Ghumatkar PJ, Patil SP, Jain PD, Tambe RM, Sathaye S (2015) Nootropic, neuroprotective and neurotrophic effects of phloretin in scopolamine induced amnesia in mice. Pharmacol Biochem Behav 135:182–191. https://doi.org/10.1016/j.pbb.2015.06.005
Ghumatkar P, Peshattiwar V, Patil S, Muke S, Whitfield D, Howlett D, Francis P, Sathaye S (2018) The effect of phloretin on synaptic proteins and adult hippocampal neurogenesis in Aβ (1-42) injected male Wistar rats. J Pharm Pharmacol 70(8):1022–1030. https://doi.org/10.1111/jphp.12925
Harkany T, Mulder J, Sasvari M, Abraham I, Konya C, Zarandi M, Penke B, Luiten PGM, Nyakas C (1999) N-methyl-D-aspartate receptor antagonist MK-801 and radical scavengers protect cholinergic nucleus basalis neurons against β-amyloid neurotoxicity. Neurobiol Dis 6(2):109–121. https://doi.org/10.1006/nbdi.1998.0230
Jung M, Triebel S, Anke T, Richling E, Erkel G (2009) Influence of apple polyphenols on inflammatory gene expression. Mol Nutr Food Res 53(10):1263–1280. https://doi.org/10.1002/mnfr.200800575
Kawahara M, Kuroda Y (2001) Intracellular calcium changes in neuronal cells induced by Alzheimer’s ß-amyloid protein are blocked by estradiol and cholesterol. Cell Mol Neurobiol 21:1–13
Kim HC, Yamada K, Nitta A, Olariu A, Tran MH, Mizuno M, Nakajima A, Nagai T, Kamei H, Jhoo WK, Im DH (2003) Immunocytochemical evidence that amyloid β (1–42) impairs endogenous antioxidant systems in vivo. Neuroscience 119(2):399–419
Klementiev B, Novikova T, Novitskaya V, Walmod PS, Dmytriyeva O, Pakkenberg B, Berezin V, Bock E (2007) A neural cell adhesion molecule–derived peptide reduces neuropathological signs and cognitive impairment induced by Aβ 25-35. Neuroscience 145(1):209–224. https://doi.org/10.1016/j.neuroscience.2006.11.060
Kobayashi Y (2010) The regulatory role of nitric oxide in proinflammatory cytokine expression during the induction and resolution of inflammation. J Leukoc Biol 88(6):1157–1162. https://doi.org/10.1189/jlb.0310149
Liu Y, Zhang L, Liang J (2015) Activation of the Nrf2 defense pathway contributes to neuroprotective effects of phloretin on oxidative stress injury after cerebral ischemia/reperfusion in rats. J Neurol Sci 351(1):88–92. https://doi.org/10.1016/j.jns.2015.02.045.
LoPachin RM, Gavin T, Geohagen BC, Zhang L, Casper D, Lekhraj R, Barber DS (2011) β-Dicarbonyl enolates: a new class of neuroprotectants. J Neurochem 116(1):132–143. https://doi.org/10.1111/j.1471-4159.2010.07091.
Lu P, Mamiya T, Lu LL, Mouri A, Zou LB, Nagai T, Hiramatsu M, Ikejima T, Nabeshima T (2009) Silibinin prevents amyloid β peptide-induced memory impairment and oxidative stress in mice. Br J Pharmacol 157(7):1270–1277
Mattson MP (2004) Pathways towards and away from Alzheimer’s disease. Nature 430(7000):631–639. https://doi.org/10.1038/nature02621
Maurice T, Lockhart BP, Privat A (1996) Amnesia induced in mice by centrally administered β-amyloid peptides involves cholinergic dysfunction. Brain Res 706(2):181–193
Miguel-Hidalgo JJ, Cacabelos R (1998) β-Amyloid (1–40)-induced neurodegeneration in the rat hippocampal neurons of the CA1 subfield. Acta Neuropathol 95(5):455–465
Nandi A, Chatterjee IB (1988) Assay of superoxide dismutase activity in animal tissues. J Biosci 13(3):305–315
Olariu A, Tran MH, Yamada K, Mizuno M, Hefco V, Nabeshima T (2001) Memory deficits and increased emotionality induced by β-amyloid (25–35) are correlated with the reduced acetylcholine release and altered phorbol dibutyrate binding in the hippocampus. J Neural Transm 108(8–9):1065–1079. https://doi.org/10.1007/s007020170025
Ondrejcak T, Klyubin I, Hu NW, Barry AE, Cullen WK, Rowan MJ (2010) Alzheimer’s disease amyloid β-protein and synaptic function. NeuroMolecular Med 12(1):13–26. https://doi.org/10.1007/s12017-009-8091-0
Pike CJ, Walencewicz-Wasserman AJ, Kosmoski J, Cribbs DH, Glabe CG, Cotman CW (1995) Structure-activity analyses of β-amyloid peptides: contributions of the β25–35 region to aggregation and neurotoxicity. J Neurochem 64(1):253–265
Pocernich CB, Cardin AL, Racine CL, Lauderback CM, Butterfield DA (2001) Glutathione elevation and its protective role in acrolein-induced protein damage in synaptosomal membranes: relevance to brain lipid peroxidation in neurodegenerative disease. Neurochem Int 39(2):141–149
Rath PC, Aggarwal BB (1999) TNF-induced signaling in apoptosis. J Clin Immunol 19(6):350–364
Reilly CA, Aust SD (2001) Measurement of lipid peroxidation. Current protocols in toxicology 2–4
Schmitt U, Hiemke C (1998) Strain differences in open-field and elevated plus-maze behavior of rats without and with pretest handling. Pharmacol Biochem Behav 59(4):807–811
Selkoe DJ (2001) Alzheimer’s disease: genes, proteins, and therapy. Physiol Rev 81(2):741–766
Sinha AK (1972) Colorimetric assay of catalase. Anal Biochem 47(2):389–394
Smith IK, Vierheller TL, Thorne CA (1988) Assay of glutathione reductase in crude tissue homogenates using 5, 5′-dithiobis (2-nitrobenzoic acid). Anal Biochem 175(2):408–413
Solanki I, Parihar P, Mansuri ML, Parihar MS (2015) Flavonoid-based therapies in the early management of neurodegenerative diseases. Adv Nutr 6(1):64–72. https://doi.org/10.3945/an.114.007500
Spencer JP (2007) The interactions of flavonoids within neuronal signalling pathways. Genes Nutr 2(3):257–273. https://doi.org/10.1007/s12263-007-0056-z
Stangl V, Lorenz M, Ludwig A, Grimbo N, Guether C, Sanad W, Ziemer S, Martus P, Baumann G, Stangl K (2005) The flavonoid phloretin suppresses stimulated expression of endothelial adhesion molecules and reduces activation of human platelets. J Nutr 135(2):172–178. https://doi.org/10.1093/jn/135.2.172
Stepanichev MY, Moiseeva YV, Lazareva NA, Onufriev MV, Gulyaeva NV (2003) Single intracerebroventricular administration of amyloid-beta (25–35) peptide induces impairment in short-term rather than long-term memory in rats. Brain Res Bull 61(2):197–205
Stepanichev MY, Zdobnova IM, Zarubenko II, Moiseeva YV, Lazareva NA, Onufriev MV, Gulyaeva NV (2004) Amyloid-β (25–35)-induced memory impairments correlate with cell loss in rat hippocampus. Physiol Behav 80(5):647–655. https://doi.org/10.1016/j.physbeh.2003.11.003
Stepanichev M, Zdobnova I, Zarubenko I, Lazareva N, Gulyaeva NV (2006) Differential effects of tumor necrosis factor-alpha co-administered with amyloid beta-peptide (25–35) on memory function and hippocampal damage in rat. Behav Brain Res 175(2):352–361. https://doi.org/10.1016/j.bbr.2006.09.006
Tamagno E, Parola M, Bardini P, Piccini A, Borghi R, Guglielmotto M, Santoro G, Davit A, Danni O, Smith MA, Perry G (2005) β-Site APP cleaving enzyme up-regulation induced by 4-hydroxynonenal is mediated by stress-activated protein kinases pathways. J Neurochem 92(3):628–636. https://doi.org/10.1111/j.1471-4159.2004.02895.x.
Varadarajan S, Kanski J, Aksenova M, Lauderback C, Butterfield DA (2001) Different mechanisms of oxidative stress and neurotoxicity for Alzheimer’s Aβ (1− 42) and Aβ (25− 35). J Am Chem Soc 123(24):5625–5631
Villard V, Espallergues J, Keller E, Alkam T, Nitta A, Yamada K, Nabeshima T, Vamvakides A, Maurice T (2009) Antiamnesic and neuroprotective effects of the aminotetrahydrofuran derivative ANAVEX1-41 against amyloid β25–35-induced toxicity in mice. Neuropsychopharmacology 34(6):1552–1566. https://doi.org/10.1038/npp.2008.212
Vytal KE, Cornwell BR, Arkin NE, Letkiewicz AM, Grillon C (2013) The complex interaction between anxiety and cognition: insight from spatial and verbal working memory. Front Hum Neurosci 7:93. https://doi.org/10.3389/fnhum.2013.00093
Walf AA, Frye CA (2007) The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat Protoc 2(2):322–328. https://doi.org/10.1038/nprot.2007.44
Watson C (2004) The rat brain in stereotaxic coordinates-the new coronal set. Academic, Cambridge
Yamaguchi Y, Kawashima S (2001) Effects of amyloid-β-(25–35) on passive avoidance, radial-arm maze learning and choline acetyltransferase activity in the rat. Eur J Pharmacol 412(3):265–272
Zhu Q, Zhang NQ, Lau CF, Chao J, Sun Z, Chang RC, Chen F, Wang M (2012) In vitro attenuation of acrolein-induced toxicity by phloretin, a phenolic compound from apple. Food Chem 135(3):1762–1768. https://doi.org/10.1016/j.foodchem.2012.06.053
Acknowledgements
The authors would like to thank Mr. Pankaj Jain, Mrs. Rufi Tambe, Mr. Gauresh Somani, Mr. Madhav Seervi, Mr. Devang Shah, Ms. Divya Kanchan, Ms. Akruti Kaikini, Ms. Sneha Bagle from ICT.
Funding
This work was supported by the INSPIRE-Department of Science and Technology (DST), New Delhi, India.
Author information
Authors and Affiliations
Contributions
PG and SS conceived and designed the present study. SP and VP assisted in framing the protocols and conducting the experiments. TV, VD, and GV assisted in conducting the histopathology of the brain tissues. PG wrote the manuscript which was later corrected by SS. All authors read and approved the manuscript.
Corresponding author
Rights and permissions
About this article
Cite this article
Ghumatkar, P.J., Patil, S.P., Peshattiwar, V. et al. The modulatory role of phloretin in Aβ25–35 induced sporadic Alzheimer’s disease in rat model. Naunyn-Schmiedeberg's Arch Pharmacol 392, 327–339 (2019). https://doi.org/10.1007/s00210-018-1588-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-018-1588-z