Abstract
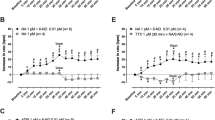
Phosphodiesterases PDE2, PDE3, and PDE4 are expressed in murine sinoatrial cells. PDE3 and/or PDE4 reduce heart rate but apparently do not influence the tachycardia mediated through sinoatrial β1- and β2-adrenoceptors despite the high content of sinoatrial cAMP. The function of PDE2 is, however, uncertain. Prostaglandin PGE1 elicits sinoatrial tachycardia through EP receptors, but the control by phosphodiesterases is unknown. We investigated on spontaneously beating right atria of mice the effects of the PDE2 inhibitors Bay 60-7550 and EHNA on basal beating and the tachycardia produced by noradrenaline (3 nM) and PGE1 (1 μM). Bay 60-7550 (1 μM), but not EHNA (10 μM), increased basal sinoatrial beating. EHNA also failed to produce tachycardia in the presence of the adenosine deaminase inhibitor 2′-deoxycoformycin (10 μM), remaining inconclusive whether PDE2 reduces basal sinoatrial beating. Rolipram (10 μM) and cilostamide (300 nM) caused moderate tachycardia. The tachycardia evoked by Bay 60-7550 was similar in the absence and presence of rolipram. Noradrenaline elicited stable tachycardia that was not increased by Bay 60-7550. A stable tachycardia caused by PGE1 was not increased by the inhibitors of PDE2, PDE3, and PDE4. Unlike PDE3 and PDE4 which reduce murine basal sinoatrial beating, a possible effect of PDE2 needs further research. The stable tachycardia produced by noradrenaline and PGE1, together with the lack potentiation by the inhibitors of PDE2, PDE3, and PDE4, suggests that cAMP generated at the receptor compartments is hardly hydrolyzed by these phophodiesterases. Evidence from human volunteers is consistent with this proposal.





Similar content being viewed by others
References
Agarwal SR, Macdougall DA, Tyser R, Pugh SD, Callaghan SD, Harvey RD (2011) Effects of cholesterol depletion on compartmentalized cAMP responses in adult cardiac myocytes. J Mol Cell Cardiol 50:500–509. doi:10.1016/j.yjmcc.2010.11.015
Boess FG, Hendrix M, Van der Staay F-J, Erb C, Schreiber R, Van Staveren W, De Vente J, Prockaerts J, Blokland A, Koenig G (2004) Inhibition of phosphodiesterase 2 increases neuronal cGMP synaptic plasticity and memory performance. Neuropharmacology 47:1081–1092. doi:10.1016/j.neuropharm.2004.07.040
Carson DA, Seegmiller JE (1976) Effect of adenosine deaminase inhibition upon human lymphocyte blastogenesis. J Clin Invest 57:274–282. doi:10.1172/JCI108278
Christ T, Galindo-Tovar A, Thoms M, Ravens U, Kaumann AJ (2009) Inotropy and L-type Ca2+ current, activated by beta1- and beta2-adrenoceptors, are differently controlled by phosphodiesterases 3 and 4 in rat heart. Br J Pharmacol 156(1):62–83. doi:10.1111/j.1476-5381.2008.00015.x
Cusack BJ, Nielson CP, Morgan ME, Vestal RE (1987) Additive effect of theophylline on the cardiac response to isoproterenol. Clin Pharmacol Ther 41:289–296. doi:10.1038/clpt.1987.30
Di Benedetto G, Zoccanato A, Lissandron V, Terrin A, Li X, Houslay MD, Baillie G, Zaccolo M (2008) Protein kinase type I and type II define distinct intracellular signalling compartments. Circ Res 103:836–844. doi:10.1161/CIRCRESAHA.108.174813
Fredholm BB, Ijzerman AP, Jacobson KA, Klotz KN, Linden J (2001) International Union of Pharmacology. XXV. Nomenclature and classification of adenosine receptors. Pharmacol Rev 53:527–552
Galindo-Tovar A, Kaumann AJ (2008) Phosphodiesterase-4 blunts inotropism and arrhythmias but not sinoatrial tachycardia of (-)-adrenaline mediated through mouse cardiac β1-adrenoceptors. Br J Pharmacol 153(4):710–20. doi:10.1038/sj.bjp.0707631
Galindo-Tovar A, Vargas ML, Escudero E, Kaumann AJ (2009) Ontogenic changes of the control by phosphodiesterases-3 and -4 of 5-HT responses in porcine heart and relevance to human atrial 5-HT4 receptors. Br J Pharmacol 156:237–249. doi:10.1111/j.1476-5381.2008.00023.x
Hua R, Adamcyk A, Robbins C, Ray G, Rose RA (2012) Distinct patterns of constistutive phosphodiesterase activity in mouse sinoatrial node and atrial myocardium. PLoS 7:e47652. doi:10.1371/journal.pone.0047652
Kaumann AJ (1986) The β1-adrenoceptor antagonist CGP 20712A unmasks β2-adrenoceptors activated by (-)-adrenaline in rat sinoatrial node. Naunyn-Schmiedeberg´s Arch Pharmacol 332:406–409
Kaumann AJ (2011) Phosphodiesterases reduce spontaneous sinoatrial beating but do not the ´´fight or flight´´ tachycardia elicited by agonists through Gs protein-coupled receptors. Trends Pharmacol Sci 32:277–283. doi:10.1016/j.tips.2011.03.003
Kaumann AJ, Birnbaumer L (1974) Prostaglandin E1 action on sinus pacemaker and adenylyl cyclase in kitten myocardium. Nature 251:515–517
Kaumann AJ, Galindo-Tovar A, Escudero E, Vargas ML (2009) Phosphodiesterases do not limit β1-adrenoceptor-mediated sinoatrial tachycardia: evidence with PDE3 and PDE4 in rabbits and PDE1-5 in rats. Naunyn-Schmiedeberg´s Arch Pharmacol 380:421–430. doi:10.1007/s00210-009-0445-5
Lakatta EG, DiFrancesco D (2009) What keeps us ticking: a funny current, a calcium clock, or both. J Mol Cell Cardiol 47:157–170. doi:10.1016/j.yjmcc.2009.03.022
Mehel H, Emons J, Vettel C, Wittköpper K, Seppelt D, Dewenter M et al (2013) Phosphodiesterase-2 is up-regulated in human failing hearts and blunts β-adrenergic responses in cardiomyocytes. JACC 62:1596–1606. doi:10.1016/j.jacc.2013.05.057
Mongillo M, Tocchetti CG, Terrin A, Lissandron V, Cheung Y-F, Dorstmann WR, Pozzan T et al (2006) Compartmentalized phosphodiesterase-2 activity blunts β-adrenergic cardiac inotropy via an NO/cGMP-dependent pathway. Circ Res 98:226–234. doi:10.1161/01.RES.0000200178.34179.93
Podzuweit T, Nennstiel P, Müller A (1995) Isoenzyme-selective inhibition of cGMP-stimulated cyclic nucleotide phosphodiesterase by EHNA (erythro-9-(2-hydroxy-3-nonyl) adenine). Cell Sign 7:733–738. doi:10.1016/0898-6568(95)00042-N
Rivet-Bastide M, Vandecasteele G, Hatem S, Verde I, Bénardeau A, Mercadier JJ, Fischmeister R (1997) cGMP-stimulated cyclic nucleotide phosphodiesterase regulates the basal calcium current in human atrial myocytes. J Clin Invest 99:2710–2718. doi:10.1172/JCI119460
Samet MK, Rutledge CO (1985) Antagonism of the positive chronotropic effect of norepinephrine by purine nucleosides in rat atria. J Pharmacol Exp Ther 232:106–110
Steinberg SF, Brunton LL (2001) Compartmentation of G protein-coupled signaling pathways in cardiac myocytes. Annu Rev Pharmacol Toxicol 41:751–773. doi:10.1146/annurev.pharmtox.41.1.751
Vandecasteele G, Verde I, Rucker-Martin C, Donzeau-Gouge P, Fischmeister R (2001) Cyclic GMP regulation of the L-type Ca2+ channel current in human atrial myocytes. J Physiol 533:329–340. doi:10.1111/j.1469-7793.2001.0329a.x
Vinogradova TM, Sirenko S, Lyashkov AE, Younes A, Li Y, Zhu W et al (2008) Constitutive phosphodiesterase activity restricts spontaneous beating rate of cardiac pacemaker cells by suppressing local Ca2+ releases. Circ Res 102:761–769. doi:10.1161/CIRCRESAHA.107.161679
Warrier S, Ramamurthy G, Eckert RL, Nikolaev VO, Lohse MJ, Harvey RD (2007) cAMP microdomains and L-type Ca2+ channel regulation in guinea-pig ventricular myocytes. J Physiol 580:765–776. doi:10.1113/jphysiol.2006.124891
Woodward DF, Jones RL, Narumiya S (2011) International Union of Basic and Clinical Pharmacology. LXXXIII: Classification of Prostanoid Receptors, Updating 15 Years of Progress. Pharmacol Rev 63:471–538. doi:10.1124/pr.110.003517
Wu AY, Tang X-B, Martinez SE, Ikeda K, Beavo JA (2004) Molecular determinants for cyclic nucleotide binding to the regulatory domains of phosphodiesterase 2A. J Biol Chem 279:37928–37938. doi:10.1074/jbc.M404287200
Acknowledgments
We are grateful to Dr. A. Peñuela for help with Fig. 5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Galindo-Tovar, A., Vargas, M.L. & Kaumann, A.J. Inhibitors of phosphodiesterases PDE2, PDE3, and PDE4 do not increase the sinoatrial tachycardia of noradrenaline and prostaglandin PGE1 in mice. Naunyn-Schmiedeberg's Arch Pharmacol 389, 177–186 (2016). https://doi.org/10.1007/s00210-015-1178-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-015-1178-2