Abstract
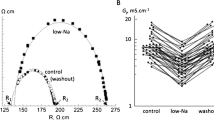
Co-ordinated electrical activation of the heart is maintained by intercellular coupling of cardiomyocytes via gap junctional channels located in the intercalated disks. These channels consist of two hexameric hemichannels, docked to each other, provided by either of the adjacent cells. Thus, a complete gap junction channel is made from 12 protein subunits, the connexins. While 21 isoforms of connexins are presently known, cardiomyocytes typically are coupled by Cx43 (most abundant), Cx40 or Cx45. Some years ago, antiarrhythmic peptides were discovered and synthesised, which were shown to increase macroscopic gap junction conductance (electrical coupling) and enhance dye transfer (metabolic coupling). The lead substance of these peptides is AAP10 (H-Gly-Ala-Gly-Hyp-Pro-Tyr-CONH2), a peptide with a horseshoe-like spatial structure as became evident from two-dimensional nuclear magnetic resonance studies. A stable d-amino-acid derivative of AAP10, rotigaptide, as well as a non-peptide analogue, gap-134, has been developed in recent years. Antiarrhythmic peptides act on Cx43 and Cx45 gap junctions but not on Cx40 channels. AAP10 has been shown to enhance intercellular communication in rat, rabbit and human cardiomyocytes. Antiarrhythmic peptides are effective against ventricular tachyarrhythmias, such as late ischaemic (type IB) ventricular fibrillation, CaCl2 or aconitine-induced arrhythmia. Interestingly, the effect of antiarrhythmic peptides is higher in partially uncoupled cells and was shown to be related to maintained Cx43 phosphorylation, while arrhythmogenic conditions like ischaemia result in Cx43 dephosphorylation and intercellular decoupling. It is still a matter of debate whether these drugs also act against atrial fibrillation. The present review outlines the development of this group of peptides and derivatives, their mode of action and molecular mechanisms, and discusses their possible therapeutic potential.






Similar content being viewed by others
References
Aonuma S, Kohama Y, Akai K, Komiyama Y, Nakajima S, Wakabayashi M et al (1980) Studies on heart. XIX. Isolation of an atrial peptide that improves the rhythmicity of cultured myocardial cell clusters. Chem Pharm Bull 28:3332–3339
Axelsen LN, Stahlhut M, Mohammed S, Larsen BD, Nielsen MS, Holstein-Rathlou NH, Andersen S, Jensen ON, Hennan JK, Kjølbye AL (2006) Identification of ischemia-regulated phosphorylation sites in connexin43: a possible target for the antiarrhythmic peptide analogue rotigaptide (ZP123). J Mol Cell Cardiol 40:790–798
Bedner P, Niessen H, Odermatt B, Willecke K, Harz H (2003) A method to determine the relative cAMP permeability of connexin channels. Exp Cell Res 291:25–35
Boldt A, Wetzel U, Lauschke J, Weigl J, Gummert J, Hindricks G, Kottkamp H, Dhein S (2004) Fibrosis in left atrial tissue of patients with atrial fibrillation with and without underlying mitral valve disease. Heart 90:400–405
Buchanan JW, Saito T, Gettes LS (1985) The effect of antiarrhythmic drugs, stimulation frequency and potassium induced resting membrane potential changes on conduction velocity and dV/dtmax in guinea pig myocardium. Circ Res 56:696–703
Butera JA, Larsen BD, Hennan JK, Kerns E, Di L, Alimardanov A, Swillo RE, Morgan GA, Liu K, Wang Q, Rossman EI, Unwalla R, McDonald L, Huselton C, Petersen JS (2009) Discovery of (2S, 4R)-1-(2-aminoacetyl)-4-benzamido-pyrrolidine-2-carboxylic acid hydrochloride (GAP-134)13, an orally active small molecule gap-junction modifier for the treatment of atrial fibrillation. J Med Chem 52:908–911
Christ GJ, Brink PR (1999) Analysis of the presence and physiological relevance of subconducting states of connxin43-derived gap junction channels in cultured human corporal vascular smooth muscle cells. Circ Res 85:797–803
Clarke TC, Thomas D, Petersen JS, Evans WH, Martin PE (2006) The antiarrhythmic peptide rotigaptide (ZP123) increases gap junction intercellular communication in cardiac myocytes and HeLa cells expressing connexin 43. Br J Pharmacol 147:486–495
Clarke TC, Williams OJ, Martin PE, Evans WH (2009) ATP release by cardiac myocytes in a simulated ischaemia model: inhibition by a connexin mimetic and enhancement by an antiarrhythmic peptide. Eur J Pharmacol 605:9–14
Cruciani V, Mikalsen SO (2007) Evolutionary selection pressure and family relationships among connexin genes. Biol Chem 388:253–264
Darrow BJ, Laing JG, Lampe PD, Saffitz JE, Beyer EC (1995) Expression of multiple connexins in cultured neonatal rat ventricular myocytes. Circ Res 76:381–387
DaTorre SD, Creer MH, Pogwizd SM, Corr PB (1991) Amphipathic lipid metabolites and their relation to arrhythmogenesis in the ischemic heart. J Mol Cell Cardiol 23(suppl.1):11–22
De Groot JR, Wilms-Schopman FJ, Opthof T, Remme CA, Coronel R (2001) Late ventricular arrhythmias during acute regional ischemia in the isolated blood perfused pig heart. Role of electrical cellular coupling. Cardiovasc Res 50:362–372
De Groot JR, Schumacher CA, Verkerk AO, Baartscheer A, Fiolet JWT, Coronel R (2003) Intrinsic heterogeneity in repolarization is increased in isolated failing rabbit cardiomyocytes during simulated ischemia. Cardiovasc Res 59:705–714
Dekker LRC, Fiolet JWT, VanBavel E, Coronel R, Opthof T, Spaan JAE, Janse MJ (1996) Intracellular Ca2+, intercellular electrical coupling and mechanical activity in ischemic rabbit papillary muscle. Effects of preconditioning and metabolic blockade. Circ Res 79:237–246
Delmar M, Michaels DC, Johnson T, Jalife J (1987) Effects of increasing intercellular resistance on transverse and longitudinal propagation in sheep epicardial muscle. Circ Res 60:780–785
Dhein S (2004) Pharmacology of gap junctions in the cardiovascular system. Cardiovasc Res 62:287–298
Dhein S (2006) Role of connexins in atrial fibrillation. Adv Cardiol 42:161–174
Dhein S, Tudyka T (1995) The therapeutic potential of antiarrhythmic peptides. Cellular coupling as a new antiarrhythmic target. Drugs 49:851–855
Dhein S, Manicone N, Müller A, Gerwin R, Ziskoven U, Irankhahi A, Minke C, Klaus W (1994) A new synthetic antiarrhythmic peptide reduces dispersion of epicardial activation recovery interval and enhances cellular communication. Antiarrhythmic properties in regional ischemia. Naunyn Schmiedebergs Arch Pharmacol 350:174–184
Dhein S, Weng S, Grover R, Tudyka T, Gottwald M, Schaefer T, Polontchouk L (2001) Protein kinase Cα mediates the effect of antiarrhythmic peptide on gap junction conductance. Cell Adhes Commun 8:257–264
Dhein S, Larsen BD, Petersen JS, Mohr FW (2003) Effects of the new antiarrhythmic peptide ZP123 on epicardial activation and repolarization pattern. Cell Commun Adhes 10:371–378
Doble BW, Ping P, Kardami E (2000) The ε subtype of protein kinase C is required for cardiomyocyte connexin-43 phosphorylation. Circ Res 86:293–301
Easton JA, Petersen JS, Martin PE (2009) The anti-arrhythmic peptide AAP10 remodels Cx43 and Cx40 expression and function. Naunyn Schmiedebergs Arch Pharmacol 80(1):11–24
Echt DS, Liebson PR, Mitchell LB et al (1991) Mortality and morbidity in patients receiving encainide, flecainide or placebo. The Cardiac Arrhythmia Suppression Trial. New Engl J Med 324:781–788
Eloff BC, Gilat E, Wan X, Rosenbaum DS (2003) Pharmacological modulation of cardiac gap junctions to enhance cardiac conduction: evidence supporting a novel target for antiarrhythmic therapy. Circulation 108:3157–3163
Falk MM (2000) Connexin-specific distribution within gap junctions revealed in living cells. J Cell Sci 113:4109–4120
Fallon RF, Goodenough DA (1981) Five hour half-life of mouse liver gap junction protein. J Cell Biol 127:343–355
Giepmans BNG (2004) Gap junctions and connexion interacting proteins. Cardiovasc Res 62:233–245
Gottwald M (1998) Verbesserung der zellulären Kopplung auf der Ebene der gap junction Kanäle alsm neuer antiarrhythmischer Therapieansatz bei Herzrhythmusstörungen. Thesis, Biology, University of Cologne.
Grover R, Dhein S (1998) Spatial structure determination of antiarrhythmic peptide using nuclear magnetic resonance spectroscopy. Peptides 19:1725–1729
Grover R, Dhein S (2001) Structure–activity relationships of novel peptides related to the antiarrhythmic peptide AAP10 which reduce the dispersion of epicardial action potential duration. Peptides 22:1011–1021
Guerra JM, Everett TH 4th, Lee KW, Wilson E, Olgin JE (2006) Effects of the gap junction modifier rotigaptide (ZP123) on atrial conduction and vulnerability to atrial fibrillation. Circulation 114:110–118
Hagen A, Dietze A, Dhein S (2009) Human cardiac gap junction coupling: effects of antiarrhythmic peptide AAP10. Cardiovascular Research 83:405–415
Haugan K, Lam HR, Knudsen CB, Petersen JS (2004) Atrial fibrillation in rats induced by rapid transesophageal atrial pacing during brief episodes of asphyxia: a new in vivo model. J Cardiovasc Pharmacol 44:125–135
Haugan K, Olsen KB, Hartvig L, Petersen JS, Holstein-Rathlou NH, Hennan JK, Nielsen MS (2005) The antiarrhythmic peptide analog ZP123 prevents atrial conduction slowing during metabolic stress. J Cardiovasc Electrophysiol 16:537–545
Haugan K, Miyamoto T, Takeishi Y, Kubota I, Nakayama J, Shimojo H, Hirose M (2006a) Rotigaptide (ZP123) improves atrial conduction slowing in chronic volume overload-induced dilated atria. Basic Clin Pharmacol Toxicol 99:71–79
Haugan K, Marcussen N, Kjølbye AL, Nielsen MS, Hennan JK, Petersen JS (2006b) Treatment with the gap junction modifier rotigaptide (ZP123) reduces infarct size in rats with chronic myocardial infarction. J Cardiovasc Pharmacol 47:236–242
Hennan JK, Swillo RE, Morgan GA, Keith JC Jr, Schaub RG, Smith RP, Feldman HS, Haugan K, Kantrowitz J, Wang PJ, Abu-Qare A, Butera J, Larsen BD, Crandall DL (2006) Rotigaptide (ZP123) prevents spontaneous ventricular arrhythmias and reduces infarct size during myocardial ischemia/reperfusion injury in open-chest dogs. J Pharmacol Exp Ther 317:236–243
Janse MJ, van Capelle FJL (1982) Electrotonic interactions across an inexcitable region as a cause of ectopic activity in acute regional myocardial ischemia. Circ Res 50:527–537
Janse MJ, Wit AL (1989) Electrophysiological mechanisms of ventricular arrhythmias resulting from myocardial ischemia and infarction. Physiol Rev 69:1049–1169
Janse MJ, van Capelle FJL, Morsink H, Kléber AG, Wilms-Schopman F, Cardinal R, Naumann d’Alnoncourt C, Durrer D (1980) Flow of "injury current" and patterns of excitation during early ventricular arrhythmias in acute myocardial ischemia in isolated porcine and canine hearts. Circ Res 47:151–165
Jongsma HJ, Wilders R (2000) Gap junctions in cardiovascular disease. Circ Res 86:1193–1197
Jørgensen NR, Teilmann SC, Henriksen Z, Meier E, Hansen SS, Jensen JE, Sørensen OH, Petersen JS (2005) The antiarrhythmic peptide analog rotigaptide (ZP123) stimulates gap junction intercellular communication in human osteoblasts and prevents decrease in femoral trabecular bone strength in ovariectomized rats. Endocrinology 146:4745–4754
Jozwiak J, Dhein S (2008) Local effects and mechanisms of antiarrhythmic peptide AAP10 in acute regional myocardial ischemia: electrophysiological and molecular findings. Naunyn Schmiedebergs Arch Pharmacol 378:459–470
Kawara T, Derksen R, de Groot JR, Coronel R, Tasseron S, Linnenbank AC, Hauer RN, Kirkels H, Janse MJ, de Bakker JM (2001) Activation delay after premature stimulation in chronically diseased human myocardium relates to the architecture of interstitial fibrosis. Circulation 104:3069–3075
Kjølbye AL, Knudsen CB, Jepsen T, Larsen BD, Petersen JS (2003) Pharmacological characterization of the new stable antiarrhythmic peptide analog Ac-D-Tyr-D-Pro-D-Hyp-Gly-D-Ala-Gly-NH2 (ZP123): in vivo and in vitro studies. J Pharmacol Exp Ther 306:1191–1199
Kohama Y, Okimoto N, Mimura T, Fukaya C, Watanabe M, Yokoyama K (1987) A new antiarrhythmic peptide, N-3-(4-hydroxyphenyl)propionyl-Pro-Hyp-Gly-Ala-Gly. Chem Pharm Bull 35:3928–3930
Kostin S, Klein G, Szalay Z, Hein S, Bauer EP, Schaper J (2002) Structural correlate of atrial fibrillation in human patients. Cardiovasc Res 54:361–379
Kreuzberg MM, Liebermann M, Segschneider S, Dobrowolski R, Dobrzynski H, Kaba R, Rowlinson G, Dupont E, Severs NJ, Willecke K (2009) Human connexin31.9, unlike its orthologous protein connexin30.2 in the mouse, is not detectable in the human cardiac conduction system. J Mol Cell Cardiol 46:553–559
Kumar NM, Gilula NB (1996) The gap junction communication channel. Cell 84:381–388
Kundu B, Rizvi SY, Mathur KB et al (1990) Antiarrhythmic activity of a novel analogue of AAP. Collect Czech Chem Commun 55:575–580
Kwak BR, Jongsma HJ (1996) Regulation of cardiac gap junction channel permeability and conductance by several phosphorylating conditions. Mol Cell Biochem 157:93–99
Kwak BR, Van Veen TAB, Analbers LJS, Jongsma HJ (1995) TPA increases conductance but decreases permeability in neonatal rat cardiomyocyte gap junction channels. Exp Cell Res 220:456–463
Laird DW (1996) The life cycle of a connexin: gap junction formation removal and degradation. J Bioenerg Biomembr 28:311–317
Lampe PD, Lau AF (2000) Regulation of gap junctions by phosphorylation of connexins. Arch Biochem Biophys 384:205–215
Lerner DL, Yamada KA, Schuessler RB, Saffitz JE (2000) Accelerated onset and increased incidence of ventricular arrhythmias induced by ischemia in Cx43-deficient mice. Circulation 101:547–552
Lesh MD, Pring M, Spear JF (1989) Cellular uncoupling can unmask dispersion of action potential duration in ventricular myocardium. Circ Res 65:1426–1440
Li DQ, Feng YB, Zhang HQ (2004) The relationship between gap junctional remodeling and atrial fibrillation in patients with rheumatic heart disease. Zhonghua Yi Xue Za Zhi 84:384–386
Lin X, Zemlin C, Hennan JK, Petersen JS, Veenstra RD (2008) Enhancement of ventricular gap–junction coupling by rotigaptide. Cardiovasc Res 79:416–426
Morley GE, Vaidya D, Samie FH, Lo C, Delmar M, Jalife J (1999) Characterization of conduction in the ventricles of normal and heterozygous Cx43 knockout mice using optical mapping. J Cardiovasc Electrophysiol 10:1361–1375
Müller A, Dhein S (1993) Sodium channel blockade enhances dispersion of the cardiac action potential duration. A computer simulation study. Basic Res Cardiol 88:11–15
Müller A, Gottwald M, Tudyka T, Linke W, Klaus W, Dhein S (1997a) Increase in gap junction conductance by an antiarrhythmic peptide. Eur J Pharmacol 327:65–72
Müller A, Schaefer T, Linke W, Tudyka T, Gottwald M, Klaus W, Dhein S (1997b) Actions of the antiarrhythmic peptide AAP10 on cellular coupling. Naunyn Schmiedebergs Arch Pharmacol 356:76–82
Müller A, Lauven M, Berkels R, Dhein S, Polder HR, Klaus W (1999) Switched single electrode voltage clamp amplifiers allow precise measurement of gap junction conductance. Am J Physiol 276:C980–C987
Musil LS, Goodenough DA (1993) Multisubunit assembly of an integral plasma membrane channel protein, gap junction connexin43, occurs after exit from the ER. Cell 74:1065–1077
Musil LS, Goodenough DA (1995) Biochemical analysis of connexon assembly. In: Kanno Y, Kataoka K, Shiba Y, Shibata Y, Shimazu T (eds) Intercellular communication through gap junctions. Progress in cell research vol. 4. Elsevier, Amsterdam, pp 327–330
Naccarelli GV, Wolbrette DL, Dell’Orfano JT, Patel HM, Luck JC (2000) Amiodarone: what have we learned from clinical trials? Clin Cardiol 23:73–82
Naccarelli GV, Wolbrette DL, Samii S, Banchs JE, Penny-Peterson E, Gonzalez MD (2007) New antiarrhythmic treatment of atrial fibrillation. Expert Rev Cardiovasc Ther 5:707–714
Neef M, Frenzel C, Mühlberg K, Dhein S, Pfeiffer D, Salameh A (2004) Growth factors and antiarrhythmic peptide AAP10 regulate connexin43 expression. Eur J Physiol 447(Suppl. 1):S131
Polontchouk L, Haefliger J-A, Ebelt B, Schaefer T, Stuhlmann D, Mehlhorn U, Kuhn-Reignier F, DeVivie ER, Dhein S (2001) Effects of chronic atrial fibrillation on gap junction distribution in human and rat atria. J Am Coll Cardiol 38:883–891
Quan XQ, Bai R, Liu N, Chen BD, Zhang CT (2007) Increasing gap junction coupling reduces transmural dispersion of repolarization and prevents torsade de pointes in rabbit LQT3 model. J Cardiovasc Electrophysiol 8:1184–1189
Ren Y, Zhang CT, Wu J, Ruan YF, Pu J, He L, Wu W, Chen BD, Wang WG, Wang L (2006) The effects of antiarrhythmic peptide AAP10 on ventricular arrhythmias in rabbits with healed myocardial infarction. Zhonghua Xin Xue Guan Bing Za Zhi 34:825–828
Rohr S (2004) Role of gap junctions in the propagation of the cardiac action potential. Cardiovasc Res 62:309–322
Rohr S, Kucera JP, Fast VG, Kleber AG (1997) Paradoxical improvement of impulse conduction in cardiac tissue by partial cellular uncoupling. Science 275(5301):841–844
Rossman EI, Liu K, Morgan GA, Swillo RE, Krueger JA, Gardell SJ, Butera J, Gruver M, Kantrowitz J, Feldman HS, Petersen JS, Haugan K, Hennan JK (2009) The gap junction modifier, GAP-134 [(2S, 4R)-1-(2-aminoacetyl)-4-benzamido-pyrrolidine-2-carboxylic acid], improves conduction and reduces atrial fibrillation/flutter in the canine sterile pericarditis model. J Pharmacol Exp Ther 329:1127–1133
Salameh A, Dhein S (2005) Pharmacology of gap junctions. New pharmacological targets for treatment of arrhythmia, seizure and cancer? Biochim Biophys Acta 1719(1–2):36–58
Severs NJ, Coppen SR, Dupont E, Yeh H-I, Ko Y-S, Matsushita T (2004) Gap junction alterations in human cardiac disease. Cardiovasc Res 62:368–377
Shaw RM, Rudy Y (1997a) Electrophysiologic effects of acute myocardial ischemia. A mechanistic investigation of action potential conduction and conduction failure. Circ Res 80:124–138
Shaw RM, Rudy Y (1997b) Ionic mechanisms of propagation in cardiac tissue: roles of the sodium and L-type calcium currents during reduced excitability and decreased gap–junction coupling. Circ Res 81:727–741
Shaw RM, Fay AJ, Puthenveedu MA, von Zastrow M, Jan YN, Jan LY (2007) Microtubule plus-end-tracking proteins target gap junctions directly from the cell interior to adherens junctions. Cell 128(3):547–560
Shiroshita-Takeshita A, Sakabe M, Haugan K, Hennan JK, Nattel S (2007) Model-dependent effects of the gap junction conduction-enhancing antiarrhythmic peptide rotigaptide (ZP123) on experimental atrial fibrillation in dogs. Circulation 115:310–318
Söhl G, Willecke K (2004) Gap junctions and the connexion protein family. Cardiovasc Res 62:228–232
Spach MS, Dolber PC (1990) Discontinuous anisotropic propagation. In: Rosen MR, Janse MJ, Wit AL (eds) Cardiac electrophysiology: a textbook. Futura, Mount Kisco, pp 517–534
Spach MS, Josephson ME (1994) Initiating reentry: the role of non-uniform anisotropy in small circuits. J Cardiovasc Electrophysiol 5:182–209
Spach MS, Dolber PC, Anderson AW (1989a) Multiple regional differences in cellular properties that regulate repolarization and contraction in the right atrium of adult and newborn dogs. Circ Res 65:1594–1611
Spach MS, Dolber PC, Heidlage JF (1989b) Interaction of inhomogeneities of repolarization with anisotropic propagation in dog atria. A mechanism for both preventing and initiating reentry. Circ Res 65:1612–1631
Spach MS, Heidlage JF, Dolber PC, Barr RC (2000) Electrophysiological effects of remodeling cardiac gap junctions and cell size: experimental and model studies of normal cardiac growth. Circ Res 86:302–311
Stahlhut M, Petersen JS, Hennan JK, Ramirez MT (2006) The antiarrhythmic peptide rotigaptide (ZP123) increases connexin 43 protein expression in neonatal rat ventricular cardiomyocytes. Cell Commun Adhes 13:21–27
Thomas SP, Kucera JP, Bircher-Lehmann L, Rudy Y, Saffitz JE, Kleber AG (2003) Impulse propagation in synthetic strands of neonatal cardiac myocytes with genetically reduced levels of connexin43. Circ Res 92:1209–1216
Unger VM, Kumar NM, Gilula NB, Yeager M (1999) Three-dimensional structure of a recombinant gap junction membrane channel. Science 283(5405):1176–1180
Van der Velden HMW, van Kempen MJA, Wijffels MCEF, van Zijverden M, Groenewegen WA, Allessie MA, Jongsma HJ (1998) Altered pattern of connexin40 distribution in persistent atrial fibrillation in the goat. J Cardiovasc Electrophysiol 9:596–607
Van der Velden HMW, Ausma J, Rook MB, Hellemous AJCGM, Van Veen TAAB, Allessie MA, Jongsma HJ (2000) Gap junctional remodeling in relation to stabilization of atrial fibrillation in the goat. Cardiovasc Res 46:476–486
Verrecchia F, Duthe F, Duval S, Duchatelle I, Sarrouilhe D, Hervé JC (1999) ATP counteracts the rundown of gap junctional channels of rat ventricular myocytes by promoting protein phosphorylation. J Physiol (Lond) 516:447–459
Waldo AL, Camm AJ, Deruyter H et al (1996) Effect of d-sotalol on mortality in patients with left ventricular dysfunction after recent and remote myocardial infarction. The SWORD Investigators. Lancet 348:7–12
Wang R, Zhang C, Ruan Y, Liu N, Wang L (2007) Changes in phosphorylation of connexin43 in rats during acute myocardial hypoxia and effects of antiarrhythmic peptide on the phosphorylation. J Huazhong Univ Sci Technolog Med Sci 27:241–244
Weingart R, Maurer P (1988) Action potential transfer in cell pairs isolated from adult rat and guinea pig ventricles. Circ Res 63:72–80
Weng S, Lauven M, Schaefer T, Polontchouk L, Grover R, Dhein S (2002) Pharmacological modulation of gap junction coupling by an antiarrhythmic peptide via protein kinase C activation. FASEB J 16:1114–1116
Wit AL, Rosen MR (1983) Pathophysiologic mechanisms of cardiac arrhythmias. Am Heart J 106(4 Pt 2):798–811
Wit AL, Duffy HS (2008) Drug development for treatment of cardiac arrhythmias: targeting the gap junctions. Am J Physiol Heart Circ Physiol 294:H16–H18
Wu J, McHowat J, Saffitz JE, Yamada KA, Corr PB (1993) Inhibition of gap junctional conductance by long chain acylcarnitines and their preferential accumulation in junctional sarcolemma during hypoxia. Circ Res 72:879–889
Xing D, Kjølbye AL, Nielsen MS, Petersen JS, Harlow KW, Holstein-Rathlou NH, Martins JB (2003) ZP123 increases gap junctional conductance and prevents reentrant ventricular tachycardia during myocardial ischemia in open chest dogs. J Cardiovasc Electrophysiol 14:510–520
Xing D, Kjølbye AL, Petersen JS, Martins JB (2005) Pharmacological stimulation of cardiac gap junction coupling does not affect ischemia-induced focal ventricular tachycardia or triggered activity in dogs. Am J Physiol Heart Circ Physiol 288:H511–H516
Yan H, Chen JZ, Zhu JH, Ni YM, Yu GW, Hu SJ, Tao QM (2004) Expression of connexin in atrium of patients with atrial fibrillation and its signal transduction pathway. Zhonghua Yi Xue Za Zhi 84:209–213
Acknowledgements
This study has been supported by the Deutsche Forschungsgemeinschaft (grant to S.D.) and by the Studienstiftung des Deutschen Volkes (grant to A.H.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dhein, S., Hagen, A., Jozwiak, J. et al. Improving cardiac gap junction communication as a new antiarrhythmic mechanism: the action of antiarrhythmic peptides. Naunyn-Schmied Arch Pharmacol 381, 221–234 (2010). https://doi.org/10.1007/s00210-009-0473-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-009-0473-1