Abstract
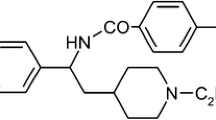
Inward rectifier potassium currents IK1 and acetylcholine activated IK,ACh are implicated in atrial fibrillation (AF) pathophysiology. In chronic AF (cAF), IK,ACh develops a receptor-independent, constitutively active component that together with increased IK1 is considered to support maintenance of AF. Here, we tested whether class I (propafenone, flecainide) and class III (dofetilide, AVE0118) antiarrhythmic drugs inhibit atrial IK1 and IK,ACh in patients with and without cAF. IK1 and IK,ACh were measured with voltage clamp technique in atrial myocytes from 58 sinus rhythm (SR) and 35 cAF patients. The M-receptor agonist carbachol (CCh; 2 µM) was employed to activate IK,ACh. In SR, basal current was not affected by either drug indicating no effect of these compounds on IK1. In contrast, all tested drugs inhibited CCh-activated IK,ACh in a concentration-dependent manner. In cAF, basal current was confirmed to be larger than in SR (at −80 mV, −15.2 ± 1.2 pA/pF, n = 88/35 vs. −6.5 ± 0.4 pA/pF, n = 194/58 [myocytes/patients]; P < 0.05), whereas CCh-activated IK,ACh was smaller (−4.1 ± 0.5 pA/pF vs. −9.5 ± 0.6 pA/pF; P < 0.05). In cAF, receptor-independent constitutive IK,ACh contributes to increased basal current, which was reduced by flecainide and AVE0118 only. This may be due to inhibition of constitutively active IK,ACh channels. In cAF, all tested drugs reduced CCh-activated IK,ACh. We conclude that in cAF, flecainide and AVE0118 reduce receptor-independent, constitutively active IK,ACh, suggesting that they may block IK,ACh channels, whereas propafenone and dofetilide likely inhibit M-receptors. The efficacy of flecainide to terminate AF may in part result from blockade of IK,ACh.





Similar content being viewed by others
References
Barry PH (1994) JPCalc, a software package for calculating liquid junction potential corrections in patch-clamp, intracellular, epithelial and bilayer measurements and for correcting junction potential measurements. J Neurosci Methods 51:107–116
Bashir Y, Thomsen PE, Kingma JH, Moller M, Wong C, Cobbe SM, Jordaens L, Campbell RW, Rasmussen HS, Camm AJ (1995) Electrophysiologic profile and efficacy of intravenous dofetilide (UK-68, 798), a new class III antiarrhythmic drug, in patients with sustained monomorphic ventricular tachycardia. Dofetilide Arrhythmia Study Group. Am J Cardiol 76:1040–1044
Birkhead JS, Vaughan Williams EM (1977) Dual effect of disopyramide on atrial and atrioventricular conduction and refractory periods. Br Heart J 39:657–660
Blaauw Y, Schotten U, van Hunnik A, Neuberger HR, Allessie MA (2007) Cardioversion of persistent atrial fibrillation by a combination of atrial specific and non-specific class III drugs in the goat. Cardiovasc Res 75:89–98
Bosch RF, Zeng X, Grammer JB, Popovic K, Mewis C, Kuhlkamp V (1999) Ionic mechanisms of electrical remodeling in human atrial fibrillation. Cardiovasc Res 44:121–131
Brandts B, Van Bracht M, Dirkmann D, Borchard R, Wickenbrock I, Prull MW, Meine M, Trappe HJ (2004) Perspectives and limitations in the treatment of vagus-induced atrial fibrillation. Insights from cellular pharmacology. Med Klin (Munich) 99:341–346
Cha TJ, Ehrlich JR, Chartier D, Qi XY, Xiao L, Nattel S (2006) Kir3-based inward rectifier potassium current: potential role in atrial tachycardia remodeling effects on atrial repolarization and arrhythmias. Circulation 113:1730–1737
Christ T, Wettwer E, Voigt N, Hala O, Radicke S, Matschke K, Varro A, Dobrev D, Ravens U (2008) Pathology-specific effects of the IKur/Ito/IK,ACh blocker AVE0118 on ion channels in human chronic atrial fibrillation. Br J Pharmacol 154:1619–1630
Dobrev D (2008) New concepts in understanding and modulating atrial repolarisation in patients with atrial fibrillation. J Interv Card Electrophysiol 22:107–110
Dobrev D, Friedrich A, Voigt N, Jost N, Wettwer E, Christ T, Knaut M, Ravens U (2005) The G protein-gated potassium current IK,ACh is constitutively active in patients with chronic atrial fibrillation. Circulation 112:3697–3706
Dobrev D, Graf E, Wettwer E, Himmel HM, Hala O, Doerfel C, Christ T, Schuler S, Ravens U (2001) Molecular basis of downregulation of G-protein-coupled inward rectifying K+ current (IK,ACh) in chronic human atrial fibrillation: decrease in GIRK4 mRNA correlates with reduced IK,ACh and muscarinic receptor-mediated shortening of action potentials. Circulation 104:2551–2557
Dobrev D, Ravens U (2003) Remodeling of cardiomyocyte ion channels in human atrial fibrillation. Basic Res Cardiol 98:137–148
Dobrev D, Wettwer E, Himmel HM, Kortner A, Kuhlisch E, Schuler S, Siffert W, Ravens U (2000) G-Protein β3-subunit 825T allele is associated with enhanced human atrial inward rectifier potassium currents. Circulation 102:692–697
Dobrev D, Wettwer E, Kortner A, Knaut M, Schuler S, Ravens U (2002) Human inward rectifier potassium channels in chronic and postoperative atrial fibrillation. Cardiovasc Res 54:397–404
Dobrzynski H, Marples DD, Musa H, Yamanushi TT, Henderson Z, Takagishi Y, Honjo H, Kodama I, Boyett MR (2001) Distribution of the muscarinic K+ channel proteins Kir3.1 and Kir3.4 in the ventricle, atrium, and sinoatrial node of heart. J Histochem Cytochem 49:1221–1234
Duan D, Fermini B, Nattel S (1993) Potassium channel blocking properties of propafenone in rabbit atrial myocytes. J Pharmacol Exp Ther 264:1113–1123
Ehrlich JR (2008) Inward rectifier potassium currents as a target for atrial fibrillation therapy. J Cardiovasc Pharmacol 52:129–135
Ehrlich JR, Cha TJ, Zhang L, Chartier D, Villeneuve L, Hebert TE, Nattel S (2004) Characterization of a hyperpolarization-activated time-dependent potassium current in canine cardiomyocytes from pulmonary vein myocardial sleeves and left atrium. J Physiol 557:583–597
Follmer CH, Colatsky TJ (1990) Block of delayed rectifier potassium current, IK, by flecainide and E-4031 in cat ventricular myocytes. Circulation 82:289–293
Fuster V, Ryden LE, Cannom DS, Crijns HJ, Curtis AB, Ellenbogen KA, Halperin JL, Le Heuzey JY, Kay GN, Lowe JE, Olsson SB, Prystowsky EN, Tamargo JL, Wann S, Smith SC Jr, Jacobs AK, Adams CD, Anderson JL, Antman EM, Hunt SA, Nishimura R, Ornato JP, Page RL, Riegel B, Priori SG, Blanc JJ, Budaj A, Camm AJ, Dean V, Deckers JW, Despres C, Dickstein K, Lekakis J, McGregor K, Metra M, Morais J, Osterspey A, Zamorano JL (2006) ACC/AHA/ESC 2006 guidelines for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines (writing committee to revise the 2001 guidelines for the management of patients with atrial fibrillation): developed in collaboration with the European Heart Rhythm Association and the Heart Rhythm Society. Circulation 114:e257–e354
Gogelein H, Brendel J, Steinmeyer K, Strubing C, Picard N, Rampe D, Kopp K, Busch AE, Bleich M (2004) Effects of the atrial antiarrhythmic drug AVE0118 on cardiac ion channels. Naunyn Schmiedebergs Arch Pharmacol 370:183–192
Guillemare E, Marion A, Nisato D, Gautier P (2000) Inhibitory effects of dronedarone on muscarinic K+ current in guinea pig atrial cells. J Cardiovasc Pharmacol 36:802–805
Hohnloser SH, Crijns HJ, van Eickels M, Gaudin C, Page RL, Torp-Pedersen C, Connolly SJ (2009) Effect of dronedarone on cardiovascular events in atrial fibrillation. N Engl J Med 360:668–678
Inomata N, Ishihara T, Akaike N (1991) Mechanisms of the anticholinergic effect of SUN 1165 in comparison with flecainide, disopyramide and quinidine in single atrial myocytes isolated from guinea-pig. Br J Pharmacol 104:1007–1011
Inomata N, Ohno T, Ishihara T, Akaike N (1993) Antiarrhythmic agents act differently on the activation phase of the ACh-response in guinea-pig atrial myocytes. Br J Pharmacol 108:111–115
Kiehn J, Villena P, Beyer T, Brachmann J (1994) Differential effects of the new class III agent dofetilide on potassium currents in guinea pig cardiomyocytes. J Cardiovasc Pharmacol 24:566–572
Miyasaka Y, Barnes ME, Gersh BJ, Cha SS, Bailey KR, Abhayaratna WP, Seward JB, Tsang TS (2006) Secular trends in incidence of atrial fibrillation in Olmsted County, Minnesota, 1980 to 2000, and implications on the projections for future prevalence. Circulation 114:119–125
Nattel S (2002) New ideas about atrial fibrillation 50 years on. Nature 415:219–226
Nattel S, Burstein B, Dobrev D (2008) Atrial remodelling and atrial fibrillation: mechanisms and implications. Circ Arrhythmia Electrophysiol 1:62–73
Pandit SV, Berenfeld O, Anumonwo JM, Zaritski RM, Kneller J, Nattel S, Jalife J (2005) Ionic determinants of functional reentry in a 2-D model of human atrial cells during simulated chronic atrial fibrillation. Biophys J 88:3806–3821
Piovan D, Padrini R, Furlanut M, Moretto R, Ferrari M (1986) Plasma and tissue levels of flecainide in rats. Pharmacol Res Commun 18:739–745
Roden DM (2005) Antiarrhythmic drugs. In: Brunton LL, Goodman LS, Gilman A, Blumenthal D, Parker KL, Buxton I (eds) The Goodman & Gilman manual of pharmacology and therapeutics. McGraw-Hill, New York, pp 899–932
Schneider HJ, Husser O, Rihm M, Fredersdorf S, Birner C, Dhein S, Muders F, Jeron A, Goegelein H, Riegger GA, Luchner A (2009) Safety of the novel atrial-selective K+-channel blocker AVE0118 in experimental heart failure. Naunyn Schmiedebergs Arch Pharmacol 379:225–232
Steurer G, Weber H, Schmidinger H, Plass H, Frey B, Purerfellner H, Probst P (1991) Plasma propafenone concentration in the evaluation of anti-arrhythmic efficacy. Eur Heart J 12:526–532
Van Wagoner DR, Pond AL, McCarthy PM, Trimmer JS, Nerbonne JM (1997) Outward K+ current densities and Kv1.5 expression are reduced in chronic human atrial fibrillation. Circ Res 80:772–781
Voigt N, Friedrich A, Bock M, Wettwer E, Christ T, Knaut M, Strasser RH, Ravens U, Dobrev D (2007) Differential phosphorylation-dependent regulation of constitutively active and muscarinic receptor-activated IK,ACh channels in patients with chronic atrial fibrillation. Cardiovasc Res 74:426–437
Voigt N, Maguy A, Yeh YH, Qi X, Ravens U, Dobrev D, Nattel S (2008) Changes in IK,ACh single-channel activity with atrial tachycardia remodelling in canine atrial cardiomyocytes. Cardiovasc Res 77:35–43
Wirth KJ, Paehler T, Rosenstein B, Knobloch K, Maier T, Frenzel J, Brendel J, Busch AE, Bleich M (2003) Atrial effects of the novel K+-channel-blocker AVE0118 in anesthetized pigs. Cardiovasc Res 60:298–306
Workman AJ, Kane KA, Rankin AC (2001) The contribution of ionic currents to changes in refractoriness of human atrial myocytes associated with chronic atrial fibrillation. Cardiovasc Res 52:226–235
Acknowledgments
The authors thank Trautlinde Thurm, Ulrike Heinrich and Annegret Häntzschel for excellent technical assistance.
Sources of funding
These studies were supported by the Deutsche Forschungsgemeinschaft (Do 769/1-1-3), the German Federal Ministry of Education and Research through the Atrial Fibrillation Competence Network (01Gi0204), the European Union (“NORMACOR”, grant LSHM-CT-2006-018676), and a grant of Fondation Leducq (07 CVD 03).
Disclosures
Ursula Ravens is a consultant to MEDA Pharma GmbH & Co. KG, Bad Homburg (flecainide).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Voigt, N., Rozmaritsa, N., Trausch, A. et al. Inhibition of IK,ACh current may contribute to clinical efficacy of class I and class III antiarrhythmic drugs in patients with atrial fibrillation. Naunyn-Schmied Arch Pharmacol 381, 251–259 (2010). https://doi.org/10.1007/s00210-009-0452-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-009-0452-6