Abstract.
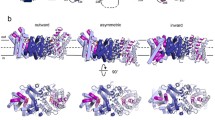
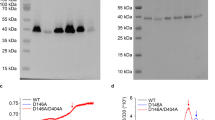
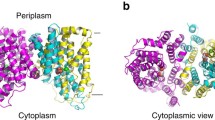
The Na+-dependent citrate carrier of Klebsiella pneumoniae (CitS) is a member of the 2-hydroxycarboxylate transporter family. Within the highly conserved helix Vb region, Asn-185 of CitS was mutated to Val and Glu-194 was mutated to Gln. The wild-type and mutant proteins were synthesised in Escherichia coli DH5α or C43(DE3) as biotinylated or His-tagged CitS-fusions, respectively. The synthesis and purification procedure yielded 6.5 mg pure CitS per litre culture. The fusion proteins were characterised with E. coli cell suspensions or after reconstitution of the purified enzymes into proteoliposomes. The E194Q mutation had almost no effect on the kinetics of Na+ or citrate transport. In contrast, aberrant citrate transport kinetics were found for the N185V mutant. The apparent K m value for the citrate species H-citrate2- was increased about nine-fold, whereas the apparent V max value and the effect of Na+ on the transport kinetics were comparable to the wild-type. Asn-185 of CitS appears therefore to participate in the binding of H-citrate2–.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Kästner, C., Dimroth, P. & Pos, K. The Na+-dependent citrate carrier of Klebsiella pneumoniae: high-level expression and site-directed mutagenesis of asparagine-185 and glutamate-194. Arch Microbiol 174, 67–73 (2000). https://doi.org/10.1007/s002030000175
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002030000175