Abstract
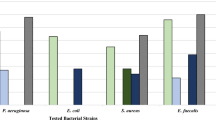
During the investigation, soil actinomycetes were isolated from Kathlekanu swamp forest and the crude ethyl acetate extract from the potent isolate KF15 was analysed with GC–MS and HPTLC to identify bioactive metabolites. The crude extract was examined for in-vitro antifungal activity on pathogens of chilli; MTT cytotoxicity assay was performed against HeLa cell line to determine the anticancer potential. The isolate Streptomyces sp. strain KF15 exhibited antagonistic activity against fungal pathogens by inhibiting growth and altering growth pattern with increased antimicrobial activity in dose-dependent manner. GC–MS revealed many bioactive compounds and HPTLC depicted metabolite fingerprint. The IC50 of 99.85 µg/ml indicated the high potential of KF15 extract to prevent proliferation of HeLa cells. Therefore, the findings of this study indicate that the crude extract from Streptomyces sp. strain KF15 contains antifungal and anticancer metabolites; further study on purification could help in controlling many fungal diseases as well as cervical cancer in humans.









Similar content being viewed by others
Data availability statement
Not applicable.
References
Ahsan T, Chen J, Zhao X, Irfan M, Wu Y (2017) Extraction and identification of bioactive compounds (eicosane and dibutyl phthalate) produced by Streptomyces strain KX852460 for the biological control of Rhizoctonia solani AG-3 strain KX852461 to control target spot disease in tobacco leaf. AMB Expr 7:54. https://doi.org/10.1186/s13568-017-0351-z
Al-Rubaye TS, Risan MH, Al-Rubaye D (2020) Gas chromatography-mass-spectroscopy analysis of bioactive compounds from Streptomyces spp. isolated from Tigris river sediments in Baghdad city. J Biotechnol Res Center 14:1–10. https://doi.org/10.24126/jobrc.2020.14.1.590
Alqahtani SS, Moni SS, Sultan MH, Bakkari MA, Madkhali OA, Alshahrani S, Makeen HA, Menachery SJ, Rehman Z, Alam MS, Mohan S, Elmobark ME, Banji D, Ahmed MZS (2022) Potential bioactive secondary metabolites of actinomycetes sp. isolated from rocky soils of the heritage village Rijal Alma, Saudi Arabia. Arabian J Chem 15:103793. https://doi.org/10.1016/j.arabjc.2022.103793
Ambarvati A, Wahyuono S, Moeljopawiro S, Yuwono T (2020) Antimicrobial activity of ethyl acetate extracts of Streptomyces sp. CRB46 and the prediction of their bioactive compounds chemical structure. Biodivers 21:3380–3390. https://doi.org/10.13057/biodiv/d210763
Anusree T, Bhai RS, Shabeer TPA, Oulkar D (2019) Streptomyces spp. from black pepper rhizosphere: a boundless reservoir of antimicrobial and growth promoting metabolites. J Biol Act Prod Nat 9:1–23. https://doi.org/10.1080/22311866.2018.1561327
Baltz RH (2007) Antimicrobials from actinomycetes: back to the future. Microbe 2:125–131
Belghit S, Driche EH, Bijani C, Zitouni A, Sabaou N, Badji B, Mathieu F (2016) Activity of 2,4-Di-tert-butyl phenol produced by a strain of Streptomyces mutabilis isolated from a Saharan soil against Candida albicans and other pathogenic fungi. J Mycol Med 26:160–169. https://doi.org/10.1016/j.mycmed.2016.03.001
Berdy J (2005) Bioactive microbial metabolites. J Antibiot 58:1–26. https://doi.org/10.1038/ja.2005.1
Biswas K, Bhattarcharya D, Saha M, Mukherjee J, Karmakar S (2022) Evaluation of antimicrobial activity of the extract of Streptomyces euryhalinus isolated from the Indian Sundarban. Arch Microbiol 204:34. https://doi.org/10.1007/s00203-021-02698-5
Chakraborty B, Kumar RS, Almansour AI, Gunasekaran P, Nayaka S (2022) Bioprospection and secondary metabolites profiling of marine Streptomyces levis strain KS46. Saudi J Biol Sci 29:667–679. https://doi.org/10.1016/j.sjbs.2021.11.055
El-Naggar NEA, El-Bindary AAA, Abdel-Mogib M, Nour NS (2017) In vitro activity, extraction, separation and structure elucidation of antibiotic produced by Streptomyces anulatus NEAE-94 active against multidrug-resistant Staphylococcus aureus. Biotechnol Biotechnol Equip 31:418–430. https://doi.org/10.1080/13102818.2016.1276412
Goodfellow M, Williams ST (1983) Ecology of actinomycetes. Ann Rev Microbial 37:189–216. https://doi.org/10.1146/annurev.mi.37.100183.001201
Goodfellow M, Kämpfer P, Busse HJ, Trujillo ME, Suzuki K, Ludwig W, Whitman WB (2012) Bergey’s manual of systematic bacteriology; The Actinobacteria, 2nd Ed.; Springer: New York, pp. 1455–1456. https://doi.org/10.1007/978-0-387-68233-4.
Hui MLY, Tan LTH, Letchumanan V, He YW, Fang CM, Chan KG, Law JWF, Lee LH (2021) The extremophilic actinobacteria: from microbes to medicine. Antibiot 10:682. https://doi.org/10.3390/antibiotics10060682
Hussain SJ, Nowshad MM, Thajuddin N, Tamilarasan TK, Azees PAA (2021) Biodegradation and characterization of Streptomyces sp. (JMCACA3) from acid corroded iron plate. Curr Microbiol 78:1245–1255. https://doi.org/10.1007/s00284-021-02374-3
Jaroszewicz W, Bielanska P, Lubomska D, Kwasnicka KK, Golec P, Grabowski L, Wieczerzak E, Drozdz W, Gaffke L, Pierzynowska K, Wegrzyn G, Wegrzyn A (2021) Antibacterial, antifungal and anticancer activities of compounds produced by newly isolated Streptomyces strains from the Szczelina Chocholowska cave (Tatra mountains, Poland). Antibiot 10:1212. https://doi.org/10.3390/antibiotics10101212
Kekuda PTR, Onkarappa R, Gautham SA, Mesta SC, Raghavendra HL (2015) Antimicrobial, antioxidant and cytotoxic activity of Streptomyces species from western ghat soils of Karnataka, India. Sci Technol Arts Res J 4:164–180. https://doi.org/10.4314/star.v4i2.20
Kitouni M, Boudemagh A, Oulmi L, Reghioua S, Boughachiche F, Zerizer F, Hamdiken H, Couble A, Mouniee D, Boulahrouf A, Boiron P (2005) Isolation of actinomycetes producing bioactive substances from water, soil and tree bark samples of the north-east of Algeria. J Mycol Med 15:45–51
Krasnov C, Ziv C (2022) Non-chemical approaches to control postharvest gray mold disease in bell peppers. Agron 12:216. https://doi.org/10.3390/agronomy12010216
Krishnamoorthy M, Dharmaraj D, Rajendran K, Karuppiah K, Balasubramanian M, Ethiraj K (2020) Pharmacological activities of coral reef associated actinomycetes, Saccharopolyspora sp. IMA1. Biocatal Agric Biotechnol 28:101748. https://doi.org/10.1016/j.bcab.2020.101748
Kumar P, Chauhan A, Kumar M, Kuanr BK, Kundu A, Solanki R, Kapur MK (2021) In vitro and in silico anticancer potential analysis of Streptomyces sp. extract against human lung cancer cell line, A549. 3 Biotech 11:254. https://doi.org/10.1007/s13205-021-02812-w
Kumar V, Naik B, Gusain O, Bisht GS (2014) An actinomycete isolate from solitary wasp mud nest having strong antibacterial activity and kills the Candida cells due to the shrinkage and the cytosolic loss. Front Microbiol 5:446. https://doi.org/10.3389/fmicb.2014.00446
Lammers A, Zweers H, Sandfeld T, Bilde T, Garbeva P, Schramm A, Lalk M (2021) Antimicrobial compounds in the volatilome of social spider communities. Front Microbiol 12:700693. https://doi.org/10.3389/fmicb.2021.700693
Li X, Jing T, Zhou D, Zhang M, Qi D, Zang X, Zhao Y, Li K, Tang W, Chen Y, Qi C, Wang W, Xie J (2021) Biocontrol efficacy and possible mechanism of Streptomyces sp. H4 against postharvest anthracnose caused by Colletotrichum fragariae on strawberry fruit. Postharvest Biol Technol 175:111401. https://doi.org/10.1016/j.postharvbio.2020.111401
Ma A, Jiang K, Chen B, Chen S, Qi X, Lu H, Liu J, Zhou X, Gao T, Li J, Zhao C (2021) Evaluation of the anticarcinogenic potential of the endophyte, Streptomyces sp. LRE541 isolated from Lilium davidii var. unicolor (Hoog) cotton. Microb Cell Fact 20:217. https://doi.org/10.1186/s12934-021-01706-z
Marimuthu S, Karthic C, Mostafa AA, Al-Enazi NM, Abdel-Raouf N, Sholkamy EN (2020) Antifungal activity of Streptomyces sp. SLR03 against tea fungal plant pathogen Pestalotiopsis theae. J King Saud Univ Sci 32:3258–3264. https://doi.org/10.1016/j.jksus.2020.08.027
Meletiadis J, Dorsthorst DTA, Verweij PE (2003) Use of turbidimetric growth curves for early determination of antifungal drug resistance of filamentous fungi. J Clin Microbiol 40:4718–4725. https://doi.org/10.1128/JCM.41.10.4718-4725.2003
Mohamed H, Hassane A, Rawway M, El-Sayed M, Gomaa AR, Abdul-Raouf U, Shah AM, Abdelmotaal H, Song Y (2021) Antibacterial and cytotoxic potency of thermophilic Streptomyces werraensis MI-S.24-3 isolated from an Egyptian extreme environment. Arch Microbiol 203:4961–4972. https://doi.org/10.1007/s00203-021-02487-0
Muthuraj R, Bhat MP, Dhanyakumara SB, Halaswamy HM, Shashiraj KN, Chakraborty B, Pallavi SS, Nayaka S (2021) Isolation, identification and characterization of antimicrobial activity exhibiting actinomycete Streptomyces paradoxus strain KUASN-7 from soil. Int J Curr Microbiol App Sci 10:164–176. https://doi.org/10.20546/ijcmas.2021.1008.021
Nandhini US, Sangareshwari S, Lata K (2015) Gas chromatography-mass spectrometry analysis of bioactive constituents from the marine Streptomyces. Asian J Pharm Clin Res 8:244–246
Nayaka S, Chakraborty B, Bhat MP, Nagaraja SK, Airodagi D, Swamy PS, Rudrappa M, Hiremath H, Basavarajappa DS, Kanakannanavar B (2020) Biosynthesis, characterization, and in vitro assessment on cytotoxicity of actinomycete-synthesized silver nanoparticles on Allium cepa root tip cells. Beni-Suef Univ J Basic Appl Sci 9:51. https://doi.org/10.1186/s43088-020-00074-8
Pallavi SS, Bhat MP, Nayaka S (2021) Microbial synthesis of silver nanoparticles using Streptomyces sp. PG12 and their characterization, antimicrobial activity and cytotoxicity assessment against human lung (A549) and breast (MCF-7) cancer cell lines. Int J Pharm Pharm Sci 13:94–102. https://doi.org/10.22159/ijpps.2021v13i8.41876
Raja A, Prabakarana P (2011) Actinomycetes and drugs—an overview. Am J Drug Discovery Dev 1:75–84. https://doi.org/10.3923/ajdd.2011.75.84
Reygaert WC (2018) An overview of the antimicrobial resistance mechanisms of bacteria. AIMS Microbiol. 4:482–501. https://doi.org/10.3934/microbiol.2018.3.482
Sharma S, Fulke AB, Chaubey A (2019) Bioprospection of marine actinomycetes: recent advances, challenges and future perspectives. Acta Oceanol Sin 38(6):1–17. https://doi.org/10.1007/s13131-018-1340-z
Shekar P, Kumar KS, Gopikrishnan V, Radhakrishnan M (2016) HPTLC fingerprint profile of antibacterial compound produced from forest soil Streptomyces SFA5. Bangladesh J Pharmacol 11:295–300. https://doi.org/10.3329/bjp.v11i2.25478
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72:7–33. https://doi.org/10.3322/caac.21708
Singh M, Jha RP, Shri N, Bhattacharyya K, Patel P, Dhamnetiya D (2022) Secular trends in incidence and mortality of cervical cancer in India and its states, 1990–2019: data from the Global Burden of Disease 2019 Study. BMC Cancer 22:149. https://doi.org/10.1186/s12885-022-09232-w
Sonawane VB, Shinde HP (2021) Anthracnose disease of Capsicum annuum L. and its bio control management: a review. Appl Ecol Environ Sci 9:172–176. https://doi.org/10.12691/aees-9-2-8
Taddei A, Rodriguez MJ, Marquez-Vilchez E, Castelli C (2006) Isolation and identification of Streptomyces spp. from Venezuelan soils: morphological and biochemical studies. Microbiol Res 161:222–231. https://doi.org/10.1016/j.micres.2005.08.004
Taechowisan T, Samsawat T, Puckdee W, Phutdhawong WS (2021) Cytotoxicity and antibacterial activities of crude extract of Streptomyces sp. W08, an endophyte of Amomum krervanh Pierre. J Appl Pharm Sci 11:134–138. https://doi.org/10.7324/JAPS.2021.110818
Tan LTH, Chan KG, Pusparajah P, Yin WF, Khan TM, Lee LH, Goh BH (2019) Mangrove habitat derived Streptomyces sp. MUM265 as a potential source of antioxidant and anticolon-cancer agents. BMC Microbiol 19:38. https://doi.org/10.1186/s12866-019-1409-7
Thakur D, Yadav A, Gogoi BK, Bora TC (2007) Isolation and screening of Streptomyces in soil of protected forest areas from the states of Assam and Tripura, India, for antimicrobial metabolites. J Mycol Med 17:242–249. https://doi.org/10.1016/j.mycmed.2007.08.001
Vijayakumar R, Panneerselvam K, Muthukumar C, Thajuddin N, Panneerselvam A, Saravanamuthu R (2012) Optimization of antimicrobial production by a marine actinomycete Streptomyces afghaniensis VPTS3–1 isolated from Palk Strait, east coast of India. Indian J Microbiol 52:230–239. https://doi.org/10.1007/s12088-011-0138-x
Zou N, Zhou D, Chen Y, Lin P, Chen Y, Wang W, Xie J, Wang M (2021) A novel antifungal actinomycete Streptomyces sp. strain H3–2 effectively controls banana Fusarium wilt. Front Microbiol 12:706647. https://doi.org/10.3389/fmicb.2021.706647
Acknowledgements
The authors are thankful to P.G. Department of Studies in Botany for extending laboratory facility and to Sophisticated Analytical Instrumentation Facility (SAIF), University Scientific Instrumentation Centre, Karnatak University, Dharwad, for providing necessary instrumentation facilities. The authors are thankful to Department of Plant pathology, U.A.S., Dharwad, for providing fungal pathogens of chilli.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
The research concept was designed by SN and MPB with supervision done by SN. MPB carried out the experimental methodologies and SN and RSK did the data validation. MPB wrote the original draft of the manuscript along with review and editing. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bhat, M.P., Nayaka, S. & Kumar, R.S. A swamp forest Streptomyces sp. strain KF15 with broad spectrum antifungal activity against chilli pathogens exhibits anticancer activity on HeLa cells. Arch Microbiol 204, 540 (2022). https://doi.org/10.1007/s00203-022-03147-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03147-7