Abstract
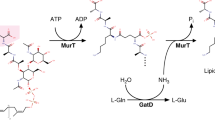
Chlamydiaceae are obligate intracellular bacteria that do not synthesise detectable peptidoglycan although they possess an almost complete arsenal of genes encoding peptidoglycan biosynthetic activities. In this paper, the murF gene from Chlamydia trachomatis was shown to be capable of complementing a conditional Escherichia coli mutant impaired in UDP-MurNAc-tripeptide:d-Ala-d-Ala ligase activity. Recombinant MurF from C. trachomatis was overproduced and purified from E. coli. It exhibited ATP-dependent UDP-MurNAc-X-γ-d-Glu-meso-A2pm:d-Ala-d-Ala ligase activity in vitro. No significant difference of kinetic parameters was seen when X was l-Ala, l-Ser or Gly. The l-Lys-containing UDP-MurNAc-tripeptide was a poorer substrate as compared to the meso-A2pm-containing one. Based on the respective substrate specificities of the chlamydial MurC, MurE, MurF and Ddl enzymes, a sequence l-Ala/l-Ser/Gly-γ-d-Glu-meso-A2pm-d-Ala-d-Ala is expected for the chlamydial pentapeptide stem, with Gly at position 1 being less likely.



Similar content being viewed by others
References
Amann E, Ochs B, Abel KJ (1988) Tightly regulated tac promoter vectors useful for the expression of unfused and fused proteins in Escherichia coli. Gene 69:301–315
Amrein KE, Takacs B, Stieger M, Molnos J, Flint NA, Burn P (1995) Purification and characterization of recombinant human p50csk protein-tyrosine kinase from an Escherichia coli expression system overproducing the bacterial chaperones GroES and GroEL. Proc Natl Acad Sci USA 92:1048–1052
Anderson MS, Eveland SS, Onishi HR, Pompliano DL (1996) Kinetic mechanism of the Escherichia coli UDPMurNAc-tripeptide d-alanyl-d-alanine-adding enzyme: use of a glutathione S-transferase fusion. Biochemistry 35:16264–16269
Auger G et al (1998) Large-scale preparation, purification, and crystallization of UDP-N-acetylmuramoyl-l-alanine: d-glutamate ligase from Escherichia coli. Protein Expr Purif 13:23–29
Babič A et al (2007) Chemoenzymatic synthesis of the nucleotide substrates of the Mur ligases. In: Kikelj D (ed) 5th Joint meeting on medicinal chemistry, June 17–21, Portorož, Slovenia. Medimond Srl, Bologna, Italy, pp 1–4
Barreteau H, Kovač A, Boniface A, Sova M, Gobec S, Blanot D (2008) Cytoplasmic steps of peptidoglycan biosynthesis. FEMS Microbiol Rev 32:168–207
Bolanos-Garcia VM, Davies OR (2006) Structural analysis and classification of native proteins from E. coli commonly co-purified by immobilised metal affinity chromatography. Biochim Biophys Acta 1760:1304–1313
Boniface A, Bouhss A, Mengin-Lecreulx D, Blanot D (2006) The MurE synthetase from Thermotoga maritima is endowed with an unusual d-lysine adding activity. J Biol Chem 281:15680–15686
Caravano A, Mengin-Lecreulx D, Brondello JM, Vincent SP, Sinaÿ P (2003) Synthesis and inhibition properties of conformational probes for the mutase-catalyzed UDP-galactopyranose/furanose interconversion. Chem Eur J 9:5888–5898
Chopra I, Storey C, Falla TJ, Pearce JH (1998) Antibiotics, peptidoglycan synthesis and genomics: the chlamydial anomaly revisited. Microbiology 144:2673–2678
Dagert M, Ehrlich SD (1979) Prolonged incubation in calcium chloride improves the competence of Escherichia coli cells. Gene 6:23–28
Dementin S (2001) Etude du mécanisme réactionnel des Mur synthétases, enzymes impliquées dans la biosynthèse du peptidoglycane bactérien. Ph.D. Thesis, Université Paris-Sud, Orsay, France
Dementin S et al (2001) Evidence of a functional requirement for a carbamoylated lysine residue in MurD, MurE and MurF synthetases as established by chemical rescue experiments. Eur J Biochem 268:5800–5807
Dover LG (2007) What genomics has taught about bacterial cell wall biosynthesis. In: Pallen MJ, Nelson KE, Preston GM (eds) Bacterial pathogenomics. ASM Press, Washington, pp 327–360
Ghuysen JM, Goffin C (1999) Lack of cell wall peptidoglycan versus penicillin sensitivity: new insights into the chlamydial anomaly. Antimicrob Agents Chemother 43:2339–2344
Gordon E, Flouret B, Chantalat L, van Heijenoort J, Mengin-Lecreulx D, Dideberg O (2001) Crystal structure of UDP-N-acetylmuramoyl-l-alanyl-d-glutamate: meso-diaminopimelate ligase from Escherichia coli. J Biol Chem 276:10999–11006
Hatch TP (1996) Disulfide cross-linked envelope proteins: the functional equivalent of peptidoglycan in chlamydiae? J Bacteriol 178:1–5
Healy VL, Park IS, Walsh CT (1998) Active-site mutants of the VanC2 d-alanyl-d-serine ligase, characteristic of one vancomycin-resistant bacterial phenotype, revert towards wild-type d-alanyl-d-alanine ligases. Chem Biol 5:197–207
Hesse L, Bostock J, Dementin S, Blanot D, Mengin-Lecreulx D, Chopra I (2003) Functional and biochemical analysis of Chlamydia trachomatis MurC, an enzyme displaying UDP-N-acetylmuramate: amino acid ligase activity. J Bacteriol 185:6507–6512
Liger D, Masson A, Blanot D, van Heijenoort J, Parquet C (1995) Over-production, purification and properties of the uridine-diphosphate-N-acetylmuramate: l-alanine ligase from Escherichia coli. Eur J Biochem 230:80–87
Longenecker KL et al (2005) Structure of MurF from Streptococcus pneumoniae co-crystallized with a small molecule inhibitor exhibits interdomain closure. Protein Sci 14:3039–3047
Lugtenberg EJJ, van Schijndel-van Dam A (1972) Temperature-sensitive mutants of Escherichia coli K-12 with low activities of the l-alanine adding enzyme and the d-alanyl-d-alanine adding enzyme. J Bacteriol 110:35–40
Lugtenberg EJJ, De Haas-Menger L, Ruyters WHM (1972) Murein synthesis and identification of cell wall precursors of temperature-sensitive lysis mutants of Escherichia coli. J Bacteriol 109:326–335
McCoy AJ, Maurelli AT (2005) Characterization of Chlamydia MurC-Ddl, a fusion protein exhibiting d-alanyl-d-alanine ligase activity involved in peptidoglycan synthesis and d-cycloserine sensitivity. Mol Microbiol 57:41–52
McCoy AJ, Maurelli AT (2006) Building the invisible wall: updating the chlamydial peptidoglycan anomaly. Trends Microbiol 14:70–77
Mengin-Lecreulx D, Falla T, Blanot D, van Heijenoort J, Adams DJ, Chopra I (1999) Expression of the Staphylococcus aureus UDP-N-acetylmuramoyl-l-alanyl-d-glutamate: l-lysine ligase in Escherichia coli and effects on peptidoglycan biosynthesis and cell growth. J Bacteriol 181:5909–5914
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, pp 431–435
Moulder JW (1993) Why is Chlamydia sensitive to penicillin in the absence of peptidoglycan? Infect Agents Dis 2:87–99
Park IS, Walsh CT (1997) d-alanyl-d-lactate and d-alanyl-d-alanine synthesis by d-alanyl-d-alanine ligase from vancomycin-resistant Leuconostoc mesenteroides. Effects of a phenylalanine 261 to tyrosine mutation. J Biol Chem 272:9210–9214
Patin D, Bostock J, Blanot D, Mengin-Lecreulx D, Chopra I (2009) Functional and biochemical analysis of the Chlamydia trachomatis ligase MurE. J Bacteriol 191:7430–7435
Patin D et al (2010) Purification and biochemical characterization of Mur ligases from Staphylococcus aureus. Biochimie 92:1793–1800
Pompeo F, van Heijenoort J, Mengin-Lecreulx D (1998) Probing the role of cysteine residues in glucosamine-1-phosphate acetyltransferase activity of the bifunctional GlmU protein from Escherichia coli: site-directed mutagenesis and characterization of the mutant enzymes. J Bacteriol 180:4799–4803
Press WH, Flannery BP, Teukolsky SA, Vetterling WT (1986) Numerical recipes: the art of scientific computing. Cambridge University Press, Cambridge
Saka HA, Valdivia RH (2010) Acquisition of nutrients by Chlamydiae: unique challenges of living in an intracellular compartment. Curr Opin Microbiol 13:4–10
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schachter J (1992) Chlamydia. In: Gorbach SL, Bartlett JG, Blacklow NR (eds) Infectious diseases. W. B. Saunders, Philadelphia, pp 1633–1641
Schleifer KH, Kandler O (1972) Peptidoglycan types of bacterial cell walls and their taxonomic implications. Bacteriol Rev 36:407–477
Storey C, Chopra I (2001) Affinities of β-lactams for penicillin binding proteins of Chlamydia trachomatis and their antichlamydial activities. Antimicrob Agents Chemother 45:303–305
Vollmer W, Blanot D, de Pedro MA (2008) Peptidoglycan structure and architecture. FEMS Microbiol Rev 32:149–167
Yan Y, Munshi S, Leiting B, Anderson MS, Chrzas J, Chen Z (2000) Crystal structure of Escherichia coli UDPMurNAc-tripeptide d-alanyl-d-alanine-adding enzyme (MurF) at 2.3 Å resolution. J Mol Biol 304:435–445
Acknowledgments
This work was supported by grants from the Centre National de la Recherche Scientifique (UMR 8619 and PICS 2950) and the European Union EUR-INTAFAR project (LSHM-CT-2004512138).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jean-Luc Pernodet.
Rights and permissions
About this article
Cite this article
Patin, D., Bostock, J., Chopra, I. et al. Biochemical characterisation of the chlamydial MurF ligase, and possible sequence of the chlamydial peptidoglycan pentapeptide stem. Arch Microbiol 194, 505–512 (2012). https://doi.org/10.1007/s00203-011-0784-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-011-0784-8