Abstract
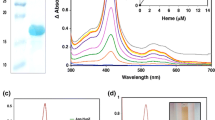
Escherichia coli Hmp is a homologue of Ralstonia eutropha FHP, the first reported bacterial flavohaemoglobin, and functions in NO detoxification. Photolysis of CO-ligated Hmp in the presence of oxygen gave a photodissociable oxy species with kon 2.82×107 M−1 s−1 and koff 4.49×103 s−1. The dissociation constant of the primary O2 compound was 160 μM (25°C, pH 7.0). In order to detect superoxide formation, ferric horseradish peroxidase was used. Hmp formed the oxy compound within milliseconds, followed by formation of compound III, arising from superoxide formation. The rate of superoxide formation was independent of oxygen concentration between 0.05 and 0.7 mM oxygen, suggesting a Km <0.05 mM. During prolonged oxidation of NADH, the spectral signals of Hmp decayed and iron was released in a process prevented by superoxide dismutase or catalase. NADH oxidation by purified Hmp was characterised by progressive slowing of oxygen uptake. Inclusion of NO, superoxide dismutase or catalase during NADH oxidation partially protected oxygen uptake, consistent with the formation, in the absence of NO, of reactive oxygen species that inhibit Hmp function. The results are discussed in relation to the tight control exerted on Hmp synthesis in vivo.








Similar content being viewed by others
References
Andrews SC, Shipley D, Keen JN, Findlay JBC, Harrison PM, Guest JR (1992) The haemoglobin-like protein (HMP) of Escherichia coli has ferrisiderophore reductase activity and its C-terminal domain shares homology with ferredoxin NADP+ reductases. FEBS Lett 302:247–252
Bolann BJ, Ulvik RJ (1990) On the limited ability of superoxide to release iron from ferritin. Eur J Biochem 193:899–904
Cooper CE, Ioannidis N, D’mello R, Poole RK (1994) Haem, flavin and oxygen interactions in Hmp, a flavohaemoglobin from Escherichia coli. Biochem Soc Trans 22:709–713
Cramm R, Siddiqui RA, Friedrich B (1994) Primary sequence and evidence for a physiological function of the flavohemoprotein of Alcaligenes eutrophus. J Biol Chem 269:7349–7354
Cruz-Ramos H, Crack J, Wu G, Hughes MN, Scott S, Thomson AJ, Green J, Poole RK (2002) NO sensing by Fnr: regulation of the Escherichia coli NO-detoxifying flavohaemoglobin, Hmp. EMBO J 21:3235–3244
Ermler U, Siddiqui RA, Cramm R, Friedrich B (1995) Crystal structure of the flavohemoglobin from Alcaligenes eutrophus at 1.75 angstrom resolution. EMBO J 14:6067–6077
Gardner PR, Gardner AM, Martin LA, Salzman AL (1998) Nitric oxide dioxygenase: an enzymic function for flavohemoglobin. Proc Natl Acad Sci USA 95:10378–10383
Gardner AM, Martin LA, Gardner PM, Dou Y, Olson JS (2000) Steady-state and transient kinetics of Escherichia coli nitric-oxide dioxygenase (flavohemoglobin). The B10 tyrosine hydroxyl is essential for dioxygen binding and catalysis. J Biol Chem 275:12581–12589
Greenwood C, Gibson QH (1967) The reaction of reduced cytochrome c oxidase with oxygen. J Biol Chem 242:1782–1787
Hausladen A, Gow AJ, Stamler JS (1998) Nitrosative stress: metabolic pathway involving the flavohemoglobin. Proc Natl Acad Sci USA 95:14100–14105
Hernández-Urzua E, Mills CE, White GP, Contreras-Zentella ML, Escamilla E, Vasudevan SG, Membrillo-Hernández J, Poole RK (2003) Flavohaemoglobin Hmp, but not its individual domains, confers protection from respiratory inhibition by nitric oxide in Escherichia coli. J Biol Chem 278:34975–34982
Ilari A, Bonamore A, Farina A, Johnson KA, Boffi A (2002) The X-ray structure of ferric Escherichia coli flavohaemoglobin reveals an unexpected geometry of the distal heme pocket. J Biol Chem 277:23725–23732
Imlay JA (2002) How oxygen damages microbes: oxygen tolerance and obligate anaerobiosis. Adv Microb Physiol 46:111–153
Ioannidis N, Cooper CE, Poole RK (1992) Spectroscopic studies on an oxygen-binding haemoglobin-like flavohaemoprotein from Escherichia coli. Biochem J 288:649–655
Isogai Y, Iizuka T, Makino R, Iyanagi T, Orii Y (1993) Superoxide-producing cytochrome b. Enzymatic and electron paramagnetic resonance properties of cytochrome b558 purified from neutrophils. J Biol Chem 268:4025–4031
Kalnenieks U, Galinina N, Bringer-Meyer S, Poole RK (1998) Membrane d-lactate oxidase in Zymomonas mobilis: evidence for a branched respiratory chain. FEMS Microbiol Lett 168:91–97
Membrillo-Hernández J, Ioannidis N, Poole RK (1996) The flavohaemoglobin (HMP) of Escherichia coli generates superoxide in vitro and causes oxidative stress in vivo. FEBS Lett 382:141–144
Membrillo-Hernández J, Kim SO, Cook GM, Poole RK (1997) Paraquat regulation of hmp (flavohaemoglobin) gene expression in Escherichia coli K-12 is SoxRS independent but modulated by σs. J Bacteriol 179:3164–3170
Membrillo-Hernández J, Coopamah MD, Channa A, Hughes MN, Poole RK (1998) A novel mechanism for upregulation of the Escherichia coli K-12 hmp (flavohaemoglobin) gene by the “NO releaser”, S-nitrosoglutathione: nitrosation of homocysteine and modulation of MetR binding to the glyA-hmp intergenic region. Mol Microbiol 29:1101–1112
Messner KR, Imlay JA (1999) The identification of primary sites of superoxide and hydrogen peroxide formation in the aerobic respiratory chain and sulfite reductase complex of Escherichia coli. J Biol Chem 274:10119–10128
Mills CE, Sedelnikova S, Soballe B, Hughes MN, Poole RK (2001) Escherichia coli flavohaemoglobin (Hmp) with equistoichiometric FAD and haem contents has a low affinity for dioxygen in the absence or presence of nitric oxide. Biochem J 353:207–213
Mogi T, Hirano T, Nakamura H, Anraku Y, Orii Y (1995) CuB promotes both binding and reduction of dioxygen at the heme-copper binuclear center in the Escherichia coli bo-type ubiquinol oxidase. FEBS Lett 370:259–263
Nauseef WM (1999) The NADPH-dependent oxidase of phagocytes. Proc Assoc Am Phys 111:373–382
Orii Y (1993) Immediate reduction of cytochrome c by photoexcited NADH: reaction mechanism as revealed by flow-flash and rapid-scan studies. Biochemistry 32:11910–11914
Orii Y, Ioannidis N, Poole RK (1992) The oxygenated flavohaemoglobin from Escherichia coli: evidence from photodissociation and rapid-scan studies for two kinetic and spectral forms. Biochem Biophys Res Commun 187:94–100
Oshino R, Asakura T, Takio K, Oshino N, Chance B, Hagihara B (1973) Purification and molecular properties of yeast hemoglobin. Eur J Biochem 39:581–590
Poole RK, Hughes MN (2000) New functions for the ancient globin family: bacterial responses to nitric oxide and nitrosative stress. Mol Microbiol 36:775–783
Poole RK, Ioannidis N, Orii Y (1994) Reactions of the Escherichia coli flavohaemoglobin (Hmp) with oxygen and reduced nicotinamide adenine dinucleotide: evidence for oxygen switching of flavin oxidoreduction and a mechanism for oxygen sensing. Proc R Soc Lond Ser B Biol Sci 255:251–258
Poole RK, Anjum MF, Membrillo-Hernández J, Kim SO, Hughes MN, Stewart V (1996a) Nitric oxide, nitrite, and fnr regulation of hmp (flavohemoglobin) gene expression in Escherichia coli K-12. J Bacteriol 178:5487–5492
Poole RK, Ioannidis N, Orii Y (1996b) Reactions of the Escherichia coli flavohaemoglobin (Hmp) with NADH and near-micromolar oxygen: oxygen affinity of NADH oxidase activity. Microbiology 142:1141–1148
Poole RK, Rogers NJ, D’Mello RAM, Hughes MN, Orii Y (1997) Escherichia coli flavohaemoglobin (Hmp) reduces cytochrome c and Fe(III)-hydroxamate K by electron transfer from NADH via FAD: sensitivity of oxidoreductase activity to haem-bound dioxygen. Microbiology 143:1557–1565
Prasad MR, Engelman RM, Jones RM, Das DK (1989) Effects of oxyradicals on oxymyoglobin. Deoxygenation, haem removal and iron release. Biochem J 263:731–736
Probst I, Schlegel HG (1976) Respiratory components and oxidase activities in Alcaligenes eutrophus. Biochim Biophys Acta 440:412–428
Probst I, Wolf G, Schlegel HG (1979) An oxygen-binding flavohemoprotein from Alcaligenes eutrophus. Biochim Biophys Acta 576:471–478
Stevanin T, Poole RK, Demoncheaux EAG, Read RC (2002) Flavohemoglobin Hmp protects Salmonella enterica serovar typhimurium from nitric oxide-related killing by human macrophages. Infect Immun 70:4399–4405
Stookey L (1970) Ferrozine-a new spectrophotometric reagent for iron. Anal Chem 42:779–781
Vasudevan SG, Armarego WLF, Shaw DC, Lilley PE, Dixon NE, Poole RK (1991) Isolation and nucleotide sequence of the hmp gene that encodes a haemoglobin-like protein in Escherichia coli K-12. Mol Gen Genet 226:49–58
Wakabayashi S, Matsubara H, Webster DA (1986) Primary sequence of a dimeric bacterial haemoglobin from Vitreoscilla. Nature 322:481–483
Wu G, Wainwright LM, Poole RK (2003) Microbial globins. Adv Microb Physiol 47:255–310
Xu F, Quandt KS, Hultquist DE (1992) Characterization of NADPH-dependent methemoglobin reductase as a heme-binding protein present in erythrocytes and liver. Proc Natl Acad Sci 89:2130–2134
Yamazaki I (1974) Peroxidase. In: Hayaishi O (ed) Molecular mechanisms of oxygen activation. Academic, New York, pp 535–558
Acknowledgements
This work was supported in RKP’s laboratory by BBSRC Research Grant P18939.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is dedicated to Professor Dr Hans G. Schlegel, on the occasion of his 80th birthday.
Rights and permissions
About this article
Cite this article
Wu, G., Corker, H., Orii, Y. et al. Escherichia coli Hmp, an “oxygen-binding flavohaemoprotein”, produces superoxide anion and self-destructs. Arch Microbiol 182, 193–203 (2004). https://doi.org/10.1007/s00203-004-0699-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-004-0699-8