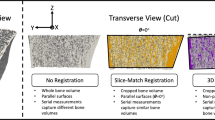
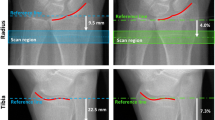
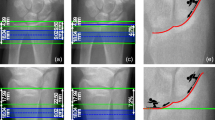
Abstract
Summary
High-resolution peripheral quantitative computed tomography (HR-pQCT) is a powerful tool to assess bone health. To determine how an individual’s or population of interest’s HR-pQCT outcomes compare to expected, reference data are required. This study provides reference data for HR-pQCT measures acquired in a population of White adults.
Purpose
To provide age- and sex-specific reference data for high-resolution peripheral quantitative computed tomography (HR-pQCT) measures of the distal and diaphyseal radius and tibia acquired using a second-generation scanner and percent-of-length offsets proximal from the end of the bone.
Methods
Data were acquired in White adults (aged 18–80 years) living in the Midwest region of the USA. HR-pQCT scans were performed at the 4% distal radius, 30% diaphyseal radius, 7.3% distal tibia, and 30% diaphyseal tibia. Centile curves were fit to the data using the LMS approach.
Results
Scans of 867 females and 317 males were included. The fitted centile curves reveal HR-pQCT differences between ages, sexes, and sites. They also indicate differences when compared to data obtained by others using fixed length offsets. Excel-based calculators based on the current data were developed and are provided to enable computation of subject-specific percentiles, z-scores, and t-scores and to plot an individual’s outcomes on the fitted curves. In addition, regression equations are provided to convert estimated failure load acquired with the conventional criteria utilized with first-generation scanners and those specifically developed for second-generation scanners.
Conclusion
The current study provides unique data and resources. The combination of the reference data and calculators provide clinicians and investigators an ability to assess HR-pQCT outcomes in an individual or population of interest, when using the described scanning and analysis procedure. Ultimately, the expectation is these data will be expanded over time so the wealth of information HR-pQCT provides becomes increasingly interpretable and utilized.







Similar content being viewed by others
References
Mikolajewicz N, Bishop N, Burghardt AJ, Folkestad L, Hall A, Kozloff KM et al (2020) HR-pQCT measures of bone microarchitecture predict fracture: systematic review and meta-analysis. J Bone Miner Res 35(3):446–459. https://doi.org/10.1002/jbmr.3901
Samelson EJ, Broe KE, Xu H, Yang L, Boyd S, Biver E et al (2019) Cortical and trabecular bone microarchitecture as an independent predictor of incident fracture risk in older women and men in the bone microarchitecture international consortium (BoMIC): a prospective study. Lancet Diabetes Endocrinol 7(1):34–43. https://doi.org/10.1016/s2213-8587(18)30308-5
Alvarenga JC, Fuller H, Pasoto SG, Pereira RM (2017) Age-related reference curves of volumetric bone density, structure, and biomechanical parameters adjusted for weight and height in a population of healthy women: an HR-pQCT study. Osteoporos Int 28(4):1335–1346. https://doi.org/10.1007/s00198-016-3876-0
Burghardt AJ, Kazakia GJ, Ramachandran S, Link TM, Majumdar S (2010) Age- and gender-related differences in the geometric properties and biomechanical significance of intracortical porosity in the distal radius and tibia. J Bone Miner Res 25(5):983–993. https://doi.org/10.1359/jbmr.091104
Burt LA, Liang Z, Sajobi TT, Hanley DA, Boyd SK (2016) Sex- and site-specific normative data curves for HR-pQCT. J Bone Miner Res 31(11):2041–2047. https://doi.org/10.1002/jbmr.2873
Burt LA, Macdonald HM, Hanley DA, Boyd SK (2014) Bone microarchitecture and strength of the radius and tibia in a reference population of young adults: an HR-pQCT study. Arch Osteoporos 9:183. https://doi.org/10.1007/s11657-014-0183-2
Dalzell N, Kaptoge S, Morris N, Berthier A, Koller B, Braak L et al (2009) Bone micro-architecture and determinants of strength in the radius and tibia: age-related changes in a population-based study of normal adults measured with high-resolution pQCT. Osteoporos Int 20(10):1683–1694. https://doi.org/10.1007/s00198-008-0833-6
Gabel L, Macdonald HM, Nettlefold LA, McKay HA (2018) Sex-, ethnic-, and age-specific centile curves for pQCT- and HR-pQCT-derived measures of bone structure and strength in adolescents and young adults. J Bone Miner Res 33(6):987–1000. https://doi.org/10.1002/jbmr.3399
Hansen S, Shanbhogue V, Folkestad L, Nielsen MM, Brixen K (2014) Bone microarchitecture and estimated strength in 499 adult Danish women and men: a cross-sectional, population-based high-resolution peripheral quantitative computed tomographic study on peak bone structure. Calcif Tissue Int 94(3):269–281. https://doi.org/10.1007/s00223-013-9808-5
Hung VW, Zhu TY, Cheung WH, Fong TN, Yu FW, Hung LK et al (2015) Age-related differences in volumetric bone mineral density, microarchitecture, and bone strength of distal radius and tibia in Chinese women: a high-resolution pQCT reference database study. Osteoporos Int 26(6):1691–1703. https://doi.org/10.1007/s00198-015-3045-x
Khosla S, Riggs BL, Atkinson EJ, Oberg AL, McDaniel LJ, Holets M et al (2006) Effects of sex and age on bone microstructure at the ultradistal radius: a population-based noninvasive in vivo assessment. J Bone Miner Res 21(1):124–131. https://doi.org/10.1359/jbmr.050916
Macdonald HM, Nishiyama KK, Kang J, Hanley DA, Boyd SK (2011) Age-related patterns of trabecular and cortical bone loss differ between sexes and skeletal sites: a population-based HR-pQCT study. J Bone Miner Res 26(1):50–62. https://doi.org/10.1002/jbmr.171
Stuck AK, Schenk D, Zysset P, Bütikofer L, Mathis A, Lippuner K (2020) Reference values and clinical predictors of bone strength for HR-pQCT-based distal radius and tibia strength assessments in women and men. Osteoporos Int 31(10):1913–1923. https://doi.org/10.1007/s00198-020-05405-0
Vilayphiou N, Boutroy S, Sornay-Rendu E, Van Rietbergen B, Chapurlat R (2016) Age-related changes in bone strength from HR-pQCT derived microarchitectural parameters with an emphasis on the role of cortical porosity. Bone 83:233–240. https://doi.org/10.1016/j.bone.2015.10.012
Whittier DE, Burt LA, Hanley DA, Boyd SK (2020) Sex- and site-specific reference data for bone microarchitecture in adults measured using second-generation HR-pQCT. J Bone Miner Res 35(11):2151–2158. https://doi.org/10.1002/jbmr.4114
Yu F, Xu Y, Hou Y, Lin Y, Jiajue R, Jiang Y et al (2020) Age-, site-, and sex-specific normative centile curves for HR-pQCT-derived microarchitectural and bone strength parameters in a Chinese mainland population. J Bone Miner Res 35(11):2159–2170. https://doi.org/10.1002/jbmr.4116
Zhu TY, Yip BH, Hung VW, Choy CW, Cheng KL, Kwok TC et al (2018) Normative standards for HRpQCT parameters in Chinese men and women. J Bone Miner Res 33(10):1889–1899. https://doi.org/10.1002/jbmr.3481
Manske SL, Zhu Y, Sandino C, Boyd SK (2015) Human trabecular bone microarchitecture can be assessed independently of density with second generation HR-pQCT. Bone 79:213–221. https://doi.org/10.1016/j.bone.2015.06.006
Bonaretti S, Majumdar S, Lang TF, Khosla S, Burghardt AJ (2017) The comparability of HR-pQCT bone measurements is improved by scanning anatomically standardized regions. Osteoporos Int 28(7):2115–2128. https://doi.org/10.1007/s00198-017-4010-7
Ghasem-Zadeh A, Burghardt A, Wang XF, Iuliano S, Bonaretti S, Bui M et al (2017) Quantifying sex, race, and age specific differences in bone microstructure requires measurement of anatomically equivalent regions. Bone 101:206–213. https://doi.org/10.1016/j.bone.2017.05.010
Shanbhogue VV, Hansen S, Halekoh U, Brixen K (2015) Use of relative vs fixed offset distance to define region of interest at the distal radius and tibia in high-resolution peripheral quantitative computed tomography. J Clin Densitom 18(2):217–225. https://doi.org/10.1016/j.jocd.2014.12.002
Boyd SK (2008) Site-specific variation of bone micro-architecture in the distal radius and tibia. J Clin Densitom 11(3):424–430. https://doi.org/10.1016/j.jocd.2007.12.013
Whittier DE, Boyd SK, Burghardt AJ, Paccou J, Ghasem-Zadeh A, Chapurlat R et al (2020) Guidelines for the assessment of bone density and microarchitecture in vivo using high-resolution peripheral quantitative computed tomography. Osteoporos Int 31(9):1607–1627. https://doi.org/10.1007/s00198-020-05438-5
Pistoia W, van Rietbergen B, Lochmuller EM, Lill CA, Eckstein F, Ruegsegger P (2002) Estimation of distal radius failure load with micro-finite element analysis models based on three-dimensional peripheral quantitative computed tomography images. Bone 30(6):842–848
Arias-Moreno AJ, Hosseini HS, Bevers M, Ito K, Zysset P, van Rietbergen B (2019) Validation of distal radius failure load predictions by homogenized- and micro-finite element analyses based on second-generation high-resolution peripheral quantitative CT images. Osteoporos Int 30(7):1433–1443. https://doi.org/10.1007/s00198-019-04935-6
van den Bergh JP, Szulc P, Cheung AM, Bouxsein M, Engelke K, Chapurlat R (2021) The clinical application of high-resolution peripheral computed tomography (HR-pQCT) in adults: state of the art and future directions. Osteoporos Int. https://doi.org/10.1007/s00198-021-05999-z
Auerbach BM, Ruff CB (2006) Limb bone bilateral asymmetry: variability and commonality among modern humans. J Hum Evol 50(2):203–218. https://doi.org/10.1016/j.jhevol.2005.09.004
Peters M (1988) Footedness: asymmetries in foot preference and skill and neuropsychological assessment of foot movement. Psychol Bull 103(2):179–192. https://doi.org/10.1037/0033-2909.103.2.179
Warden SJ, Wright CS, Fuchs RK (2021) Bone microarchitecture and strength adaptation to physical activity: a within-subject controlled. HRpQCT study. Med Sci Sports Exerc 53(6):1179–1187. https://doi.org/10.1249/MSS.0000000000002571
Sode M, Burghardt AJ, Pialat JB, Link TM, Majumdar S (2011) Quantitative characterization of subject motion in HR-pQCT images of the distal radius and tibia. Bone 48(6):1291–1297. https://doi.org/10.1016/j.bone.2011.03.755
Cole TJ, Green PJ (1992) Smoothing reference centile curves: the LMS method and penalized likelihood. Stat Med 11(10):1305–1319. https://doi.org/10.1002/sim.4780111005
Rigby RA, Stasinopoulos DM (2005) Generalized additive models for location, scale and shape. J Roy Stat Soc Ser C (Appl Stat) 54(3):507–554. https://doi.org/10.1111/j.1467-9876.2005.00510.x
Centers for Disease Control and Prevention. Modified z-scores in the CDC growth charts. https://www.cdc.gov/nccdphp/dnpa/growthcharts/resources/biv-cutoffs.pdf. Accessed Mar 5 2021
Cole TJ (2021) Sample size and sample composition for constructing growth reference centiles. Stat Methods Med Res 30(2):488–507. https://doi.org/10.1177/0962280220958438
Whittier DE, Manske SL, Kiel DP, Bouxsein M, Boyd SK (2018) Harmonizing finite element modelling for non-invasive strength estimation by high-resolution peripheral quantitative computed tomography. J Biomech 80:63–71. https://doi.org/10.1016/j.jbiomech.2018.08.030
Kawalilak CE, Johnston JD, Olszynski WP, Kontulainen SA (2014) Characterizing microarchitectural changes at the distal radius and tibia in postmenopausal women using HR-pQCT. Osteoporos Int 25(8):2057–2066. https://doi.org/10.1007/s00198-014-2719-0
Zebaze RMD, Ghasem-Zadeh A, Bohte A, Iuliano-Burns S, Mirams M, Price RI et al (2010) Intracortical remodelling and porosity in the distal radius and post-mortem femurs of women: a cross-sectional study. Lancet 375(9727):1729–1736. https://doi.org/10.1016/S0140-6736(10)60320-0
Zebaze R, Seeman E (2015) Cortical bone: a challenging geography. J Bone Miner Res 30(1):24–29. https://doi.org/10.1002/jbmr.2419
Burt LA, Hanley DA, Boyd SK (2017) Cross-sectional versus longitudinal change in a prospective HR-pQCT study. J Bone Miner Res 32(7):1505–1513. https://doi.org/10.1002/jbmr.3129
Zebaze R, Ghasem-Zadeh A, Mbala A, Seeman E (2013) A new method of segmentation of compact-appearing, transitional and trabecular compartments and quantification of cortical porosity from high resolution peripheral quantitative computed tomographic images. Bone 54(1):8–20. https://doi.org/10.1016/j.bone.2013.01.007
Baumbach SF, Schmidt R, Varga P, Heinz T, Vécsei V, Zysset PK (2011) Where is the distal fracture line location of dorsally displaced distal radius fractures? J Orthop Res 29(4):489–494. https://doi.org/10.1002/jor.21268
Acknowledgements
The authors thank Patrick Johnson, Tyler Branford, and Steffi Farkas for assistance with participant recruitment, and scan acquisition and processing.
Funding
This contribution was made possible by support from the National Institutes of Health (NIH/NIAMS P30 AR072581) and the Indiana Clinical Translational Science Award/Institute (NCATS UL1TR002529-01).
Author information
Authors and Affiliations
Contributions
Study conceptualization, funding acquisition, and project administration: SJW and SMM. Methodology, investigation, and supervision: SJW, RKF and SMM. Resources and software: SJW, ZL and BvR. Data curation and formal analysis: SJW and ZL. Writing—original draft: SJW and RKF. Writing—review and editing: SJW, ZL, RKF, BvR, SMM.
Corresponding author
Ethics declarations
Ethics approval
Institutional Review Board approval was obtained from Indiana University.
Conflict of interest
Bert van Rietbergen is a consultant for Scanco Medical AG. Stuart Warden, Ziyue Liu, Robyn Fuchs, and Sharon Moe declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Warden, S.J., Liu, Z., Fuchs, R.K. et al. Reference data and calculators for second-generation HR-pQCT measures of the radius and tibia at anatomically standardized regions in White adults. Osteoporos Int 33, 791–806 (2022). https://doi.org/10.1007/s00198-021-06164-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-021-06164-2