Abstract
Summary
The Risk-stratified Osteoporosis Strategy Evaluation (ROSE) study investigated the effectiveness of a two-step screening program for osteoporosis in women. We found no overall reduction in fractures from systematic screening compared to the current case-finding strategy. The group of moderate- to high-risk women, who accepted the invitation to DXA, seemed to benefit from the program.
Introduction
The purpose of the ROSE study was to investigate the effectiveness of a two-step population-based osteoporosis screening program using the Fracture Risk Assessment Tool (FRAX) derived from a self-administered questionnaire to select women for DXA scan. After the scanning, standard osteoporosis management according to Danish national guidelines was followed.
Methods
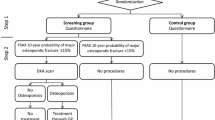
Participants were randomized to either screening or control group, and randomization was stratified according to age and area of residence. Inclusion took place from February 2010 to November 2011. Participants received a self-administered questionnaire, and women in the screening group with a FRAX score ≥ 15% (major osteoporotic fractures) were invited to a DXA scan. Primary outcome was incident clinical fractures. Intention-to-treat analysis and two per-protocol analyses were performed.
Results
A total of 3416 fractures were observed during a median follow-up of 5 years. No significant differences were found in the intention-to-treat analyses with 34,229 women included aged 65–80 years. The per-protocol analyses showed a risk reduction in the group that underwent DXA scanning compared to women in the control group with a FRAX ≥ 15%, in regard to major osteoporotic fractures, hip fractures, and all fractures. The risk reduction was most pronounced for hip fractures (adjusted SHR 0.741, p = 0.007).
Conclusions
Compared to an office-based case-finding strategy, the two-step systematic screening strategy had no overall effect on fracture incidence. The two-step strategy seemed, however, to be beneficial in the group of women who were identified by FRAX as moderate- or high-risk patients and complied with DXA.


Similar content being viewed by others
References
Johnell O, Kanis J (2005) Epidemiology of osteoporotic fractures. Osteoporos Int 16(Suppl 2):S3–S7. https://doi.org/10.1007/s00198-004-1702-6
Vestergaard P, Lindholm J, Jorgensen JO, Hagen C, Hoeck HC, Laurberg P et al (2002) Increased risk of osteoporotic fractures in patients with Cushing’s syndrome. Eur J Endocrinol 146(1):51–56
Brenneman SK, Barrett-Connor E, Sajjan S, Markson LE, Siris ES (2006) Impact of recent fracture on health-related quality of life in postmenopausal women. J Bone Miner Res 21(6):809–816. https://doi.org/10.1359/jbmr.060301
Viswanathan HN, Curtis JR, Yu J, White J, Stolshek BS, Merinar C, Balasubramanian A, Kallich JD, Adams JL, Wade SW (2012) Direct healthcare costs of osteoporosis-related fractures in managed care patients receiving pharmacological osteoporosis therapy. Appl Health Econ Health Policy 10(3):163–173. https://doi.org/10.2165/11598590-000000000-00000
Hansen L, Mathiesen AS, Vestergaard P, Ehlers LH, Petersen KDA (2013) Health economic analysis of osteoporotic fractures: who carries the burden? Arch Osteoporos 8(1-2):126. https://doi.org/10.1007/s11657-013-0126-3
Vestergaard P, Rejnmark L, Mosekilde L (2005) Osteoporosis is markedly underdiagnosed: a nationwide study from Denmark. Osteoporos Int 16(2):134–141. https://doi.org/10.1007/s00198-004-1680-8
Rubin KH, Abrahamsen B, Hermann AP, Bech M, Gram J, Brixen K (2011) Prevalence of risk factors for fractures and use of DXA scanning in Danish women. A regional population-based study. Osteoporos Int 22(5):1401–1409. https://doi.org/10.1007/s00198-010-1348-5
Wells GA, Cranney A, Peterson J, Boucher M, Shea B, Robinson V et al (2008) Alendronate for the primary and secondary prevention of osteoporotic fractures in postmenopausal women. Cochrane Database Syst Rev 1:CD001155
Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, Christiansen C, Delmas PD, Zanchetta JR, Stakkestad J, Glüer CC, Krueger K, Cohen FJ, Eckert S, Ensrud KE, Avioli LV, Lips P, Cummings SR (1999) Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. Multiple Outcomes of Raloxifene Evaluation (MORE) investigators. JAMA 282(7):637–645. https://doi.org/10.1001/jama.282.7.637
Meunier PJ, Roux C, Seeman E, Ortolani S, Badurski JE, Spector TD, Cannata J, Balogh A, Lemmel EM, Pors-Nielsen S, Rizzoli R, Genant HK, Reginster JY (2004) The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N Engl J Med 350(5):459–468. https://doi.org/10.1056/NEJMoa022436
Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, Cosman F, Lakatos P, Leung PC, Man Z, Mautalen C, Mesenbrink P, Hu H, Caminis J, Tong K, Rosario-Jansen T, Krasnow J, Hue TF, Sellmeyer D, Eriksen EF, Cummings SR, HORIZON Pivotal Fracture Trial (2007) Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med 356(18):1809–1822. https://doi.org/10.1056/NEJMoa067312
Cummings SR, San MJ, McClung MR, Siris ES, Eastell R, Reid IR et al (2009) Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med 361(8):756–765. https://doi.org/10.1056/NEJMoa0809493
Christensen PM, Brixen K, Gyrd-Hansen D, Kristiansen IS (2005) Cost-effectiveness of alendronate in the prevention of osteoporotic fractures in Danish women. Basic Clin Pharmacol Toxicol 96(5):387–396
Jonsson B, Strom O, Eisman JA, Papaioannou A, Siris ES, Tosteson A et al (2011) Cost-effectiveness of denosumab for the treatment of postmenopausal osteoporosis. Osteoporos Int 22(3):967–982. https://doi.org/10.1007/s00198-010-1424-x
Aboyoussef M, Vierkoetter KR (2007) Underdiagnosis and under-treatment of osteoporosis following fragility fracture. Hawaii Med J 66(7):185–187
Gardner MJ, Flik KR, Mooar P, Lane JM (2002) Improvement in the undertreatment of osteoporosis following hip fracture. J Bone Joint Surg Am 84-A(8):1342–1348
Kling JM, Clarke BL, Sandhu NP (2014) Osteoporosis prevention, screening, and treatment: a review. J Women’s Health (2002) 23(7):563–572. https://doi.org/10.1089/jwh.2013.4611
US Preventive Services Task Force Guides to Clinical Preventive Services (2012) The Guide to Clinical Preventive Services 2012: Recommendations of the US Preventive Services Task Force. Agency for Healthcare Research and Quality (US), Rockville
Cosman F, de Beur SJ, LeBoff MS, Lewiecki EM, Tanner B, Randall S, Lindsay R, National Osteoporosis Foundation (2014) Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 25(10):2359–2381. https://doi.org/10.1007/s00198-014-2794-2
Rubin KH, Holmberg T, Rothmann MJ, Hoiberg M, Barkmann R, Gram J et al (2015) The risk-stratified osteoporosis strategy evaluation study (ROSE): a randomized prospective population-based study. Design and baseline characteristics. Calcif Tissue Int 96(2):167–179. https://doi.org/10.1007/s00223-014-9950-8
Rubin KH, Abrahamsen B, Hermann AP, Bech M, Gram J, Brixen K (2011) Fracture risk assessed by fracture risk assessment tool (FRAX) compared with fracture risk derived from population fracture rates. Scand J Public Health 39(3):312–318
ISCD. The International Society for Clinical Densitometry—2015 official positions—adults. https://iscd.app.box.com/v/op-iscd-2015-adult 2015
Andersen TF, Madsen M, Jorgensen J, Mellemkjoer L, Olsen JH (1999) The Danish National Hospital Discharge Register. A valuable source of data for modern health. Science:263–268
Charlson ME, Pompei P, Ales KL, MacKenzie CRA (1987) New method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383. https://doi.org/10.1016/0021-9681(87)90171-8
Thygesen SK, Christiansen CF, Christensen S, Lash TL, Sorensen HT (2011) The predictive value of ICD-10 diagnostic coding used to assess Charlson comorbidity index conditions in the population-based Danish National Registry of patients. BMC Med Res Methodol 11(1):83. https://doi.org/10.1186/1471-2288-11-83
Dansk Knoglemedicinsk S, Danish Bonemedical S (2012) Vejledning til udredning og behandling of Osteoporose. [Instructions to diagnosis and treatment of osteoporosis]
Rothmann MJ, Moller S, Holmberg T, Hojberg M, Gram J, Bech M et al (2017) Non-participation in systematic screening for osteoporosis—the ROSE trial. Osteoporos Int 28(12):3389–3399. https://doi.org/10.1007/s00198-017-4205-y
National Osteoporosis F (2013) Clinician’s guide to prevention and treatment of osteoporosis. National Osteoporosis Foundation, Washington DC
World Health O (2017) WHO Fractures risk assessment tool; http://www.shef.ac.uk/FRAX/index.aspx
Shepstone L, Fordham R, Lenaghan E, Harvey I, Cooper C, Gittoes N, Heawood A, Peters TJ, O’Neill T, Torgerson D, Holland R, Howe A, Marshall T, Kanis JA, McCloskey E (2012) A pragmatic randomised controlled trial of the effectiveness and cost-effectiveness of screening older women for the prevention of fractures: rationale, design and methods for the SCOOP study. Osteoporos Int. 23(10):2507–2515. https://doi.org/10.1007/s00198-011-1876-7
McCloskey EV, Lenaghan E, Clarke S, Fordham R, Gittoes N, Harvey I et al (2016) Screening based on FRAX fracture risk assessment reduces the incidence of hip fractures in older community-dwelling women—results from the SCOOP study in the UK. ASBMR: J Bone Miner Res 31(Suppl 1):2016
Barr RJ, Stewart A, Torgerson DJ, Seymour DG, Reid DM (2005) Screening elderly women for risk of future fractures—participation rates and impact on incidence of falls and fractures. Calcif Tissue Int 76(4):243–248
Lacroix AZ, Buist DS, Brenneman SK, Abbott TA III (2005) Evaluation of three population-based strategies for fracture prevention: results of the osteoporosis population-based risk assessment (OPRA) trial. Med Care 43(3):293–302
Dargent-Molina P, Piault S, Breart G (2003) A comparison of different screening strategies to identify elderly women at high risk of hip fracture: results from the EPIDOS prospective study. Osteoporos Int. 14(12):969–977. https://doi.org/10.1007/s00198-003-1506-0
Schneider DL, Worley K, Beard MK, Iannini M, Ko M, McCallum J et al (2010) The primary care osteoporosis risk of fracture screening (POROS) study: design and baseline characteristics. Contemp Clin Trials 31(4):336–344
Mosbech J, Jorgensen J, Madsen M, Rostgaard K, Thornberg K, Poulsen TD (1995) The national patient registry. Evaluation of data quality. UgeskrLaeger 157(26):3741–3745
Thygesen LC, Daasnes C, Thaulow I, Bronnum-Hansen H (2011) Introduction to Danish (nationwide) registers on health and social issues: structure, access, legislation, and archiving. Scand J Public Health 39(7 Suppl):12–16
Vestergaard P, Mosekilde L (2002) Fracture risk in patients with celiac disease, Crohn’s disease, and ulcerative colitis: a nationwide follow-up study of 16,416 patients in Denmark. Am J Epidemiol 156(1):1–10. https://doi.org/10.1093/aje/kwf007
Acknowledgements
We thank the participants in the ROSE study and the technical staff in the four involved hospitals: Odense University Hospital, Odense and Nyborg; Hospital of Southwest Denmark, Esbjerg; and Sygehus Lillebælt Hospital, Kolding, Denmark. Claire Gudex is thanked for linguistic editing and proofreading of the manuscript.
Funding
The ROSE study was supported by INTERREG (4A JNR 08/4177), the Region of Southern Denmark (JNR 08/8133), and Odense University Hospital (JNR 11/5761). The funding agencies had no direct role in the conduct of the study, data collection, analysis, and interpretation of the data or the preparation, review, and final approval of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
SM, TH, MB, JG, RB, CCG, and KHR have no conflict of interest. MJR has received speaker fee from Eli Lilly. MH is a full-time employee of Boehringer-Ingelheim Norway KS (currently). APH serves on advisory boards for Eli Lilly and Amgen, and she has received research funding from Eli Lilly, speaker fee from Eli Lilly, GSK, Genzyme, and Amgen; KB reports other from Merck, Sharpe, and Dohme, other from Amgen, other from Novartis, and other from NPS, all outside the submitted work.
Rights and permissions
About this article
Cite this article
Rubin, K.H., Rothmann, M.J., Holmberg, T. et al. Effectiveness of a two-step population-based osteoporosis screening program using FRAX: the randomized Risk-stratified Osteoporosis Strategy Evaluation (ROSE) study. Osteoporos Int 29, 567–578 (2018). https://doi.org/10.1007/s00198-017-4326-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-017-4326-3