Abstract
Summary
More than 45 % of long-term childhood cancer survivors (CCS) were diagnosed with osteopenia. Our data suggest that greater awareness for osteopenia is warranted in long-term CCS, especially in survivors who are older than 30 years, male, and underweight and were treated with cranial-spinal radiotherapy and/or steroids.
Introduction
Osteopenia is a potential complication of childhood cancer treatment, but the magnitude of this problem in survivors is unknown. We examined (determinants of) bone mineral density (BMD) status in long-term survivors of adult childhood cancer.
Methods
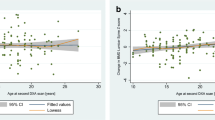
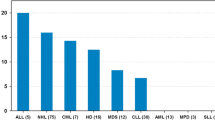
This retrospective single-centre cohort study included 346 subjects with the most common types of childhood cancer. Subjects had a median age at diagnosis of 7.0 years (range 0.1–16.8 years), a median age at follow-up of 24.5 years (range 18.0–47.6 years) and a median follow-up time of 16.7 years (range 5.6–39.9 years). Total body BMD (BMDTB) and BMD of the lumbar spine (BMDLS) were measured by dual X-ray absorptiometry. Osteopenia was defined as BMD standardized deviation score (SDS) below −1.
Results
Survivors had a lower BMDTB and BMDLS (mean SDS −0.55; p < 0.001 and −0.30; p < 0.001, respectively) as compared to healthy peers. Osteopenia (BMDTB and/or BMDLS) was present in 45 % of the survivors. Multivariate logistic regression analyses identified age at diagnosis <12 years, age >30 years at follow-up, male gender, underweight at follow-up and treatment with cranial-spinal radiotherapy or prednisone as independent prognostic factors for osteopenia.
Conclusions
This large cohort of childhood cancer survivors identified osteopenia in 45 % of CCS. This indicates that greater awareness is warranted, especially in survivors who are older than 30 years, male, have underweight and were treated with cranial-spinal radiotherapy and/or steroids.

Similar content being viewed by others
References
Gatta G, Botta L, Rossi S, Aareleid T, Bielska-Lasota M, Clavel J, Dimitrova N, Jakab Z, Kaatsch P, Lacour B, Mallone s, Marcos-Gragera R, Minicozzi P, Sanchez-Perez MJ, Sant M, Santaquilani M, Stiller C, Tavilla A, Trama A, Peris-Bonet R, Group EW (2014) Childhood cancer survival in Europe 1999-2007: results of EUROCARE-5—a population-based study. Lancet Oncol 15(1):35–47. doi:10.1016/S1470-2045(13)70548-5
Chemaitilly W, Sklar CA (2010) Endocrine complications in long-term survivors of childhood cancers. Endocr Relat Cancer 17(20453080):141–159
Lie Fong S, Lugtenburg PJ, Schipper I, Themmen APN, de Jong FH, Sonneveld P, Laven JSE (2008) Anti-mullerian hormone as a marker of ovarian function in women after chemotherapy and radiotherapy for haematological malignancies. Hum Reprod 23(18216040):674–678
van Beek RD, van den Heuvel-Eibrink MM, Hakvoort-Cammel FG, van den Bos C, van der Pal HJ, Krenning EP, de Rijke YB, Pieters R, de Muinck Keizer-Schrama SM (2009) Bone mineral density, growth, and thyroid function in long-term survivors of pediatric Hodgkin’s lymphoma treated with chemotherapy only. J Clin Endocrinol Metab 94(6):1904–1909. doi:10.1210/jc.2008-0622
van Beek RD, van den Heuvel-Eibrink MM, Laven JS, de Jong FH, Themmen AP, Hakvoort-Cammel FG, van den Bos C, van den Berg H, Pieters R, de Muinck Keizer-Schrama SM (2007) Anti-Mullerian hormone is a sensitive serum marker for gonadal function in women treated for Hodgkin’s lymphoma during childhood. J Clin Endocrinol Metab 92(10):3869–3874. doi:10.1210/jc.2006-2374
van Casteren NJ, van der Linden GH, Hakvoort-Cammel FG, Hahlen K, Dohle GR, van den Heuvel-Eibrink MM (2009) Effect of childhood cancer treatment on fertility markers in adult male long-term survivors. Pediatr Blood Cancer 52(1):108–112. doi:10.1002/pbc.21780
van Waas M, Neggers SJCMM, Pieters R, van den Heuvel-Eibrink MM (2010) Components of the metabolic syndrome in 500 adult long-term survivors of childhood cancer. Ann Oncol 21(19850641):1121–1126
Karaman S, Ercan O, Yildiz I, Bolayirli M, Celkan T, Apak H, Ozkan A, Onal H, Canbolat A (2010) Late effects of childhood ALL treatment on body mass index and serum leptin levels. J Pediatr Endocrinol Metab 23(20857839):669–674
te Winkel ML, Pieters R, Hop WC, Roos JC, Bokkerink JP, Leeuw JA, Bruin MC, Kollen WJ, Veerman AJ, de Groot-Kruseman HA, van der Sluis IM, van den Heuvel-Eibrink MM (2014) Bone mineral density at diagnosis determines fracture rate in children with acute lymphoblastic leukemia treated according to the DCOG-ALL9 protocol. Bone 59:223–228. doi:10.1016/j.bone.2013.11.017
Wilson CL, Ness KK (2013) Bone mineral density deficits and fractures in survivors of childhood cancer. Curr Osteoporos Rep 11(4):329–337. doi:10.1007/s11914-013-0165-0
Ruble K, Hayat MJ, Stewart KJ, Chen AR (2010) Bone mineral density after bone marrow transplantation in childhood: measurement and associations. Biol Blood Marrow Transplant 16(10):1451–1457. doi:10.1016/j.bbmt.2010.04.010
Le Meignen M, Auquier P, Barlogis V, Sirvent N, Contet A, Simeoni MC, Galambrun C, Poiree M, Chastagner P, Play B, Villes V, Berbis J, Chambost H, Bordigoni P, Michel G (2011) Bone mineral density in adult survivors of childhood acute leukemia: impact of hematopoietic stem cell transplantation and other treatment modalities. Blood 118(6):1481–1489. doi:10.1182/blood-2011-01-332866
Choi YJ, Park SY, Cho WK, Lee JW, Cho KS, Park SH, Hahn SH, Jung MH, Chung NG, Cho B, Suh BK, Kim HK (2013) Factors related to decreased bone mineral density in childhood cancer survivors. J Korean Med Sci 28(11):1632–1638. doi:10.3346/jkms.2013.28.11.1632
Benmiloud S, Steffens M, Beauloye V, de Wandeleer A, Devogelaer J-P, Brichard B, Vermylen C, Maiter D (2010) Long-term effects on bone mineral density of different therapeutic schemes for acute lymphoblastic leukemia or non-Hodgkin lymphoma during childhood. Horm Res Paediatr 74(20395671):241–250
Mandel K, Atkinson S, Barr RD, Pencharz P (2004) Skeletal morbidity in childhood acute lymphoblastic leukemia. J Clin Oncol 22(15051768):1215–1221
Kaste SC, Jones-Wallace D, Rose SR, Boyett JM, Lustig RH, Rivera GK, Pui CH, Hudson MM (2001) Bone mineral decrements in survivors of childhood acute lymphoblastic leukemia: frequency of occurrence and risk factors for their development. Leukemia 15(5):728–734
Gurney JG, Kaste SC, Liu W, Srivastava DK, Chemaitilly W, Ness KK, Lanctot JQ, Ojha RP, Nottage KA, Wilson CL, Li Z, Robison LL, Hudson MM (2014) Bone mineral density among long-term survivors of childhood acute lymphoblastic leukemia: results from the St. Jude Lifetime Cohort Study. Pediatr Blood Cancer. doi:10.1002/pbc.25010
Makitie O, Heikkinen R, Toiviainen-Salo S, Henriksson M, Puukko-Viertomies LR, Jahnukainen K (2013) Long-term skeletal consequences of childhood acute lymphoblastic leukemia in adult males: a cohort study. Eur J Endocrinol 168(2):281–288. doi:10.1530/eje-12-0702
Lim JS, Kim DH, Lee JA, Cho J, Cho WH, Lee SY, Jeon DG (2013) Young age at diagnosis, male sex, and decreased lean mass are risk factors of osteoporosis in long-term survivors of osteosarcoma. J Pediatr Hematol Oncol 35(1):54–60. doi:10.1097/MPH.0b013e318275193b
Joyce ED, Nolan VG, Ness KK, Ferry RJ Jr, Robison LL, Pui CH, Hudson MM, Kaste SC (2011) Association of muscle strength and bone mineral density in adult survivors of childhood acute lymphoblastic leukemia. Arch Phys Med Rehabil 92(6):873–879. doi:10.1016/j.apmr.2010.12.039
Polgreen LE, Petryk A, Dietz AC, Sinaiko AR, Leisenring W, Goodman P, Steffen LM, Perkins JL, Dengel DR, Baker KS, Steinberger J (2012) Modifiable risk factors associated with bone deficits in childhood cancer survivors. BMC Pediatr 12:40. doi:10.1186/1471-2431-12-40
Assembly WMAG (2008) The Declaration of Helsinki—Sixth Revision. Helsinki, Finland
de Vet A, Laven JSE, de Jong FH, Themmen APN, Fauser BCJM (2002) Antimullerian hormone serum levels: a putative marker for ovarian aging. Fertil Steril 77(11821097):357–362
Crabtree NJ, Shaw NJ, Boivin CM, Oldroyd B, Truscott JG (2005) Pediatric in vivo cross-calibration between the GE Lunar Prodigy and DPX-L bone densitometers. Osteoporos Int: J Established Result Cooperation Between Eur Found Osteoporos Natl Osteoporos Found USA 16(12):2157–2167. doi:10.1007/s00198-005-2021-2
Oldroyd B, Smith AH, Truscott JG (2003) Cross-calibration of GE/Lunar pencil and fan-beam dual energy densitometers—bone mineral density and body composition studies. Eur J Clin Nutr 57(8):977–987. doi:10.1038/sj.ejcn.1601633
Boot AM, de Ridder MAJ, van der Sluis IM, van Slobbe I, Krenning EP, Keizer-Schrama SMPFM (2010) Peak bone mineral density, lean body mass and fractures. Bone 46(19833245):336–341
Lewiecki EM, Gordon CM, Baim S, Binkley N, Bilezikian JP, Kendler DL, Hans DB, Silverman S, Bishop NJ, Leonard MB, Bianchi ML, Kalkwarf HJ, Langman CB, Plotkin H, Rauch F, Zemel BS (2008) Special report on the 2007 adult and pediatric Position Development Conferences of the International Society for Clinical Densitometry. Osteoporos Int: J Established Result Cooperation Between Eur Found Osteoporos Natl Osteoporos Found USA 19(10):1369–1378. doi:10.1007/s00198-008-0689-9
Nysom K, Holm K, Michaelsen KF, Hertz H, Muller J, Molgaard C (2001) Bone mass after treatment of malignant lymphoma in childhood. Med Pediatr Oncol 37(6):518–524
Holzer G, Krepler P, Koschat MA, Grampp S, Dominkus M, Kotz R (2003) Bone mineral density in long-term survivors of highly malignant osteosarcoma. J Bone Joint Surg (Br) 85(12678358):231–237
Kaste SC, Ahn H, Liu T, Liu W, Krasin MJ, Hudson MM, Spunt SL (2008) Bone mineral density deficits in pediatric patients treated for sarcoma. Pediatr Blood Cancer 50(5):1032–1038. doi:10.1002/pbc.21281
Kang MJ, Kim SM, Lee YA, Shin CH, Yang SW, Lim JS (2012) Risk factors for osteoporosis in long-term survivors of intracranial germ cell tumors. Osteoporos Int: J Established Result Cooperation Between Eur Found Osteoporos Natl Osteoporos Found USA 23(7):1921–1929. doi:10.1007/s00198-011-1821-9
Nysom K, Holm K, Michaelsen KF, Hertz H, Jacobsen N, Muller J, Molgaard C (2000) Bone mass after allogeneic BMT for childhood leukaemia or lymphoma. Bone Marrow Transplant 25(2):191–196. doi:10.1038/sj.bmt.1702131
Ho PT, Zimmerman K, Wexler LH, Blaney S, Jarosinski P, Weaver-McClure L, Izraeli S, Balis FM (1995) A prospective evaluation of ifosfamide-related nephrotoxicity in children and young adults. Cancer 76(12):2557–2564
Muller HL, Schneider P, Bueb K, Etavard-Gorris N, Gebhardt U, Kolb R, Sorensen N (2003) Volumetric bone mineral density in patients with childhood craniopharyngioma. Exp Clin Endocrinol Diabetes 111(3):168–173. doi:10.1055/s-2003-39789
Watsky MA, Carbone LD, An Q, Cheng C, Lovorn EA, Hudson MM, Pui CH, Kaste SC (2014) Bone turnover in long-term survivors of childhood acute lymphoblastic leukemia. Pediatr Blood Cancer. doi:10.1002/pbc.25025
Kanis JA (2002) Diagnosis of osteoporosis and assessment of fracture risk. Lancet 359(9321):1929–1936. doi:10.1016/S0140-6736(02)08761-5
Kaste SC, Rai SN, Fleming K, McCammon EA, Tylavsky FA, Danish RK, Rose SR, Sitter CD, Pui C-H, Hudson MM (2006) Changes in bone mineral density in survivors of childhood acute lymphoblastic leukemia. Pediatr Blood Cancer 46(16106430):77–87
Bours SP, van Geel TA, Geusens PP, Janssen MJ, Janzing HM, Hoffland GA, Willems PC, van den Bergh JP (2011) Contributors to secondary osteoporosis and metabolic bone diseases in patients presenting with a clinical fracture. J Clin Endocrinol Metab 96(5):1360–1367. doi:10.1210/jc.2010-2135
Frank GR (2003) Role of estrogen and androgen in pubertal skeletal physiology. Med Pediatr Oncol 41(3):217–221. doi:10.1002/mpo.10340
Sala A, Talsma D, Webber C, Posgate S, Atkinson S, Barr R (2007) Bone mineral status after treatment of malignant lymphoma in childhood and adolescence. Eur J Cancer Care 16(4):373–379. doi:10.1111/j.1365-2354.2006.00757.x
van Leeuwen BL, Kamps WA, Jansen HW, Hoekstra HJ (2000) The effect of chemotherapy on the growing skeleton. Cancer Treat Rev 26(5):363–376. doi:10.1053/ctrv.2000.0180
Ferrari S, Bianchi ML, Eisman JA, Foldes AJ, Adami S, Wahl DA, Stepan JJ, de Vernejoul MC, Kaufman JM, Pathophysiology IOFCoSAWGoO (2012) Osteoporosis in young adults: pathophysiology, diagnosis, and management. Osteoporos Int: J Established Result Cooperation Between Eur Found Osteoporos Natl Osteoporos Found USA 23(12):2735–2748. doi:10.1007/s00198-012-2030-x
Suh E, Daugherty CK, Wroblewski K, Lee H, Kigin ML, Rasinski KA, Ford JS, Tonorezos ES, Nathan PC, Oeffinger KC, Henderson TO (2014) General internists’ preferences and knowledge about the care of adult survivors of childhood cancer: a cross-sectional survey. Ann Intern Med 160(1):11–17
Children’s Oncology Group COG (2014) Version 3.0 of long-term follow-up guidelines http://www.survivorshipguidelines.org/. Accessed 5 Jun 2014
Stichting Kinderoncologie Nederland SKION (2010) Richtlijn follow-up na kinderkanker. https://www.skion.nl/voor-patienten-en-ouders/late-effecten/533/richtlijn-follow-up-na-kinderkanker/. Accessed 5 Jun 2014
SIGN (2014) SIGN 132: Long term follow up of survivors of childhood cancer. http://www.sign.ac.uk/guidelines/fulltext/132/index.html. Accessed 5 Jun 2014
Acknowledgments
MvW, MtW and SN were supported by grants from the Foundation Kinderenkankervrij (KIKA) and WvD is supported by the Paediatric Oncology Centre Society for Research (KOCR), Rotterdam, The Netherlands.
Ethical standards
This study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All persons gave their informed consent prior to their inclusion in the study.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
M.A.H. den Hoed and B.C. Klap shared first authorship and S.M.F. Pluijm and M.M. van den Heuvel-Eibrink shared last authorship.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental table 1
Body height and bone mineral density in 346 adult long term childhood cancer survivors (DOCX 37 kb)
Supplemental table 2
Bone mineral density adjusted for height SDS in 298 adult long term childhood cancer survivors (DOCX 36 kb)
Rights and permissions
About this article
Cite this article
den Hoed, M.A.H., Klap, B.C., te Winkel, M.L. et al. Bone mineral density after childhood cancer in 346 long-term adult survivors of childhood cancer. Osteoporos Int 26, 521–529 (2015). https://doi.org/10.1007/s00198-014-2878-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-014-2878-z