Abstract
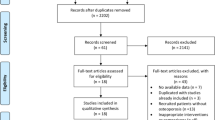
Current treatments for osteoporosis are limited by lack of effect on cortical bone, side effects, and, in some cases, cost. Organic nitrates, which act as nitric oxide donors, may be a potential alternative. This systematic review summarizes the clinical data that reports on the effects of organic nitrates and bone. Organic nitrates, which act as nitric oxide donors, are novel agents that have several advantages over the currently available treatments for osteoporosis. This systematic review summarizes the clinical data that reports on the effects of organic nitrates on bone. We searched Medline (1966 to November 2012), EMBASE (1980 to November 2012), and the Cochrane Central Register of Controlled Trials (Issue 11, 2012). Keywords included nitrates, osteoporosis, bone mineral density (BMD), and fractures. We identified 200 citations. Of these, a total of 29 were retrieved for more detailed evaluation and we excluded 19 manuscripts: 15 because they did not present original data and four because they did not provide data on the intervention or outcome of interest. As such, we included ten studies in literature review. Of these ten studies two were observational cohort studies reporting nitrate use was associated with increased BMD; two were case control studies reporting that use of nitrates were associated with lower risk of hip fracture; two were randomized controlled trials (RCT) comparing alendronate to organic nitrates for treatment of postmenopausal women and demonstrating that both agents increased lumbar spine BMD. The two largest RCT with the longest follow-up, both of which compared effects of organic nitrates to placebo on BMD in women without osteoporosis, reported conflicting results. Headaches were the most common adverse event among women taking nitrates. No studies have reported on fracture efficacy. Further research is needed before recommending organic nitrates for the treatment of postmenopausal osteoporosis.

Similar content being viewed by others
References
Melton LJI, Cooper C (2001) Magnitude and impact of osteoporosis and fractures. In: Marcus R, Feldman D, Kelsey J (eds) Osteoporosis. Academic Press, San Diego, pp 557–567
Gullberg B, Johnell O, Kanis JA (1997) World-wide projections for hip fracture. Osteoporos Int 7:407–413
Cummings SR (2006) A 55-year-old woman with osteopenia. JAMA 296:2601–2610
Black DM, Cummings SR, Karpf DB, Cauley JA, Thompson DE, Nevitt MC, Bauer DC, Genant HK, Haskell WL, Marcus R, Ott SM, Torner JC, Quandt SA, Reiss TF, Ensrud KE (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 348:1535–1541
Cummings SR, Black DM, Thompson DE, Applegate WB, Barrett-Connor E, Musliner TA, Palermo L, Prineas R, Rubin SM, Scott JC, Vogt T, Wallace R, Yates AJ, LaCroix AZ (1998) Effect of alendronate on risk of fracture in women with low bone density but without vertebral fractures: results from the Fracture Intervention Trial. JAMA 280:2077–2082
Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, Chesnut CH 3rd, Brown J, Eriksen EF, Hoseyni MS, Axelrod DW, Miller PD (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. JAMA 282:1344–1352
Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, Cosman F, Lakatos P, Leung PC, Man Z, Mautalen C, Mesenbrink P, Hu H, Caminis J, Tong K, Rosario-Jansen T, Krasnow J, Hue TF, Sellmeyer D, Eriksen EF, Cummings SR (2007) Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med 356:1809–1822
Cummings SR, San Martin J, McClung MR, Siris ES, Eastell R, Reid IR, Delmas P, Zoog HB, Austin M, Wang A, Kutilek S, Adami S, Zanchetta J, Libanati C, Siddhanti S, Christiansen C (2009) Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med 361:756–765
Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, Christiansen C, Delmas PD, Zanchetta JR, Stakkestad J, Gluer CC, Krueger K, Cohen FJ, Eckert S, Ensrud KE, Avioli LV, Lips P, Cummings SR (1999) Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. Multiple Outcomes of Raloxifene Evaluation (MORE) Investigators. JAMA 282:637–645
Neer RM, Arnaud CD, Zanchetta JR et al (2001) Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 344:1434–1441
Cummings SR, Palermo L, Browner W, Marcus R, Wallace R, Pearson J, Eckert S, Black D (2000) Monitoring osteoporosis therapy with bone densitometry: misleading changes and regression to the mean. Fracture Intervention Trial Research Group. JAMA 8:1318–1321
Cryer R, Bauer DC (2002) Oral bisphosphonates and upper gastrointestinal tract problems: what is the evidence? Mayo Clin Proc 77:1031–1043
Bauer DC, Black D, Ensrud K, Thompson D, Hochberg M, Nevitt M, Musliner T, Freedholm D (2000) Upper gastrointestinal tract safety profile of alendronate: the fracture intervention trial. Arch Intern Med 160:517–525
Jamal SA, Bauer DC, Ensrud KE, Cauley JA, Hochberg M, Ishani A, Cummings SR (2007) Alendronate treatment in women with normal to severely impaired renal function: an analysis of the fracture intervention trial. J Bone Miner Res 22:503–508
Shane E, Burr D, Ebeling PR, Abrahamsen B, Adler RA, Brown TD, Cheung AM, Cosman F, Curtis JR, Dell R, Dempster D, Einhorn TA, Genant HK, Geusens P, Klaushofer K, Koval K, Lane JM, McKiernan F, McKinney R, Ng A, Nieves J, O'Keefe R, Papapoulos S, Sen HT, van der Meulen MC, Weinstein RS, Whyte M (2010) Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 25:2267–2294.
Women's Health Initiative (2002) Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women's Health Initiative randomized controlled trial. JAMA 288:321–333.
Cummings SR, Cosman F, Jamal SA (eds) (2002) Osteoporosis. An evidenced-based guide to prevention and management. American College of Physicians, Philadelphia
Cummings SR, Eckert S, Krueger KA, Grady D, Powles TJ, Cauley JA, Norton L, Nickelsen T, Bjarnason NH, Morrow M, Lippman ME, Black D, Glusman JE, Costa A, Jordan VC (1999) The effect of raloxifene on risk of breast cancer in postmenopausal women: results from the MORE randomized trial. Multiple outcomes of raloxifene evaluation. JAMA 281:2189–2197
Das-Gupta V, Williamson RA, Pitsillides AA (2012) Expression of endothelial nitric oxide synthase protein is not necessary for mechanical strain-induced nitric oxide production by cultured osteoblasts. Osteoporos Int 23:2635–2647.
Zheng H, Yu X, Collin-Osdoby P, Osdoby P (2006) RANKL stimulates inducible nitric-oxide synthase expression and nitric oxide production in developing osteoclasts. An autocrine negative feedback mechanism triggered by RANKL-induced interferon-beta via NF-kappaB that restrains osteoclastogenesis and bone resorption. J Biol Chem 281:15809–15820
MacIntyre I, Zaidi M, Alam AS, Datta HK, Moonga BS, Lidbury PS, Hecker M, Vane JR (1991) Osteoclastic inhibition: an action of nitric oxide not mediated by cyclic GMP. Proc Natl Acad Sci U S A 88:2936–2940
Lowik CW, Nibbering PH, van de Ruit M, Papapoulos SE (1994) Inducible production of nitric oxide in osteoblast-like cells and in fetal mouse bone explants is associated with suppression of osteoclastic bone resorption. J Clin Investig 93:1465–1472
Collin-Osdoby P, Li L, Rothe L, Anderson F, Kirsch D, Oursler MJ, Osdoby P (1998) Inhibition of avian osteoclast bone resorption by monoclonal antibody 121 F: a mechanism involving the osteoclast free radical system. J Bone Miner Res 13:67–78
Ralston SH (1997) The Michael Mason Prize Essay (1997) Nitric oxide and bone: what a gas! Br J Rheumatol 36:831–838
Ralston SH, Ho LP, Helfrich MH, Grabowski PS, Johnston PW, Benjamin N (1995) Nitric oxide: a cytokine-induced regulator of bone resorption. J Bone Miner Res 10:1040–1049
Brandi ML, Hukkanen M, Umeda T, Moradi-Bidhendi N, Bianchi S, Gross SS et al (1995) Bidirectional regulation of osteoclast function by nitric oxide synthase isoforms. Proc Natl Acad Sci U S A 92:2954–2958
Chow JW, Fox SW, Lean JM, Chambers TJ (1998) Role of nitric oxide and prostaglandins in mechanically induced bone formation. J Bone Miner Res 13:1039–1044
Kasten TP, Collin-Osdoby P, Patel N, Osdoby P, Krukowski M, Misko TP, Settle SL, Currie MG, Nickols GA (1994) Potentiation of osteoclast bone-resorption activity by inhibition of nitric oxide synthase. Proc Natl Acad Sci U S A 91:3569–3573
Ake Y, Saegusa Y, Matsubara T, Mizuno K (1994) Cultured osteoblast synthesize nitric oxide in response to cytokines and lipopolysaccharide. Kobe J Med Sci 40:125–137
Grassi F, Fan X, Rahnert J, Weitzmann MN, Pacifici R, Nanes MS, Rubin J (2006) Bone re/modeling is more dynamic in the endothelial nitric oxide synthase(−/−) mouse. Endocrinology 147:4392–4399
Aguirre J, Buttery L, O'Shaughnessy M, Afzal F, Fernandez de Marticorena I, Hukkanen M, Huang P, MacIntyre I, Polak J (2001) Endothelial nitric oxide synthase gene-deficient mice demonstrate marked retardation in postnatal bone formation, reduced bone volume, and defects in osteoblast maturation and activity. Am J Pathol 158:247–257
Armour KE, Armour KJ, Gallagher ME, Godecke A, Helfrich MH, Reid DM, Ralston SH (2001) Defective bone formation and anabolic response to exogenous estrogen in mice with targeted disruption of endothelial nitric oxide synthase. Endocrinology 142:760–766
Wimalawansa SJ, De Marco G, Gangula P, Yallampalli C (1996) Nitric oxide donor alleviates ovariectomy-induced bone loss. Bone 18:301–304
Wimalawansa SJ, Chapa MT, Yallampalli C, Zhang R, Simmons DJ (1997) Prevention of corticosteroid-induced bone loss with nitric oxide donor nitroglycerin in male rats. Bone 21:275–280
Jamal SA, Browner WS, Bauer DC, Cummings SR (1998) Intermittent use of nitrates increases bone mineral density: the study of osteoporotic fractures. J Bone Miner Res 13:1755–1759
Abshagen A, Sporl-Radun S (1981) First data on effects and pharmacokinetics of isosorbide-5-mononitrate in normal man. Eur J Clin Pharmacol 19:423–429
Wimalawansa SJ, Chapa T, Wimalawansa S, Fang L, Yallampalli C (1997) Dose and frequency effects of nitric oxide donor nitroglycerine on bone. Seventy-ninth Annual Meeting of the Endocrine Society, Minneapolis, USA. (abst P3–248).
Cummings SR, Nevitt MC, Browner WS, Stone K, Fox KM, Ensrud KE, Cauley J, Black D, Vogt TM (1995) Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N Engl J Med 332:767–773
Jamal SA, Goltzman D, Hanley DA, Papaioannou A, Prior JC, Josse RG (2009) Nitrate use and changes in bone mineral density: the Canadian Multicentre Osteoporosis Study. Osteoporos Int 20:737–744
Rejnmark L, Vestergaard P, Mosekilde L (2006) Decreased fracture risk in users of organic nitrates: a nationwide case–control study. J Bone Miner Res 21:1811–1817
Pouwels S, Lalmohamed A, van Staa T, Cooper C, Souverein P, Leufkens HG, Rejnmark L, de Boer A, Vestergaard P, de Vries F (2010) Use of organic nitrates and the risk of hip fracture: a population-based case–control study. The Journal of Clinical Endocrinology and Metabolism 95:1924–1931.
Jamal SA, Cummings SR, Hawker GA (2004) The effects of isosorbide mononitrate on bone turnover: a randomized controlled trial. J Bone Miner Res 19:1512–1517
Hochberg MC, Greenspan S, Wasnich RD, Miller P, Thompson DE, Ross PD (2002) Changes in bone density and turnover explain the reductions in incidence of nonvertebral fractures that occur during treatment with antiresorptive agents. J Clin Endocrinol Metab 87:1586–1592
Looker AC, Bauer DC, Chesnut CH 3rd, Gundberg CM, Hochberg MC, Klee G, Kleerekoper M, Watts NB, Bell NH (2000) Clinical use of biochemical markers of bone remodeling: current status and future directions. Osteoporos Int 11:467–480
Wimalawansa SJ (2000) Nitroglycerin therapy is as efficacious as standard estrogen replacement therapy (Premarin) in prevention of oophorectomy-induced bone loss: a human pilot clinical study. J Bone Miner Res 15:2240–2244
Jamal SA, Hamilton CJ, Eastell R, Cummings SR (2011) Effect of nitroglycerin ointment on bone density and strength in postmenopausal women: a randomized trial. JAMA 305:800–807.
Wimalawansa SJ, Grimes JP, Wilson AC, Hoover DR (2009) Transdermal nitroglycerin therapy may not prevent early postmenopausal bone loss. J Clin Endocrinol Metab 94:3356–3364
Duhan N, Siwach RC, Yadav K, Dahiya K, Nanda S, Sirohiwal D (2010) Comparative evaluation of isosorbide mononitrate and alendronate in management of postmenopausal osteoporosis. Archives of Gynecology and Obstetrics 285:1019–1023.
Nabhan AF, Rabie NH (2008) Isosorbide mononitrate versus alendronate for postmenopausal osteoporosis. Int J Gynaecol Obstet 103:213–216
Li X, Zhang Y, Kang H, Liu W, Liu P, Zhang J, Harris SE, Wu D (2005) Sclerostin binds to LRP5/6 and antagonizes canonical Wnt signaling. J Biol Chem 280:19883–19887
Balemans W, Ebeling M, Patel N, Van Hul E, Olson P, Dioszegi M, Lacza C, Wuyts W, Van Den Ende J, Willems P, Paes-Alves AF, Hill S, Bueno M, Ramos FJ, Tacconi P, Dikkers FG, Stratakis C, Lindpaintner K, Vickery B, Foernzler D, Van Hul W (2001) Increased bone density in sclerosteosis is due to the deficiency of a novel secreted protein (SOST). Hum Mol Genet 10:537–543
Arasu A, Hamilton CJ, Cummings S, Eastell R, Jamal SA (2012) Nitroglycerin May Improve Bone Mass by Suppressing Sclerostin Levels. In The Endocrine Society Annual Meeting. Endocrine Reviews
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
A comment to this article is available at http://dx.doi.org/10.1007/s00198-016-3852-8.
Appendix 1 search strategy
Appendix 1 search strategy
-
a.
Medline (PubMed) (1966 to November 2012)
(Nitrates[53] OR nitrate[tiab] OR nitrates[tiab] OR Nitroglycerin[53] OR nitroglycerin[tiab] OR Glyceryl-Trinitrate[tiab] OR transderm-nitro[tiab] OR nitroderm[tiab] OR Nitric Oxide Donors[53] OR Nitric Oxide[53] OR Isosorbide Dinitrate[53] OR Isosorbide[53] OR oxide-donor[tiab] OR isosorbide-dinitrate[tiab] OR isosorbide[tiab] OR dinitrate[tiab] OR monoxide[tiab] OR mononitrogen[tiab] OR endogenous-nitrate[tiab] OR ((Vasodilator Agents[53] OR vasodilator[tiab] OR vasodilators[tiab] OR vasorelaxant[tiab] OR vasodilatator[tiab] OR Organic Chemicals[53] OR organic[tiab]) AND (nitrate[tiab] OR nitrates[tiab] OR nitric[tiab])) OR nitrogen-monoxide[all fields] OR mononitrate[tiab] OR organic-nitrate[tiab] OR organic-salt[tiab]) AND (osteoporosis[all fields] OR osteopenia[tiab] OR osteoporotic[tiab] OR Bone Diseases, Metabolic[53] OR Bone Density/drug effects[53] OR Bone Resorption[53] OR Bone Density Conservation Agents[53] OR bone-loss[tiab] OR bone-resorption[tiab] OR osteolysis[tiab])
(Therapy/Broad[filter]) AND ((Nitrates[53] OR nitrate[tiab] OR nitrates[tiab] OR Nitroglycerin[53] OR nitroglycerin[tiab] OR Glyceryl-Trinitrate[tiab] OR transderm-nitro[tiab] OR nitroderm[tiab] OR Nitric Oxide Donors[53] OR Nitric Oxide[53] OR Isosorbide Dinitrate[53] OR Isosorbide[53] OR oxide-donor[tiab] OR isosorbide-dinitrate[tiab] OR isosorbide[tiab] OR dinitrate[tiab] OR monoxide[tiab] OR mononitrogen[tiab] OR endogenous-nitrate[tiab] OR ((Vasodilator Agents[53] OR vasodilator[tiab] OR vasodilators[tiab] OR vasorelaxant[tiab] OR vasodilatator[tiab] OR Organic Chemicals[53] OR organic[tiab]) AND (nitrate[tiab] OR nitrates[tiab] OR nitric[tiab])) OR nitrogen-monoxide[all fields] OR mononitrate[tiab] OR organic-nitrate[tiab] OR organic-salt[tiab]) AND (osteoporosis[all fields] OR osteopenia[tiab] OR osteoporotic[tiab] OR Bone Diseases, Metabolic[53] OR Bone Density/drug effects[53] OR Bone Resorption[53] OR Bone Density Conservation Agents[53] OR bone-loss[tiab] OR bone-resorption[tiab] OR osteolysis[tiab]))
(Therapy/Broad[filter]) AND (Nitrates[53] OR nitrate[tiab] OR nitrates[tiab] OR Nitroglycerin[53] OR nitroglycerin[tiab] OR Glyceryl-Trinitrate[tiab] OR transderm-nitro[tiab] OR nitroderm[tiab] OR Nitric Oxide Donors[53] OR Nitric Oxide[53] OR Isosorbide Dinitrate[53] OR Isosorbide[53] OR oxide-donor[tiab] OR isosorbide-dinitrate[tiab] OR isosorbide[tiab] OR dinitrate[tiab] OR monoxide[tiab] OR mononitrogen[tiab] OR endogenous-nitrate[tiab] OR ((Vasodilator Agents[53] OR vasodilator[tiab] OR vasodilators[tiab] OR vasorelaxant[tiab] OR vasodilatator[tiab] OR Organic Chemicals[53] OR organic[tiab]) AND (nitrate[tiab] OR nitrates[tiab] OR nitric[tiab])) OR nitrogen-monoxide[all fields] OR mononitrate[tiab] OR organic-nitrate[tiab] OR organic-salt[tiab]) AND (osteoporosis[all fields] OR osteopenia[tiab] OR osteoporotic[tiab] OR Bone Diseases, Metabolic[53] OR Bone Density/drug effects[53] OR Bone Resorption[53] OR Bone Density Conservation Agents[53] OR bone-loss[tiab] OR bone-resorption[tiab] OR osteolysis[tiab]) AND Human[53]
-
b.
Cochrane Central Register of Controlled Trials (Issue 11, 2012)
(Nitrates OR Nitroglycerin OR Nitric Oxide Donors OR Nitric Oxide OR Isosorbide Dinitrate OR Isosorbide OR Vasodilator Agents OR nitrate* OR nitric oxide OR nitroglycerin OR isosorbide OR dinitrate OR monoxide OR mononitrogen OR endogenous nitrate OR nitrogen monoxide OR mononitrate) AND (osteoporosis OR osteopenia OR osteoporotic OR Bone Diseases, Metabolic OR Bone Density OR Bone Resorption OR Bone Density Conservation Agents)
-
c.
EMBASE (Ovid) (1980 to November 2012)
-
1.
nitrate$.mp. or exp nitrate/ (76389)
-
2.
nitroglycerin.mp. or exp glyceryl trinitrate/ (33093)
-
3.
"nitric oxide donor$".mp. or exp nitric oxide/ or exp nitric oxide donor/ (109316)
-
4.
isosorbide dinitrate.mp. (8858)
-
5.
(osteoporosis or osteopenia or osteoporotic).mp. (102897)
-
6.
"bone density conservation agent$".mp. or exp bone density conservation agent/ (2410)
-
7.
"metabolic bone disease$".mp. or exp metabolic bone disease/ (103492)
-
8.
"bone resorption".mp. or exp osteolysis/ (51435)
-
9.
nitric.mp. (173554)
-
10.
nitrat$.mp. (80571)
-
11.
9 or 10 (239764)
-
12.
"vasodilator agent$".mp. (26093)
-
13.
"organic chemicals".mp. (2201)
-
14.
12 or 13 (28294)
-
15.
11 and 14 (5042)
-
16.
1 or 2 or 3 or 4 or 15 (209373)
-
17.
5 or 6 or 7 or 8 (157666)
-
18.
16 and 17 (880)
-
19.
limit 18 to "therapy (maximizes sensitivity)" (266)
-
20.
limit 19 to exclude medline journals (46)
-
21.
limit 19 to human (236)
-
1.
Rights and permissions
About this article
Cite this article
Jamal, S.A., Reid, L.S. & Hamilton, C.J. The effects of organic nitrates on osteoporosis: a systematic review. Osteoporos Int 24, 763–770 (2013). https://doi.org/10.1007/s00198-012-2262-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-012-2262-9