Abstract
Introduction
Quantifying the determinants of bone strength is essential to understanding if or how the structure will fail under load. Determining failure requires knowledge of material and geometric properties. However, characterizing the relative contributions of geometric parameters of bone to overall bone strength has been difficult to date because of limitations in imaging technology. Peripheral quantitative computed tomography (pQCT) uses digital images to derive estimates of bone strength in the peripheral skeleton and is a relatively safe technique to differentiate cortical from trabecular bone and assess bone geometry and density. However, in a compromised osteoporotic bone, thin cortices and low scan resolution can limit accurate analysis.
Methods
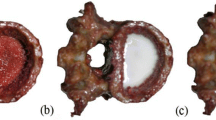
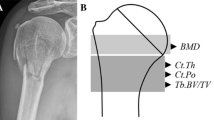
Therefore, in this two-part investigation we scanned ten pairs (n=20) of fresh-frozen radial specimens [female, mean (SD) age 79(6) years] using pQCT (XCT 2000) at the 4 and 30% sites of the distal radius. We investigated the accuracy of four different acquisition resolutions (200, 300, 400, 500 μm) and several analysis modes and thresholds. We evaluated (1) the accuracy of the Norland/Stratec XCT 2000 pQCT in assessing low-density bones by comparing pQCT outcomes to ashing and histomorphometry and (2) the association of geometric parameters by pQCT and areal bone mineral density (aBMD) by dual-energy X-ray absorptiometry (DXA) to failure load at the distal radius.
Results
Using histomorphometry and ashing as reference standards, we found that pQCT scans varied systematically and underestimated or overestimated total area and mineral content at the radial midshaft depending on the analysis algorithm and selected threshold. Overall, most pQCT analysis modes were accurate. In the mechanical testing studies, bone mineral content and cortical bone content at the midshaft were strongly associated with failure load. The pQCT parameters that best accounted for failure load were total content at the 4% site and cortical thickness at the 30% site and they accounted for up to 81% of the variance. The best DXA predictor of failure load was total density at the distal third site and it explained 75% of the variance.
Conclusions
In summary, analysis mode, resolution and thresholding affected pQCT outputs at the radial midshaft. This study extends our understanding of pQCT analysis and provides important data regarding determinants of bone strength at the distal radius.







Similar content being viewed by others
References
Genant HK, Engelke K, Fuerst T, Gluer CC, Grampp S, Harris ST, Jergas M, Lang T, Lu Y, Majumdar S, Mathur A, Takada M (1996) Noninvasive assessment of bone mineral and structure: state of the art. J Bone Miner Res 6:707–730
Marshall D, Johnell O, Wedel H (1996) Meta-analysis of how well measures of bone density predict fractures. Br Med J 312:1254–1259
Myers ER, Hecker AT, Rooks DS, Hipp JA, Hayes WC (1993) Geometric variables from DXA of the radius predict forearm fracture load in vitro. Calcif Tissue Int 52(3):199–204
Lochmuller EM, Lill CA, Kuhn V, Schneider E, Eckstein F (2002) Radius bone strength in bending, compression, and falling and its correlation with clinical densitometry at multiple sites. J Bone Miner Res 17(9):1629–1638
Augat P, Iida H, Jiang Y, Diao E, Genant HK (1998) Distal radius fractures: mechanisms of injury and strength prediction by bone mineral assessment. J Orthop Res 16(5):629–635
Augat P, Reeb H, Claes LE (1996) Prediction of fracture load at different skeletal sites by geometric properties of the cortical shell. J Bone Miner Res 11(9):1356–1363
Bonel HM, Lochmuller EM, Well H, Kuhn V, Hudelmaier M, Reiser M, Eckstein F (2004) Multislice computed tomography of the distal radius metaphysis: relationship of cortical bone structure with gender, age, osteoporotic status, and mechanical competence. J Clin Densitom 7(2):169–182
Eckstein F, Kuhn V, Lochmuller EM (2004) Strength prediction of the distal radius by bone densitometry-evaluation using biomechanical tests. Ann Biomed Eng 32(3):487–503
Hudelmaier M, Kuhn V, Lochmuller EM, Well H, Priemel M, Link TM, Eckstein F (2004) Can geometry-based parameters from pQCT and material parameters from quantitative ultrasound (QUS) improve the prediction of radial bone strength over that by bone mass (DXA)? Osteoporos Int 15(5):375–381
Siu WS, Qin L, Leung KS (2003) pQCT bone strength index may serve as a better predictor than bone mineral density for long bone breaking strength. J Bone Miner Metab 21(5):316–322
Wigderowitz CA, Paterson CR, Dashti H, McGurty D, Rowley DI (2000) Prediction of bone strength from cancellous structure of the distal radius: can we improve on DXA? Osteoporos Int 11(10):840–846
Wu C, Hans D, He Y, Fan B, Njeh CF, Augat P, Richards J, Genant HK (2000) Prediction of bone strength of distal forearm using radius bone mineral density and phalangeal speed of sound. Bone 26(5):529–533
Augat P, Gordon CL, Lang TF, Iida H, Genant HK (1998) Accuracy of cortical and trabecular bone measurements with peripheral quantitative computed tomography (pQCT). Phys Med Biol 43:2873–2883
Takada M, Engelke K, Hagiwara S, Grampp S, Genant HK (1996) Accuracy and precision study in vitro for peripheral quantitative computed tomography. Osteoporos Int 6(3):207–212
Louis O, Boulpaep J, Willnecker J, Van Den Winkel P, Osteaux M (1995) Cortical mineral content of the radius assessed by peripheral QCT predicts compressive strength on biomechanical testing. Bone 16(3):375–379
Louis O, Soykens S, Willnecker J, Van Den Winkel P, Osteaux M (1996) Cortical and total bone mineral content of the radius: accuracy of peripheral computed tomography. Bone 18(5):467–472
Louis O, Willnecker J, Soykens S, Van den Winkel P, Osteaux M (1995) Cortical thickness assessed by peripheral quantitative computed tomography: accuracy evaluated on radius specimens. Osteoporos Int 5(6):446–449
Hangartner TN, Gilsanz V (1996) Evaluation of cortical bone by computed tomography. J Bone Miner Res 11(10):1518–1525
Binkley TL, Specker BL (2000) pQCT measurement of bone parameters in young children: validation of technique. J Clin Densitom 3(1):9–14
Gatti D, Rossini M, Zamberlan N, Braga V, Fracassi E, Adami S (1996) Effect of aging on trabecular and compact bone components of proximal and ultradistal radius. Osteoporos Int 6(5):355–360
Kang Q, An YH, Friedman RJ (1997) Effects of multiple freezing-thawing cycles on ultimate indentation load and stiffness of bovine cancellous bone. Am J Vet Res 58(10):1171–1173
Stratec XCT (2000) Manual; Software Version 5.5,Pforzheim, Germany.http://www.stratec-med.com/literatur/manuals/man3k55e.pdf.
Spoor CF, Zonneveld FW, Macho GA (1993) Linear measurements of cortical bone and dental enamel by computed tomography: applications and problems. Am J Phys Anthropol 91(4):469–484
Hologic (1996) model QDR-4500 user’s guide, Waltham, MA
Muller ME, Webber CE, Bouxsein ML (2003) Predicting the failure load of the distal radius. Osteoporos Int 14(4):345–352
Mall G, Hubig M, Buttner A, Kuznik J, Penning R, Graw M (2001) Sex determination and estimation of stature from the long bones of the arm. Forensic Sci Int 117(1-2):23–30
Hotchkiss CE (1999) Use of peripheral quantitative computed tomography for densitometry of the femoral neck and spine in cynomolgus monkeys (Macaca fascicularis). Bone 24(2):101–107
Ballester M, Zisserman A, Brady M (2002) Estimation of partial volume effect. Medical 6:385–405
Hwang SN, Wehrli FW (2002) Subvoxel processing: a method for reducing partial volume blurring with application to in vivo MR images of trabecular bone. Magn Reson Med 47(5):948–957
Veitch SW, Findlay SC, Ingle BM, Ibbotson CJ, Barrington A, Hamer AJ, Eastell R (2004) Accuracy and precision of peripheral quantitative computed tomography measurements at the tibial metaphysis. J Clin Densitom 7(2):209–217
Spadaro JA, Werner FW, Brenner RA, Fortino MD, Fay LA, Edwards WT (1994) Cortical and trabecular bone contribute to the strength of the osteopenic distal radius. J Orthop Res 12:211–218
Courtney AC, Wachtel EF, Myers ER, Hayes WC (1994) Effects of loading rate on strength of the proximal femur. Calcif Tissue Int 55(1):53–58
Rockoff S, Sweet E, Bleustein J (1969) The relative contribution of trabecular and cortical bone to the strength of human lumbar vertebrae. Calcif Tissue Res 3(2):167–175
Lochmuller EM, Muller R, Kuhn V, Lill CA, Eckstein F (2003) Can novel clinical densitometric techniques replace or improve DXA in predicting bone strength in osteoporosis at the hip and other skeletal sites? J Bone Miner Res 18(5):906–912
Acknowledgements
We gratefully acknowledge the support of the Canadian Institutes of Health Research, Michael Smith Foundation for Health Research and the British Columbia Medical Services Foundation. We thank Dr. Neil White, Dr. Kathy Keiver, Dr. Gilles Galzi, Ms. Cecelia Tang, Ms. Jen Davis and Mr. Jesse Chen for their helpful assistance.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
There are several steps involved in analyzing a pQCT scan. The first step is to define the region of interest (ROI) after scan acquisition. Whatever is contained within the ROI will be included for analysis. Once the ROI is defined, the analysis parameters must be determined. This includes setting the thresholds for separating trabecular from cortical and “subcortical” bone. The XCT 2000 software version 5.50 provides the operator the choice of many combinations of analysis modes. CALCBD refers to the analysis of cancellous or trabecular bone and total bone, and CORTBD to the analysis of cortical bone. Tables 4 and 5 provide an overview of the modes available with the XCT 2000. Within CALCBD there are two parts: Contour Mode and Peel Mode. The software provides three Contour Mode and eight Peel Mode algorithms. Contour Mode (edge detection) separates bone from the surrounding soft tissue. Peel Mode separates trabecular bone from cortical/subcortical bone and CORTBD determines the cortical bone parameters.
Rights and permissions
About this article
Cite this article
Ashe, M.C., Khan, K.M., Kontulainen, S.A. et al. Accuracy of pQCT for evaluating the aged human radius: an ashing, histomorphometry and failure load investigation. Osteoporos Int 17, 1241–1251 (2006). https://doi.org/10.1007/s00198-006-0110-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-006-0110-5