Abstract
Introduction and hypothesis
This systematic review and meta-analysis aim to evaluate the outcomes between SNM and BTX in the treatment of refractory OAB.
Methods
PubMed, Embase, and CENTRAL were comprehensively searched from their inception to December 2019. Randomized and nonrandomized controlled trials evaluating OAB patients who underwent SNM and BTX were included. Data extraction and quality assessment were conducted by two independent reviewers. The outcomes, side effects, and cost-effectiveness values of both procedures were compared in meta-analysis.
Results
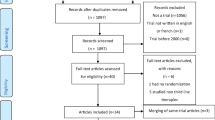
This review involved six articles (2629 patients). Specifically, three articles were based on the same trial, and the other studies were retrospective cohort studies. No significant difference was found in successful treatment between BTX and SNM at 6 months after procedures [risk ratio (RR) = 0.93, 95% confidence interval (CI) 0.63–1.39]. BTX exhibited a significantly higher total adverse event rate than SNM through 6 months (RR = 1.55, 95% CI 1.28–1.88). Patients suffered more urinary tract infection (UTI) risk under BTX injection at the early stage (RR = 1.58, 95% CI 1.10–2.25); however, the difference in UTI events was not significant between the two groups (RR = 1.13, 95% CI 1.10–2.25) during the period of 7–12 months postoperatively. Obviously, the short-term cost (1–2 years) of BTX was significantly lower than that of the SNM procedure.
Conclusions
Both treatments were effective; however, because of the high complication rate of BTX, it may not be a better way to treat refractory OAB than SNM, although BTX is more cost-effective for short-term treatment at present.


Similar content being viewed by others
Abbreviations
- OAB:
-
Overactive bladder
- QoL:
-
Quality of life
- UUI:
-
Urgency urinary incontinence
- BTX:
-
OnabotulinumtoxinA
- SNM:
-
Sacral neuromodulation
- RCT:
-
Randomized controlled trial
- CI:
-
Confidence interval
- LE:
-
Level of evidence
- RR:
-
Risk ratio
- AE:
-
Adverse event
References
Stewart WF, Van Rooyen JB, Cundiff GW, Abrams P, Herzog AR, Corey R, et al. Prevalence and burden of overactive bladder in the United States. World J Urol. 2003;20(6):327–36. https://doi.org/10.1007/s00345-002-0301-4.
Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, et al. The standardisation of terminology of lower urinary tract function: report from the standardisation sub-committee of the international continence society. Neurourol Urodyn. 2002;21(2):167–78. https://doi.org/10.1002/nau.10052.
Coyne KS, Wein A, Nicholson S, Kvasz M, Chen CI, Milsom I. Economic burden of urgency urinary incontinence in the United States: a systematic review. J Manag Care Pharm. 2014;20(2):130–40. https://doi.org/10.18553/jmcp.2014.20.2.130.
Tutolo M, Ammirati E, Heesakkers J, Kessler TM, Peters KM, Rashid T, et al. Efficacy and safety of sacral and percutaneous Tibial Neuromodulation in non-neurogenic lower urinary tract dysfunction and chronic pelvic pain: a systematic review of the literature. Eur Urol. 2018. https://doi.org/10.1016/j.eururo.2017.11.002.
Siegel S, Noblett K, Mangel J, Griebling TL, Sutherland SE, Bird ET, et al. Results of a prospective, randomized, multicenter study evaluating sacral neuromodulation with InterStim therapy compared to standard medical therapy at 6-months in subjects with mild symptoms of overactive bladder. Neurourol Urodyn. 2015;34(3):224–30. https://doi.org/10.1002/nau.22544.
Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, et al. The standardisation of terminology in lower urinary tract function: report from the standardisation sub-committee of the international continence society. Urology. 2003;61(1):37–49. https://doi.org/10.1016/S0090-4295(02)02243-4.
Lightner DJ, Gomelsky A, Souter L, Vasavada SP. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults: AUA/SUFU guideline amendment 2019. J Urol. 2019;202(3):558–63. https://doi.org/10.1097/ju.0000000000000309.
Herschorn S, Kohan A, Aliotta P, McCammon K, Sriram R, Abrams S, et al. The efficacy and safety of onabotulinumtoxinA or solifenacin compared with placebo in solifenacin naive patients with refractory overactive bladder: results from a multicenter, randomized, double-blind phase 3b trial. J Urol. 2017;198(1):167–75. https://doi.org/10.1016/j.juro.2017.01.069.
Biers SM, Venn SN, Greenwell TJ. The past, present and future of augmentation cystoplasty. BJU Int. 2012;109(9):1280–93. https://doi.org/10.1111/j.1464-410X.2011.10650.x.
Amundsen CL, Komesu YM, Chermansky C, Gregory WT, Myers DL, Honeycutt EF, et al. Two-year outcomes of sacral neuromodulation versus onabotulinumtoxinA for refractory urgency urinary incontinence: a randomized trial. Eur Urol. 2018;74(1):66–73. https://doi.org/10.1016/j.eururo.2018.02.011.
Amundsen CL, Richter HE, Menefee SA, Komesu YM, Arya LA, Gregory WT, et al. OnabotulinumtoxinA vs sacral neuromodulation on refractory urgency urinary incontinence in women: a randomized clinical trial. Jama. 2016;316(13):1366–74. https://doi.org/10.1001/jama.2016.14617.
Harvie HS, Amundsen CL, Neuwahl SJ, Honeycutt AA, Lukacz ES, Sung VW, et al. Cost effectiveness of sacral neuromodulation versus onabotulinumtoxinA for refractory urgency urinary incontinence: results of the ROSETTA randomized trial. J Urol. 2019. https://doi.org/10.1097/ju.0000000000000656.
Chughtai B, Clemens JQ, Thomas D, Sun T, Ghomrawi H, Sedrakyan A. Real world performance of sacral neuromodulation and onabotulinumtoxinA for overactive bladder: focus on safety and cost. J Urol. 2020;203(1):179–84. https://doi.org/10.1097/ju.0000000000000462.
Elmer-Lyon CG, Streit JA, Takacs EB, Ten Eyck PP, Bradley CS. Urinary tract infection and drug-resistant urinary tract infection after intradetrusor onabotulinumtoxinA injection versus sacral neuromodulation. Int Urogynecol J. 2019. https://doi.org/10.1007/s00192-019-04007-7.
Singh R, El Nashar SA, Trabuco EC, Klingele CJ, Gebhart JB, Occhino JA. Comparison of short term outcomes of sacral nerve stimulation and intradetrusor injection of onabotulinumtoxinA (Botox) in women with refractory overactive bladder. Female Pelvic Med Reconstr Surg. 2015;21(6):369–73. https://doi.org/10.1097/spv.0000000000000200.
Phillips B, Ball C, Sackett D, Banenoch D, Straus S, Haynes B. Oxford Centre for Evidence-based Medicine-Levels of Evidence. 2009:2009 http://www.cebm.net/index.aspx?o=1025.
Amundsen CL, Richter HE, Menefee S, Vasavada S, Rahn DD, Kenton K, et al. The refractory overactive bladder: sacral neuromodulation vs. botulinum toxin assessment: ROSETTA trial. Contemp Clin Trials. 2014;37(2):272–83. https://doi.org/10.1016/j.cct.2014.01.009.
Siegel S. OnabotulinumtoxinA is preferred over sacral neuromodulation for refractory overactive bladder: con. J Urol. 2017;197(1):11–3. https://doi.org/10.1016/j.juro.2016.10.032.
Carrington EV, Evers J, Grossi U, Dinning PG, Scott SM, O'Connell PR, et al. A systematic review of sacral nerve stimulation mechanisms in the treatment of fecal incontinence and constipation. Neurogastroenterol Motil. 2014;26(9):1222–37. https://doi.org/10.1111/nmo.12388.
Dmochowski R, Chapple C, Nitti VW, Chancellor M, Everaert K, Thompson C, et al. Efficacy and safety of onabotulinumtoxinA for idiopathic overactive bladder: a double-blind, placebo controlled, randomized, dose ranging trial. J Urol. 2010;184(6):2416–22. https://doi.org/10.1016/j.juro.2010.08.021.
Nitti VW, Ginsberg D, Sievert KD, Sussman D, Radomski S, Sand P, et al. Durable efficacy and safety of long-term onabotulinumtoxinA treatment in patients with overactive bladder syndrome: final results of a 3.5-year study. J Urol. 2016;196(3):791–800. https://doi.org/10.1016/j.juro.2016.03.146.
Acknowledgements
This study was supported by grant nos. 81873601 and 81770673 from the National Natural Science Foundation of China and grant no. ZYGD18011 and ZY2017310 from the 1.3.5 Project for Disciplines of Excellence, West China Hospital, Sichuan University.
Author information
Authors and Affiliations
Contributions
Q He: Data collection, Manuscript writing.
BY Li: Data collection, Manuscript writing.
C Zhang: Data analysis.
J Zhang: Data analysis.
DY Luo: Project development, Manuscript editing.
KJ Wang: Project development, Manuscript editing.
Corresponding authors
Ethics declarations
Conflict of interest
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
He, Q., Li, B., Zhang, C. et al. Treatment for refractory overactive bladder: a systematic review and meta-analysis of sacral neuromodulation and onabotulinumtoxinA. Int Urogynecol J 32, 477–484 (2021). https://doi.org/10.1007/s00192-020-04427-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-020-04427-w