Abstract
Introduction and hypothesis
The aims of this study were to assess the anatomical, sexual and functional outcomes of women undergoing surgical intervention for complications of the trocar-guided transvaginal mesh (TVM) procedure.
Methods
This was a retrospective analysis of a clinical database of women who had developed a complication following a TVM procedure. This included dyspareunia, mesh erosion, urinary symptoms, mesh contraction and prolapse recurrence. Pre- and post-operatively, we assessed the women for prolapse, stress incontinence, urgency, defecatory difficulty, digitation, pain, dyspareunia and apareunia. We also recorded the Pelvic Organ Prolapse Quantification (POP-Q) score. The TVM was removed and a Biodesign graft was used in the majority of cases to prevent further prolapse. Follow-up was at 6 weeks, 6 months, 1 and 2 years.
Results
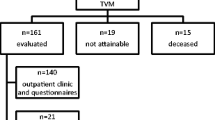
In our cohort of 21 women, 18 required surgery for pain and/or dyspareunia; 20 women had reached the 6-week follow-up at the time of analysis. At 6 weeks, two women still had pain and required a second intervention. Fifteen women had reached a 6-month follow-up and only one woman had persistent pain requiring repeat surgery. Of the 15 women, 7 were sexually active and in 6 cases the dyspareunia had resolved completely with 1 woman retaining an element of pain at intercourse. Six women had been seen at 12 months and all four of the sexually active women had no dyspareunia. There were no symptoms relating to prolapse in any of the women at 6 weeks, 6, 12 or 24 months.
Conclusions
We report satisfactory outcomes following removal of a complicated TVM kit.


Similar content being viewed by others
References
Altman D, Väyrynen T, Engh ME, Axelsen S, Falconer C, Nordic Transvaginal Mesh Group (2011) Anterior colporrhaphy versus transvaginal mesh for pelvic-organ prolapse. N Engl J Med 364(19):1826–1836
Diwadkar GB, Barber MD, Feiner B, Maher C, Jelovsek JE (2009) Complication and reoperation rates after apical vaginal prolapse surgical repair: a systematic review. Obstet Gynecol 113(2 Pt 1):367–373
Marcus-Braun N, von Theobald P (2010) Mesh removal following transvaginal mesh placement: a case series of 104 operations. Int Urogynecol J 21(4):423–430
Feiner B, Jelovsek JE, Maher C (2009) Efficacy and safety of transvaginal mesh kits in the treatment of prolapse of the vaginal apex: a systematic review. BJOG 116(1):15–24
Muffly TM, Barber MD (2010) Insertion and removal of vaginal mesh for pelvic organ prolapse. Clin Obstet Gynecol 53(1):99–114
Feiner B, Maher C (2010) Vaginal mesh contraction: definition, clinical presentation, and management. Obstet Gynecol 115(2 Pt 1):325–330
Hurtado EA, Appell RA (2009) Management of complications arising from transvaginal mesh kit procedures: a tertiary referral center’s experience. Int Urogynecol J Pelvic Floor Dysfunct 20(1):11–17
Altman D, Väyrynen T, Engh ME, Axelsen S, Falconer C, Nordic Transvaginal Mesh Group (2008) Short-term outcome after transvaginal mesh repair of pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct 19(6):787–793
Rane A, Kannan K, Barry C, Balakrishnan S, Lim Y, Corstiaans A (2008) Prospective study of the Perigee system for the management of cystocoeles–medium-term follow up. Aust N Z J Obstet Gynaecol 48(4):427–432
Barber MD, Brubaker L, Nygaard I, Wheeler TL 2nd, Schaffer J, Chen Z, Spino C, Pelvic Floor Disorders Network (2009) Defining success after surgery for pelvic organ prolapse. Obstet Gynecol 114(3):600–609
Maher C, Feiner B, Baessler K, Adams EJ, Hagen S, Glazener CM (2010) Surgical management of pelvic organ prolapse in women. Cochrane Database Syst Rev 4:CD004014
Finamore PS, Echols KT, Hunter K, Goldstein HB, Holzberg AS, Vakili B (2010) Risk factors for mesh erosion 3 months following vaginal reconstructive surgery using commercial kits vs. fashioned mesh-augmented vaginal repairs. Int Urogynecol J 21(3):285–291
US Food and Drug Administration. FDA Safety Communication: Update on serious complications associated with transvaginal placement of surgical mesh for pelvic organ prolapse. July 13, 2011
Zyczynski HM, Carey MP, Smith AR, Gauld JM, Robinson D, Sikirica V, Reisenauer C, Slack M, Prosima Study Investigators (2010) One-year clinical outcomes after prolapse surgery with nonanchored mesh and vaginal support device. Am J Obstet Gynecol 203(6):587.e1–587.e8
Tincello DG (2009) The use of synthetic meshes in vaginal prolapse surgery. BJOG 116(1):1–2
Freeman RM, Lose G (2009) The great mesh debate. Int Urogynecol J Pelvic Floor Dysfunct 20(8):889–891
Feldner PC Jr, Castro RA, Cipolotti LA, Delroy CA, Sartori MG, Girão MJ (2010) Anterior vaginal wall prolapse: a randomized controlled trial of SIS graft versus traditional colporrhaphy. Int Urogynecol J 21(9):1057–1063
de Boer TA, Gietelink DA, Vierhout ME (2008) Discrepancies between physician interview and a patient self-assessment questionnaire after surgery for pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct 19(10):1349–1352
Acknowledgements
Danette Nieuwoudt helped with data collection.
Conflicts of interest
No funding was provided for this paper. Dr. Jeffery has received travel sponsorship from Johnson and Johnson, Boston Scientific, American Medical Systems, Cook and Bard. He has also received speakers fees and honoraria from Johnson and Johnson, Boston Scientific, American Medical Systems and Bard. He is a trainer for Johnson and Johnson, Boston Scientific and American Medical Systems. Dr. Nieuwoudt has received speakers fees and honoraria from Cook Medical and Gynecare (Johnson and Johnson)—all funds were paid into the account of his employer, Stichting Zorgsaam.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeffery, S.T., Nieuwoudt, A. Beyond the complications: medium-term anatomical, sexual and functional outcomes following removal of trocar-guided transvaginal mesh. A retrospective cohort study. Int Urogynecol J 23, 1391–1396 (2012). https://doi.org/10.1007/s00192-012-1746-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-012-1746-7