Abstract
Objective
To assess the incidence of, predisposing factors for, and the rates and relative risks of mortality from acute respiratory distress syndrome (ARDS) in pediatric patients.
Design
A prospective study in 12 consecutive months from 2004 to 2005 in 25 pediatric intensive care units (PICUs).
Patients and setting
ARDS was diagnosed according to the 1994 American–European Consensus Conference definitions, applied to all severely ill admissions between 1 month and 14 years of age. The PICUs were in major municipalities and provincial cities, and half were university affiliated.
Measurements and results
From a total of 12,018 admissions, 7,269 were severely ill. One hundred and five (1.44%) patients developed ARDS and 64 (61.0%) died, which accounts for 13.2%, of the total ICU death (n = 485, 6.7%) or a nine times relative risk of dying. The median age at onset of ARDS was 24 months and 40% were less than 12 month old. Median time from PICU admission to the onset of ARDS was 16 h, and in 63% <24 h. Pneumonia (55.2%) and sepsis (22.9%) were the major predisposing factors for ARDS. These were respectively 14 and 5 times as high a death rate as those of the severely ill patients without ARDS.
Conclusions
ARDS has a high mortality in these Chinese PICUs, especially in those with pneumonia and sepsis, and adequate management including lung protective ventilation strategy is required.
Similar content being viewed by others

Introduction
Acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) are important causes of morbidity and mortality in pediatric intensive care units (PICUs). The incidence of pediatric ARDS varies from study to study depending on the patient population targeted [1–4], and mortality is usually less than 30%. In developing countries, there are very few data available regarding the incidence, clinical management and outcome of pediatric ARDS. In 2001–2002, an initial study was conducted in 15 adult ICUs in Shanghai. In this study the incidence of ARDS was approximately 2% of all ICU admissions, with a 90-day mortality >70%, almost nine times the overall ICU death rate [5]. We found a similar, high mortality in four major PICUs in Shanghai during that period [6]. In the PICU, clinical features of ARDS are different from adult patients in terms of underlying diseases, predisposing factors, management of the initial period, treatment modalities and response, and outcome. Diagnosis and treatment of ARDS based on 1994 American–European Consensus Conference (AECC) definition [7] is widely accepted. Lung protective strategies have been shown to reduce the mortality in adult ARDS, however, the benefits of such strategies have never been examined in the context of PICU in the developing world. We postulated that, by conducting a prospective epidemiologic study in a large number of PICUs, we should be able to identify the incidence and mortality of ARDS and thus provide baseline data for future studies.
Methods
Participating centers
Twenty-five PICUs with a median of 9 (range 4–32) beds participated in the study, representing municipalities and provincial cities in both southeastern and mid-western regions of China (Figure A in “Supplementary materials”). There was 24 h access to clinical radiology and laboratory investigations. Half of the centers were university based with clinical research experience and facilities, while the other half were city central hospitals with limited research commitment. All but four units admitted surgical patients, and five units admitted patients after cardiac surgery. All the centers had senior consultant pediatric intensive care specialists, and provided a clinical resident training program. Each served as the tertiary referral center in pediatric emergency and intensive care for a population of 1–5 million inhabitants. Twenty-two of these centers participated in the study from 1 January to 31 December 2004, and three from 1 June 2004 to 31 May 2005.
Study design and patient management
The study protocol was approved by the Ethics Committee of Children’s Hospital of Fudan University and adopted by each center as required by the Chinese regulations for clinical investigations. As data were collected from observational parameters and no element of specific intervention was used in the protocol, informed consent was waived. All PICU admissions for infants and children in the age group from 1 month to 14 years old were eligible for entry to the study. Only patients with a pediatric critical illness score ≤90 (see Table A in “Supplementary materials”), or who met the American guidelines for admission and discharge policies [8] were included.
Patients and respiratory care
Acute respiratory distress syndrome was defined as acute onset of respiratory failure, hypoxemia (PaO2/FiO2 ≤200 mmHg), bilateral lung infiltration, and lack of left heart failure [7]. To ensure prompt identification and accurate diagnosis of ARDS, we imposed both oxygenation criteria and chest radiographic criteria as simultaneously present within 24 h [9]. FiO2 was based on set and measured values in the ventilators for those dependent on mechanical ventilation, or on measured values obtained proximal to the patient nose and mouth using a CY100 oximeter (Lihua Scientific Instrument, Hangzhou, Zhejiang) in those who breathed spontaneously on oxygen with assisted devices. All chest X-ray films were transmitted to the coordinating center through electronic mail (within 24 h of PaO2/FiO2 ≤200 mmHg) and reviewed by two radiologists to reach agreement. In case of disagreement, X-ray evidence was re-examined in the subsequent workshop. As pulmonary catheterization was not applicable in most participating centers for determination of left atrial hypertension, echocardiographic criteria were used according to Dahlem et al. [2]. We required that echocardiographic examination should be performed within 24 h when bilateral infiltration existed on chest X-ray and when there was any doubt regarding cardiac ventricular function.
For patients with the diagnosis of ARDS who required endotracheal intubation and mechanical ventilation, tidal volume (V T), peak inspiratory pressure (PIP), positive end-expiratory pressure (PEEP), and mean airway pressure (MAP) during the first 3 days of mechanical ventilation were recorded twice daily, and the highest values were used for analysis. For those who deteriorated during non-ventilator treatment with obvious hypoxemia and respiratory distress, endotracheal intubation and mechanical ventilation were implemented. Ventilation modes were variable. The oxygenation index [OI = FiO2 × MAP (cmH2O) × 100/PaO2 (mmHg)] and ventilation index [VI = PIP (cmH2O) × ventilator frequency (cycles/min) × PaCO2 (mmHg)/1,000] were used for estimation of the severity of lung disease and the treatment response. Fluid intake or intravenous infusion was recommended initially at 70% maintenance in the first 3 days of ARDS, with additional fluids as needed for cardiac or nutritional support. For those died of respiratory failure, at least one blood gas was analyzed within 24 h before death.
Underlying diseases, complications and outcome assessment
Underlying diseases of ARDS were categorized according to routine diagnostic definitions in pediatrics. Sepsis and organ dysfunction were identified by using criteria of Levy [10] and the cut-off values of heart and respiratory rate were similar to the definitions for children from Proulx et al. [11]. Respiratory failure was defined as PaO2 ≤50 mmHg in room air at sea level or PaO2/FiO2 ≤250 mmHg under oxygen administration and in the absence of cyanotic congenital heart disease. Survivors were defined as alive at 7 days after discharge from hospitals and verified by telephone contact. Non-survivors included those who died in the hospitals or within 7 days after discharge from the hospital.
Statistical analysis
A simple tabulation was used to describe the data presented as means and SD (when normally distributed) or median and range (when not normally distributed) for continuous variables or incidence ratio in percentage and 95% confidence interval (CI) for categorical variables. Depending on the distribution of data, chi-square test and Fisher’s exact test, Wilcoxon–Mann–Whitney test, or Student’s t-test were used to make comparison of outcomes according to variables of interest and to test significance at alpha error level of 0.05.
Results
Demographic characteristics of PICU admissions
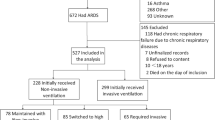
Of the 12,018 patients admitted to the PICU during the 12-month period, 7,269 cases were severely ill and eligible for the study. Among them, 5,590 were medical cases and the others were surgical ones. Average duration of stay in the PICU was 6.6 ± 3.2 days. Four hundred and eighty five (6.7%, 95% CI 6.1–7.3%) of the severely ill patients died in hospital or within 7 days after discharge from hospital.
ARDS patients
Of 7,269 severely ill cases, 105 (1.44%, 95% CI 1.18–1.75%) were diagnosed as having ARDS. Major clinical characteristics of the ARDS patients are presented in Table 1. At the onset of ARDS 42 (40%) were less than 12 months old. In all ARDS patients, 31 (29.5%) had diffuse bilateral infiltrates or white lung on chest X-ray. None of the 47 (44.8%) patients who underwent an echocardiographic examination had elevated left atrium pressure. Thirty-four patients (33.3%) had co-existing clinical conditions (Table 2).
Time and risk factors of ARDS onset
The median (interquartile range) and 95th percentile time from the onset of underlying disease to the onset of ARDS were 72 (24, 120) and 168 h, respectively. The cumulative incidences of ARDS for patients with pneumonia, non-pulmonary sepsis and other disorders are shown in Fig. 1.
Predisposing illnesses associated with the development of ARDS are presented in Table 3. One of the major clinical risk factors for the development of ARDS was identified as predominant for each of the 105 ARDS patients. The major etiologies of ARDS were pneumonia and non-pulmonary sepsis (Table 4), and 62 (59%) patients had ARDS of pulmonary origin.
Respiratory support of ARDS
At the time of ARDS diagnosis, 79 (75.2%) patients were receiving supplemental oxygen through mechanical ventilation, 5 (4.8%) via nasal continuous positive airway pressure, 7 (6.7%) with face mask, 8 (7.6%) with head mask and 6 (5.7%) by nasal cannula. Forty (38.1%) received 100% (FiO2 = 1.0) supplemental oxygen at the time of ARDS diagnosis. PaO2/FiO2 was 95.7 ± 46.3 mmHg (range 14.4–197.5 mmHg) at the onset of ARDS (Table 1). Of 26 non-ventilated patients at inclusion, 18 eventually required intratracheal intubation and mechanical ventilation. In those patients not intubated and ventilated, four were because the attending physicians had not adequately judged the severity of illness, and in the other four palliative therapy was requested by the parents in view of underlying malignant diseases.
Ninety-seven patients received mechanical ventilation, including seven patients with noninvasive mechanical ventilation. The most commonly used ventilatory modes were pressure control (n = 60), synchronized intermittent mandatory ventilation (n = 20), pressure regulated volume control (n = 5), volume control (n = 4), bi-level positive airway pressure (n = 4), nasal continuous positive airway pressure (n = 3) and high frequency oscillation (n = 1). The measurements of lung mechanics at onset of invasive mechanical ventilation are listed in Table 1. The mean (SD) values of the highest PIP, PEEP, MAP and V T during the first 3 days of ventilation were 32.6 ± 10.1 cmH2O, 10.8 ± 6.8 cmH2O, 18.2 ± 6.5 cmH2O and 10.4 ± 5.4 ml/kg, respectively. During this time, approximately 23–28% patients received V T of 6–8 ml/kg, 20–30% below 6 ml/kg, and 40–50% >8 ml/kg. Approximately 30–45% patients were using PEEP levels of ≤5 cmH2O, while 60–80% had PIP ≤30 cmH2O. No statistically significant difference in ventilation parameters was found between survivors and non-survivors.
Mortality
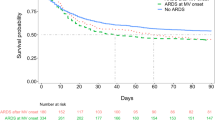
Overall, 485 (6.7%) of PICU patients admitted to the study, died in hospital or within 7 days after discharge. The non-survivors (n = 64) of ARDS represented 13.2% of all non-survivors in these PICUs, i.e., ARDS patients had ten times higher relative risk of death than non-ARDS patients. No difference in mortality rate was found between the university teaching hospitals (37/64, 57.8%) and the non-teaching hospitals (27/41, 65.9%, χ 2 = 0.679, p = 0.410), or between the municipal/provincial hospitals (53/89, 59.6%) and prefectural/regional hospitals (11/16, 68.8%) (χ 2 = 0.482, p = 0.487). Of 64 ARDS non-survivors, 23 (35.9%) died in the first 24 h after the onset of ARDS. Seven (30.4%) of these died within 24 h without mechanical ventilation. No difference in age specific mortality was found (χ 2 = 4.206, p = 0.389). Mortality varied from 79.2% among those with non-pulmonary sepsis, to 60.3% with pneumonia. As shown in Table 4, the relative risks of death in patients with pneumonia or non-pulmonary sepsis were 14.4 and 5.1 times higher, respectively, in those with ARDS.
Discussion
This study is the first to report on the incidence of ARDS in PICUs in China. There are approximately 50 PICUs in China. In the coastal regions, PICUs have been established in major cities for populations of 5–15 million, while in the mid-west regions each province has at least one PICU in the provincial children’s hospital. The participating centers in this study are representative of PICUs in the whole country, with eight of them being national university hospitals, and six of them provincial children’s hospitals of prosperous regions.
We have documented the incidence of, predisposing factors for, mortality and its relative risks from pediatric ARDS in China according to the 1994 AECC definition of ARDS. Although this definition was developed for both adult and pediatric patients, few multicenter, prospective, epidemiologic studies using this definition have been reported in PICU-based patient populations.
Approximately 40% of all patients admitted to PICU during the study periods were found to have relatively mild levels of illness. Among those who were regarded as severely ill, and comparable to those admitted to American PICUs, we found an average incidence of ARDS of 1.4%. This figure is compatible with many of the published reports of ARDS in PICU with incidences varying from 0.7 to 4.2% [12–15]; but most of these studies represent single site and retrospective descriptions of PICU admissions. In a 3-month survey in 92/94 ICUs in Germany, only 12 children with ARDS were identified [4], while Flori et al. [3] recruited 221 ARDS patients in 2 American PICUs over a 4-year period. While 1.9% of PICU admissions in New Zealand and Australia during a 1-year period had ALI [16].
The 61% mortality reported in this study is higher than that reported in 2004 in a 2-month prospective study in 36 PICUs from 7 countries [17]. In that study, there was a mortality of 50% in pediatric ARDS patients receiving mechanical ventilation, which contrasts with the mortality of 26% in the study of Flori et al. [3], but this study included non-ventilated patients. Recently Erickson et al. [16] reported a mortality of 38% in patients who received ventilatory support for ALI in New Zealand and Australia. The wide variation in mortality could be related to many factors including: infrastructure for referral, admission and discharge policy in PICU, severity of illness, patient mix, quality of patient care, resource availability, etc.
The underlying disease pattern may differ in pediatric and adult patients who develop ARDS. In adult ARDS patients, sepsis, pneumonia, trauma, and aspiration of gastric contents are the most common underlying conditions [5, 18–20]. In children with ARDS, pneumonia, sepsis, intoxication and asphyxia are the major causes [1–4, 21]. In general, respiratory and infectious diseases are the two major causes for hospital and ICU admission in infants and children [3]. Tables 3 and 4 illustrate significantly higher risks of mortality for those with ARDS due to pneumonia or sepsis than those without ARDS. Subsequently, these patients had 14 and 5 times higher relative risk of mortality, respectively, when compared to those without ARDS. In this study, only 17 patients had malignant diseases. A limitation of the study is that we did not look at age-stratified death risks compared to respective admissions and disease pattern. Our data suggest that age, underlying diseases, early and adequate management, co-existing clinical conditions such as immuno-compromised subgroup, may be related to different risk of mortality in ARDS.
The diagnostic criteria for pediatric ARDS based on the 1994 AECC definition have only recently been applied in prospective, multicenter clinical trials using surfactant, inhaled nitric oxide, and high frequency oscillation [22–26]. The AECC definitions were also used in clinical epidemiologic studies based on PICU admission [1–3], or regional population profile [4]. Compared to these reports, a limitation of this study is the lack of systematic evaluation of mechanical ventilation. Respiratory care, mechanical ventilation and supportive management were not strictly controlled.
The high mortality within the first 24 h of onset of ARDS may reflect inadequate intervention in the PICU, however, the details of therapy are not fully available. Some deaths occurred because of a conscious decision that it was not appropriate in the context to escalate therapy. A significant proportion of patients received ventilatory patterns (low PEEP, high V T and high PIP) that have been associated with poor outcomes.
In clinical trials of ALI and ARDS in PICU patients [22–26], mortality has ranged from 19–60% in both intervention and control arms. In clinical epidemiologic studies [1, 3], the mortality of ARDS or severe hypoxemic respiratory failure was <30% although in general mortality was higher than in clinical trials [17]. Using a multivariate model, Flori et al. [3] identified three major risk factors that were independent predictors of death and prolonged ventilation: non-pulmonary organ system dysfunction, central nerves system dysfunction and severe hypoxemia at early onset of ARDS, i.e., ALI phase, irrespective of whether mechanical ventilation is required. It appears that differences between our study and that of Flori’s are at least partly due to differences in study design and patient population (e.g., single or multiple centers, observational time span, ALI and/or ARDS), and social, cultural and economic background [14, 15, 21, 27].
The effective lung protective ventilation strategies in ARDS in adults include small tidal volume mechanical ventilation [18–20], non-invasive positive pressure ventilation, prone position [28, 29], and possibly therapies such as exogenous surfactant [22, 23], inhaled nitric oxide [29], high frequency oscillation ventilation [30], and extracorporeal membrane oxygenation [31]. Some of these therapies have been effective in neonates and infants but still require more studies in pediatrics.
During this study, the participating centers had various levels of modern respiratory care facilities, and attending staff steadily improved skills as part of the collaboration. It is possible that uniform implementation of evidence-based protocols for lung protective ventilation could improve patient outcomes with ARDS, and this may warrant an interventional study.
Factors in developing countries affecting the etiology, prognosis and outcome for pediatric ARDS vary from medical resource to social and economic conditions. Organization of collaborative studies in developing countries may have the potential to improve mortality from respiratory diseases in infants and children; the low mortality of ARDS in the developed world is associated with high costs and possibly efficient care. Implementation of advanced respiratory therapies and standardization of care for pediatric ARDS is a challenging and a long-term task for PICU professionals in developing countries. This collaborative study will enable the development of both infrastructure in pediatric respiratory and critical care, and structures to allow for in depth investigation of ARDS and other challenging respiratory diseases in this context.
References
Trachsel D, McCrindle BW, Nakagawa S, Bohn D (2005) Oxygenation index predicts outcome in children with acute hypoxemic respiratory failure. Am J Respir Crit Care Med 172:206–211
Dahlem P, Van Aalderen WM, Hamaker ME, Dijkgaal MGW, Bos AP (2003) Incidence and short-term outcome of acute lung injury in mechanically ventilated children. Eur Respir J 22:980–985
Flori HR, Glidden DV, Rutherford GW, Matthay MA (2005) Pediatric acute lung injury. Prospective evaluation of risk factors associated with mortality. Am J Respir Crit Care Med 171:995–1001
Bindl L, Dresbach K, Lentze MJ (2005) Incidence of acute respiratory distress syndrome in German children and adolescents: a population-based study. Crit Care Med 33:209–212
Lu Y, Song Z, Zhou X, Huang S, Zhu D, Yang X, Bai C, Sun B, Spragg R, Shanghai ARDS Study Group (2004) A 12-month clinical survey of incidence and outcome of acute respiratory distress syndrome in Shanghai intensive care units. Intensive Care Med 30:2197–2203
Lu ZJ, Wang Y, Tang DH, Mi YQ, Sun B, Shanghai Pediatric ARDS Study Group (2003) A survey of acute respiratory distress syndrome in four pediatric intensive care units in 2001. Chin J Pediatr 41:619–620
Bernard GR, Artigas A, Brigham KL, Carlet J, Falke K, Hudson L, Lamy M, Legall JR, Morris A, Spragg RG (1994) The American–European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 149:818–824
American College of Critical Care Medicine of the Society of Critical Care Medicine, American Academy of Pediatrics (1999) Guidelines for developing admission and discharge policies for pediatric intensive care unit. Crit Care Med 27:843–845
Moss M, Thompson BT (2002) Definitions and clinical risk factors. In: Matthay MA (ed) Acute respiratory distress syndrome. Marcel Dekker, New York, pp 7–36
Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, Cohen J, Steven M, Vincent JL, Ramsay G for the International Sepsis Definitions Conference (2003) 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med 31:1250–1256
Proulx F, Fayon M, Farrell CA, Lacroix J, Cauthier M (1996) Epidemiology of sepsis and multiple organ dysfunction syndrome in children. Chest 109:1033–1037
Kühl PG, Appel R, Lasch P, Moller J, Bindl L (1996) Ergebnisse einer Umfrage in deutschen Kinderklinken und gemeinsame Empfehlungen der “Arbeitsgemeinschaft ARDS im Kindesalter” zur Beatmungstherapie. Monatschr Kindrheilkd 144:1110–1116
Costil J, Cloup M, Leclerc F, Devictor D, Beaufils F, Simeonl U, Berthier JC, Berner M, Teyssier G, Rosselot JM, Ensel P, Frappat P, Lefrancois C, Roze JC (1995) Acute respiratory distress syndrome (ARDS) in children: Multicenter collaborative study of French group of pediatric intensive care. Pediatr Pulmonol Suppl 11:106–107
Goh AYT, Chan PWK, Lum LCS, Roziah M (1998) Incidence of acute respiratory distress syndrome: a comparison of two definitions. Arch Dis Child 79:256–259
Norrashidah AW, Azizi BH, Zulfiqar MA (1999) Acute respiratory distress syndrome in a paediatric intensive care unit. Med J Malaysia 54:225–229
Erickson S, Schibler A, Numa A, Nuthall G, Yung M, Pascoe E, Wilkins B on behalf of the Paediatric Study Group; Australian and New Zealand Intensive Care Society (2007) Acute lung injury in pediatric intensive care in Australia and New Zealand—a prospective, multicenter, observational study. Pediatr Crit Care Med 8:317–323
Farias JA, Frutos F, Esteban A, Flores JC, Retta A, Baltodano A, Alía I, Hatzis T, Olazarri F, Petros A, Johnson M (2004) What is the daily practice of mechanical ventilation in pediatric intensive care units? A multicenter study. Intensive Care Med 30:918–925
Brochard L, Roudot-Thoraval F, Roupie E, Delcaux C, Chastre J, Fernandez-Mondejar E, Clementi E, Mancebo J, Factor P, Mataris M, Ranieri M, Blanch L, Rodi G, Mentec H, Dryfuss D, Ferrer M, Brun-Buisson C, Tobin M, LeMaire F, Multicenter Trial Group on Tidal Volume Reduction in ARDS (1998) Tidal volume reduction for prevention of ventilator-induced lung injury in acute respiratory distress syndrome. Am J Respir Crit Care Med 158:1831–1838
The Acute Respiratory Distress Syndrome Network (2000) Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 342:1301–1306
Ferguson ND, Kacmarek RM, Chiche JD, Singh JM, Hallett DC, Mehta S, Stewart TE (2004) Screening of ARDS patients using standardized ventilator settings: influence on enrollment in a clinical trial. Intensive Care Med 30:1111–1116
Bindl L, Buderus S, Dahlem P, Demirakca S, Goldner M, Huth R, Kohl M, Krause M, Kuhl P, Lasch P, Lewandowski K, Merz U, Moeller J, Mohamad Y, Peters M, Porz W, Vierzig A, Ruchard J, Scharf J, Varnholt V (2003) Gender-based differences in children with sepsis and ARDS: the ESPNIC ARDS Database Group. Intensive Care Med 29:1770–1773
Willson DF, Thomas NJ, Markovitz BP, Bauman LA, DiCarlo JV, Pon S, Jacobs BR, Jeffeson LS, Conaway MR, Egan EA for the Pediatric Acute Lung Injury and Sepsis Investigators Network (2005) Effect of exogenous surfactant (Calfactant) in pediatric acute lung injury: a randomized clinical trial. JAMA 293:470–476
Moller JC, Schaible T, Roll C, Schiffmann J-H, Bindl L, Schrod L, Reiss I, Kohl M, Demirakca S, Hentschel R, Paul T, Vierzig A, Groneck P, von Seefeld H, Schumacher H, Gortner L, The Surfactant ARDS Study Group (2003) Treatment with bovine surfactant in severe acute respiratory distress syndrome in children: a randomized multicenter study. Intensive Care Med 29:437–446
Ream RS, Hauver JF, Lynch RE, Kountzman B, Gale GB, Mink RB (1999) Low-dose inhaled nitric oxide improves the oxygenation and ventilation of infants and children with acute, hypoxemic respiratory failure. Crit Care Med 29:989–996
Dobyns EL, Cornfield DN, Anas NG, Fortenberry JD, Tasker RC, Lynch A, Liu P, Eells PL, Griebel J, Baier M, Kinsella JP, Abman SH (1999) Multicenter randomized controlled trial of the effects of inhaled nitric oxide therapy on gas exchange in children with acute hypoxemic respiratory failure. J Pediatr 134:406–412
Dobyns EL, Anas NG, Fortenberry JD, Deshpande J, Cornfield DN, Tasker RC, Liu P, Eells PL, Griebel J, Kinsella JP, Abman SH (2002) Interactive effects of high-frequency oscillatory ventilation and inhaled nitric oxide in acute hypoxemic respiratory failure in pediatrics. Crit Care Med 30:2425–2429
Paret G, Ziv T, Augarten A, Barzilai A, Ben-Abraham R, Manisterski Y, Barzilay Z (1999) Acute respiratory distress syndrome in children: a 10 years experience. Isr Med Assoc J 1:149–153
Vieillard-Baron A, Rabiller A, Chergui K, Peyrouset O, Page B, Beauchet A, Jardin F (2005) Prone position improves mechanics and alveolar ventilation in acute respiratory distress syndrome. Intensive Care Med 31:220–226
Martinez M, Diaz E, Joseph D, Villagrá A, Mas A, Fernandez R, Blanch L (1999) Improvement in oxygenation by prone position and nitric oxide in patients with acute respiratory distress syndrome. Intensive Care Med 25:29–36
David M, Karmrodt J, Weiler N, Scholz A, Markstaller K, Eberle B (2005) High-frequency oscillatory ventilation in adults with traumatic brain injury and acute respiratory distress syndrome. Acta Anaesthesiol Scand 49:209–214
Macintosh I, Butt WW, Robertson CF, Best D, Shekerdemian LS (2005) Extending the limits of extracorporeal membrane oxygenation: lung rest for a child with non-specific interstitial pneumonia. Intensive Care Med 31:993–996
Acknowledgments
This work was supported by grants from China Medical Board of New York (No. 03-786), Shanghai Education and Development Foundation (No. 02SG02), Cheung Kong Scholar’s Program, Ministry of Education and Chair Professorship in Pediatrics of Fudan University (Dr. B. Sun). Authors are grateful to Drs. Guoying Huang and Shubao Chen for their advices in assessment of echocardiographic findings, Dr. Yue Chen, Department of Epidemiology and Community Medicine in University of Ottawa, Canada, for his consultation and advice in statistical analyses of the data, and Dr. Andrew Argent, Pediatric Intensive Care Unit of the Red Cross War Memorial Children’s Hospital, University of Cape Town, South Africa, for constructive comments and suggestions in preparation of the major scientific findings of the manuscript.
Author information
Authors and Affiliations
Consortia
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
List of institutes and investigators of participating PICUs
List of institutes and investigators of participating PICUs
Shanghai Children’s Medical Center of Shanghai Jiaotong University, Shanghai (Ying Wang, Hong Ren); Hangzhou Children’s Hospital of Zhejiang University, Zhejiang (Li-Ping Shi, Tao Liu, Mei-Yue Sun, Li-Zhong Du); Children’s Hospital of Congqing Medical University, Chongqing (Feng-Wu Kuang, Cheng-Jun Liu); Beijing Children’s Hospital of Capital University of Medical Sciences, Beijing (Su-Yun Qian, Xue-Jun Yu, Xun-Mei Fan); Guangzhou Children’s Hospital, Guangdong (Qi-Yi Zeng, Jian-Hui Zhang); Children’s Hospital of Suzhou University, Jiangsu (Min-Hui Xie); Chengdu Children’s Hospital, Sichuan (Guo-Ying Zhang, Wei He); Second Hospital of Hebei Medical University, Shijiazhuang, Hebei (Shao-Dan Zhang); Quanzhou Children’s Hospital, Fujian (De-Yi Zhuang, Rui-Quan Wang); Second Hospital of Chinese Medical University, Shenyang, Liaoning (Chun-Feng Liu, Hui Zhang); Zhengzhou Children’s Hospital, Henan (Xin-Guang Hou); Harbin Children’s Hospital, Heilongjiang (Mei Li, Dong-Yuan Zhou); Shanghai Children’s Hospital, Shanghai (Ding-Hua Tang, Liang Xu, Yu-Ming Zhang); Jiangxi Children’s Hospital, Nanchang, Jiangxi (You-Rong Zhu); Nanjing Children’s Hospital, Jiangsu (Yi Yu, Ai-Dong Cai); Shanxi Children’s Hospital, Taiyuan, Shanxi (Zheng Liu); Wenzhou Yuying Children’s Hospital of Wenzhou Medical College, Zhejiang (Chang-Chong Li, Shi-Jun He); Changchun Children’s Hospital, Jilin (Li-Hong Li, Jian Zhang); Hunan Children’s Hospital, Changsha, Hunan (Yi-Min Zhu, Xiu-Lan Lu); Children’s Hospital of Capital Institute for Pediatric Research, Beijing (Guo-Wei Song, Xiao-Zhuang Gan); Xin Hua Children’s Hospital of Shanghai Jiaotong University, Shanghai (Yue-Qun Mi, Min-Hao Zhu, Jianxin Zhu); Shenzhen Children’s Hospital, Guangdong (Xiao-Hong Liu, Ya-Jun Song); Tianjin Children’s Hospital, Tianjin (Jing-Fu Huang, Xiao-Min Wang); First Hospital of Beijing University, Beijing (Ying Wang); Children’s Hospital of Fudan University, Shanghai [Wen-Liang Yu (Coordinator), Zhu-Jin Lu, Ling-En Zhang, Bo Sun (study director)].
Rights and permissions
About this article
Cite this article
Yu, WL., Lu, ZJ., Wang, Y. et al. The epidemiology of acute respiratory distress syndrome in pediatric intensive care units in China. Intensive Care Med 35, 136–143 (2009). https://doi.org/10.1007/s00134-008-1254-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-008-1254-x