Abstract
Objective
To investigate the role of neuronal nitric oxide synthase (NOS1) in murine polymicrobial peritonitis and sepsis.
Design
Randomized experimental trial.
Setting
Animal research facility.
Subjects
B6129S NOS1+/+ and B6;129S4 NOS−/− mice.
Interventions
NOS1+/+ and NOS1−/− animals underwent cecal ligation and puncture (CLP) or sham surgery and received the NOS1 inhibitor 7-nitroindazole (7-NI) or vehicle.
Measurements and main results
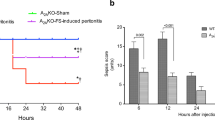
After CLP, genetic deficiency and pharmacologic inhibition of NOS1 significantly increased risk of mortality [8.69 (3.27, 23.1), p < 0.0001 and 1.71 (1.00, 2.92) p = 0.05, hazard ratio of death (95% confidence interval) for NOS1−/− and 7-NI-treated NOS1+/+ respectively] compared with NOS1+/+ animals. In 7-NI-treated NOS1+/+ animals, there were increases (6 h) and then decreases (24 h), whereas in NOS−/− animals persistent increases in blood bacteria counts ( p = 0.04 for differing effects of 7-NI and NOS1−/−) were seen compared with NOS1+/+ animals. After CLP, NOS1−/− had upregulation of inducible NOS and proinflammatory cytokines and greater increases in serum tumor necrosis factor-α and interleukin-6 levels compared with NOS1+/+ mice (all p < 0.05). Following CLP, there were similar significant decreases in circulating leukocytes and lung lavage cells ( p ≤ 0.0008) and significant increases in peritoneal lavage cells ( p = 0.0045) in all groups. Over 6 h and 24 h following CLP, compared with NOS1+/+, NOS−/− mice had significantly higher peritoneal cell concentrations {respectively 0.40 ± 0.09 vs 0.79 ± 0.15 [log(× 104cells/ml)] averaged over both times p = 0.038}.
Conclusions
Deficiency and inhibition of NOS1 increases mortality, possibly by increasing proinflammatory cytokine response and impairing bacterial clearance after CLP. These data suggest that NOS1 is important for survival, bacterial clearance, and regulation of cytokine response during infection and sepsis.







Similar content being viewed by others
References
Albuszies G, Vogt J, Wachter U, Thiemermann C, Leverve XM, Weber S, Georgieff M, Radermacher P, Barth E (2007) The effect of iNOS deletion on hepatic gluconeogenesis in hyperdynamic murine septic shock. Intensive Care Med 33:1094–1101
Siegemund M, van Bommel J, Schwarte LA, Studer W, Girard T, Marsch S, Radermacher P, Ince C (2005) Inducible nitric oxide synthase inhibition improves intestinal microcirculatory oxygenation and CO2 balance during endotoxemia in pigs. Intensive Care Med 31:985–992
Hollenberg SM, Broussard M, Osman J, Parrillo JE (2000) Increased microvascular reactivity and improved mortality in septic mice lacking inducible nitric oxide synthase. Circ Res 86:774–778
Ullrich R, Scherrer-Crosbie M, Bloch KD, Ichinose F, Nakajima H, Picard MH, Zapol WM, Quezado ZM (2000) Congenital deficiency of nitric oxide synthase 2 protects against endotoxin-induced myocardial dysfunction in mice. Circulation 102:1440–1446
Cobb JP, Hotchkiss RS, Swanson PE, Chang K, Qiu Y, Laubach VE, Karl IE, Buchman TG (1999) Inducible nitric oxide synthase (iNOS) gene deficiency increases the mortality of sepsis in mice. Surgery 126:438–442
Ichinose F, Hataishi R, Wu JC, Kawai N, Rodrigues AC, Mallari C, Post JM, Parkinson JF, Picard MH, Bloch KD, Zapol WM (2003) A selective inducible NOS dimerization inhibitor prevents systemic, cardiac, and pulmonary hemodynamic dysfunction in endotoxemic mice. Am J Physiol Heart Circ Physiol 285:H2524–2530
Connelly L, Madhani M, Hobbs AJ (2005) Resistance to endotoxic shock in endothelial nitric-oxide synthase (eNOS) knock-out mice: a pro-inflammatory role for eNOS-derived no in vivo. J Biol Chem 280:10040–10046
Yamashita T, Kawashima S, Ohashi Y, Ozaki M, Ueyama T, Ishida T, Inoue N, Hirata K, Akita H, Yokoyama M (2000) Resistance to endotoxin shock in transgenic mice overexpressing endothelial nitric oxide synthase. Circulation 101:931–937
Ichinose F, Buys ES, Neilan TG, Furutani EM, Morgan JG, Jassal DS, Graveline AR, Searles RJ, Lim CC, Kaneki M, Picard MH, Scherrer-Crosbie M, Janssens S, Liao R, Bloch KD (2007) Cardiomyocyte-specific overexpression of nitric oxide synthase 3 prevents myocardial dysfunction in murine models of septic shock. Circ Res 100:130–139
Quezado Z, Besch V, Hergen A, Rivas R, Gerstenberger E, Cui X, Fitz Y, Eichacker P (2003) Neuronal nitric oxide synthase (NOS1) is protective during bacterial peritonitis and sepsis in mice. Am J Resp Crit Care Med 167:A553
Saini R, Patel S, Saluja R, Sahasrabuddhe AA, Singh MP, Habib S, Bajpai VK, Dikshit M (2006) Nitric oxide synthase localization in the rat neutrophils: immunocytochemical, molecular, and biochemical studies. J Leukoc Biol 79:519–528
Danson EJ, Choate JK, Paterson DJ (2005) Cardiac nitric oxide: emerging role for nNOS in regulating physiological function. Pharmacol Ther 106:57–74
Greenberg SS, Ouyang J, Zhao X, Giles TD (1998) Human and rat neutrophils constitutively express neural nitric oxide synthase mRNA. Nitric Oxide 2:203–212
Huang PL, Dawson TM, Bredt DS, Snyder SH, Fishman MC (1993) Targeted disruption of the neuronal nitric oxide synthase gene. Cell 75:1273–1286
De Sanctis GT, MacLean JA, Hamada K, Mehta S, Scott JA, Jiao A, Yandava CN, Kobzik L, Wolyniec WW, Fabian AJ, Venugopal CS, Grasemann H, Huang PL, Drazen JM (1999) Contribution of nitric oxide synthases 1, 2, and 3 to airway hyperresponsiveness and inflammation in a murine model of asthma. J Exp Med 189:1621–1630
Ortiz PA, Garvin JL (2003) Cardiovascular and renal control in NOS-deficient mouse models. Am J Physiol Regul Integr Comp Physiol 284:R628–638
Saraiva RM, Minhas KM, Raju SV, Barouch LA, Pitz E, Schuleri KH, Vandegaer K, Li D, Hare JM (2005) Deficiency of neuronal nitric oxide synthase increases mortality and cardiac remodeling after myocardial infarction: role of nitroso-redox equilibrium. Circulation 112:3415–3422
Mattson DL, Meister CJ (2005) Renal cortical and medullary blood flow responses to L-NAME and ANG II in wild-type, nNOS null mutant, and eNOS null mutant mice. Am J Physiol Regul Integr Comp Physiol 289:R991–997
Huang Z, Huang PL, Panahian N, Dalkara T, Fishman MC, Moskowitz MA (1994) Effects of cerebral ischemia in mice deficient in neuronal nitric oxide synthase. Science 265:1883–1885
Ward ME, Toporsian M, Scott JA, Teoh H, Govindaraju V, Quan A, Wener AD, Wang G, Bevan SC, Newton DC, Marsden PA (2005) Hypoxia induces a functionally significant and translationally efficient neuronal NO synthase mRNA variant. J Clin Invest 115:3128–3139
Lefer DJ, Jones SP, Girod WG, Baines A, Grisham MB, Cockrell AS, Huang PL, Scalia R (1999) Leukocyte–endothelial cell interactions in nitric oxide synthase-deficient mice. Am J Physiol 276:H1943–1950
Greenberg SS, Ouyang J, Zhao X, Parrish C, Nelson S, Giles TD (1999) Effects of ethanol on neutrophil recruitment and lung host defense in nitric oxide synthase I and nitric oxide synthase II knockout mice. Alcohol Clin Exp Res 23:1435–1445
Enkhbaatar P, Murakami K, Shimoda K, Mizutani A, McGuire R, Schmalstieg F, Cox R, Hawkins H, Jodoin J, Lee S, Traber L, Herndon D, Traber D (2003) Inhibition of neuronal nitric oxide synthase by 7-nitroindazole attenuates acute lung injury in an ovine model. Am J Physiol Regul Integr Comp Physiol 285:R366–372
Alexander HR, Doherty GM, Fraker DL, Block MI, Swedenborg JE, Norton JA (1991) Human recombinant interleukin-1 alpha protection against the lethality of endotoxin and experimental sepsis in mice. J Surg Res 50:421–424
Ichinose F, Mi WD, Miyazaki M, Onouchi T, Goto T, Morita S (1998) Lack of correlation between the reduction of sevoflurane MAC and the cerebellar cyclic GMP concentrations in mice treated with 7-nitroindazole. Anesthesiology 89:143–148
Bush MA, Pollack GM (2000) Pharmacokinetics and protein binding of the selective neuronal nitric oxide synthase inhibitor 7-nitroindazole. Biopharm Drug Dispos 21:221–228
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Li E, Zhou P, Singer SM (2006) Neuronal nitric oxide synthase is necessary for elimination of Giardia lamblia infections in mice. J Immunol 176:516–521
May MJ, Ghosh S (1998) Signal transduction through NF-kappa B. Immunol Today 19:80–88
Togashi H, Sasaki M, Frohman E, Taira E, Ratan RR, Dawson TM, Dawson VL (1997) Neuronal (type I) nitric oxide synthase regulates nuclear factor kappaB activity and immunologic (type II) nitric oxide synthase expression. Proc Natl Acad Sci U S A 94:2676–2680
Qu XW, Wang H, De Plaen IG, Rozenfeld RA, Hsueh W (2001) Neuronal nitric oxide synthase (NOS) regulates the expression of inducible NOS in rat small intestine via modulation of nuclear factor kappa B. FASEB J 15:439–446
Quezado Z, Parent C, Karzai W, Depietro M, Natanson C, Hammond W, Danner RL, Cui X, Fitz Y, Banks SM, Gerstenberger E, Eichacker PQ (2001) Acute G-CSF therapy is not protective during lethal E. coli sepsis. Am J Physiol Regul Integr Comp Physiol 281:R1177–1185
Freeman BD, Quezado Z, Zeni F, Natanson C, Danner RL, Banks S, Quezado M, Fitz Y, Bacher J, Eichacker PQ (1997) rG-CSF reduces endotoxemia and improves survival during E. coli pneumonia. J Appl Physiol 83:1467–1475
Acknowledgements
The authors wish to thank Yu Cao, Yvonne Fitz, and LaShon Middleton for their expert technical support. This research was supported by the Intramural Research Program of the NIH Clinical Center, National Institutes of Health. The authors have no conflict of interest to report.
Author information
Authors and Affiliations
Corresponding author
Additional information
Drs. Cui and Besch contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cui, X., Besch, V., Khaibullina, A. et al. Neuronal nitric oxide synthase deficiency decreases survival in bacterial peritonitis and sepsis. Intensive Care Med 33, 1993–2003 (2007). https://doi.org/10.1007/s00134-007-0814-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-007-0814-9