Abstract
Background
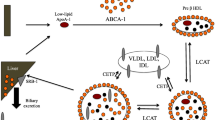
Systemic inflammation and sepsis are accompanied by severe metabolic alterations, including insulin resistance together with increased levels of triglycerides (TGs) and decreases in high- and low-density lipoproteins. Clinical studies have clearly established a link between lipid metabolism and systemic inflammation. Lipoproteins were shown to neutralize LPS and to exert direct anti-inflammatory actions. High- and low-density lipoproteins are thus thought to be important regulators of the host immune response during endotoxemia, which may also have the potential of improving the care of patients with Gram-negative sepsis.
Discussion
Nutritional lipids supplied during critical illness have been shown to modulate the host response to inflammation. In particular, inclusion of ω-3 fatty acids seems to have beneficial effects on cellular immunity and helps to maintain the balance between pro- and anti-inflammatory cytokines thereby preventing hyperinflammatory complications. In addition to improvements in the profile of lipid mediators generated, ω-3 fatty acids act as activating ligands of peroxisome proliferator-activated receptors and directly inhibit nuclear factor κB mediated proinflammatory signaling. We present an overview on the alterations in the metabolism of serum lipoproteins during sepsis and present data from clinical studies and discuss the significance of nutritional lipids and their role in immunomodulation with special emphasis on ω-3 fatty acids.




Similar content being viewed by others
References
Beigneux AP, Moser AH, Shigenaga JK, Grunfeld C, Feingold KR (2000) The acute phase response is associated with retinoid X receptor repression in rodent liver. J Biol Chem 275:16390–16399
Vänttinen M, Nuutila P, Kuulasmaa T, Pihlajamäki J, Hällsten K, Virtanen KA, Lautamäki R, Peltoniemi P, Takala T, Viljanen APM, Knuuti J, Laakso M (2005) Single nucleotide polymorphisms in the peroxisome proliferator-activated receptor δ gene are associated with skeletal muscle glucose uptake. Diabetes 54:3587–3591
Feingold K, Kim MS, Shigenaga J, Moser A, Grunfeld C (2004) Altered expression of nuclear hormone receptors and coactivators in mouse heart during the acute-phase response. Am J Physiol Endocrinol Metab 286(2):E201–207
Pascual G, Fong AL, Ogawa S, Gamliel A, Li AC, Perissi V, Rose DW, Willson TM, Rosenfeld MG, Glass CK (2005) A SUMOylation-dependent pathway mediates transrepression of inflammatory response genes by PPAR-gamma. Nature 437:759–763
Liu D, Zeng BX, Zhang SH, Yao SL (2005) Rosiglitazone, an agonist of peroxisome proliferator-activated receptor γ, reduces pulmonary inflammatory response in a rat model of endotoxemia. Inflamm Res 54:464–470
Mesotten D, Swinnen JV, Vanderhoydonc F, Woulters PJ, Van den Berghe G (2005) Contribution of circulating lipids to the improved outcome of critical illness by glycemic control with intensive insulin therapy. J Clin Endocrinol Metab 89:219–226
Khovidhunkit W, Kim MS, Memon RA, Shigenaga JK, Moser AH, Feingold KR, Grunfeld C (2004) Effects of infection and inflammation on lipid and lipoprotein metabolism: mechanisms and consequences for the host. J Lipid Res 45:1169–1196
Pussinen PJ, Metso J, Malle E, Barlage S, Palosuo T, Sattler W, Schmitz G, Jauhiainen M (2001) The role of plasma phospholipid transfer protein (PLTP) in HDL remodeling in acute-phase patients. Biochim Biophys Acta 1533:153–163
Barlage S, Fröhlich D, Böttcher A, Jauhiainen M, Müller HP, Noetzel F, Rothe G, Schütt C, Linke R, Lackner KJ, Ehnholm C, Schmitz G (2001) ApoE-containing high density lipoproteins and phospholipids transfer protein activity increase in patients with a systemic inflammatory response. J Lipid Res 42:281–290
Carpentier YA, Scruel O (2002) Changes in the concentration and composition of plasma lipoproteins during the acute phase response. Curr Opin Clin Nutr Metab Care 5:153–158
Feingold KR, Grunfeld C (1987) Tumor necrosis factor-alpha stimulates hepatic lipogenesis in the rat in vivo. J Clin Invest 80:184–190
Nonogaki K, Fuller GM, Fuentes NL, Moser AH, Staprans C, Grunfeld C, Feingold KR (1995) Interleukin-6 stimulates hepatic triglyceride secretion in rats. Endocrinology 136:2143–2149
Akgün S, Ertel NH, Mosenthal A, Oser W (1998) Postsurgical reduction of serum lipoproteins: interleukin-6 and the acute-phase response. J Lab Clin Med 131:103–108
Feingold KR, Staprans I, Memon RA, Moser AH, Shigenaga JK, Doerrler W, Dinarello CA, Grunfeld C (1992) Endotoxin reapidly induces changes in lipid metabolism that produce hypertriglyceridemia: low doses stimulate hepatic triglyceride production while high doses inhibit clearance. J Lipid Res 33:1765–1776
Bronfman M, Morales MN, Orellana A (1988) Diacylglycerol activation of protein kinase C is modulated by long-chain acyl-CoA. Biochem Biophys Res Commun 152:987–992
Dresner A, Laurent D, Marcucci M, Griffin ME, Dufour S, Cline GW, Slezak LA, Andersen DK, Hundal RS, Rothman DL, Petersen KF, Shulman GI (1999) Effects of free fatty acids on glucose transport and IRS-associated phosphatidylinositol 3-kinase activity. J Clin Invest 103:253–259
Itani SI, Ruderman NB, Schmieder F, Boden G (2002) Lipid-induced insulin resistance in human muscle is associated with changes in diacylglycerol, protein kinase C, and Iκ-Bα. Diabetes 51:2005–2011
Kitchens RL, Thompson PA, Munford RS, O'Keefe GE (2003) Acute inflammation and infection maintain circulating phospholipid levels and enhance lipopolysaccharide binding to plasma lipids. J Lipid Res 44:2339–2348
Fraunberger P, Schaefer S, Werdan K, Walli AK, Seidel D (1999) Reduction of circulating cholesterol and apolipoprotein levels during sepsis. Clin Chem Lab Med 37:357–362
Leeuwen HJ van, Heezius EC, Dallinga GM, van Strijp JA, Verhoef J, can Kessel JP (2003) Lipoprotein metabolism in patients with severe sepsis. Crit Care Med 31:1359–1366
Feingold KR, Spady DK, Pollock AS, Moser AH, Grunfeld C (1996) Endotoxin, TNF, and IL-1 decrease cholesterol 7 alpha-hydroxylase mRNA levels and activity. J Lipid Res 37:223–228
Memon RA, Moser AH, Shigenaga JK, Grunfeld C, Feingold KR (2001) In vivo and in vitro regulation of sterol 27-hydroxylase in the liver during the acute phase response. Potential role of hepatocyte nuclear factor-1. J Biol Chem 276:30118–30126
Wu A, Hinds CJ, Thiemermann C (2004) High-density lipoproteins in sepsis and septic shock: metabolism, actions, and therapeutic applications. Shock 21:210–221
Pruzanski W, Stefanski E, de Beer FC, de Beer MC, Vadas P, Ravandi A, Kuksis A (2000) Comparative analysis of lipid composition of normal and acute-phase high density lipoproteins. J Lipid Res 41:1035–1047
Cotzee GA, Strachan AF, van der Westhuyzen DR, Hoppe HC, Jeenah MS, de Beer FC (1986) Serum amyloid A-containing human high density lipoprotein 3. Density, size, and apolipoprotein composition. J Biol Chem 261:9644–9651
Cabana VG, Lukens JR, Rice KS, Hawkins TJ, Getz GS (1996) HDL content and composition in acute phase response in three species: triglyceride enrichment of HDL a factor in its decrease. J Lipid Res 37:2662–2674
Cabana VG, Reardon CA, Feng N, Neath S, Lukens J, Getz GS (2003) Serum paraoxonase: effect of the apolipoprotein composition of HDL and the acute phase response. J Lipid Res 44:780–792
Ettinger WH, Miller LD, Albers JJ, Smith TK, Parks JS (1990) Lipopolysaccharide and tumor necrosis factor cause a fall in plasma concentration of lecithin: cholesteryl acyltransferase and lipase deficiency in cynomolgus monkeys. J Lipid Res 31:1099–1107
Masucci-Magoulas L, Moulin P, Jiang XC, Richardson H, Walsh A, Breslow JL, Tall A (1995) Decreased cholesteryl ester transfer protein (CETP) mRNA and protein and increased high density lipoprotein following lipopolysaccharide administration in human CETP transgenic mice. J Clin Invest 95:1587–1594
Post SM, de Crom R, van Haperen R, van Tol A, Princen MG (2003) Increased fecal bile acid excretion in transgenic mice with elevated expression of human phospholipid transfer protein. Arterioscler Thromb Vasc Biol 23:892–897
Feingold KR, Marchall M, Gulli R, Moser AH, Grunfeld C (1994) Effect of endotoxin and cytokines on lipoprotein lipase activity in mice. Arterioscler Thromb Vasc Biol 14:1866–1872
Levels JHM, Lemaire LCJM, van den Ende AE, van Deventer SJH, Lanschot JJB (2003) Lipid composition and lypopolysaccharide binding capacity of lipoproteins in plasma and lymph of patients with systemic inflammatory response syndrome and multiple organ failure. Crit Care Med 31:1647–1653
Schatz IJ, Masaki K, Yano K, Chen R, Rodriguez BL, Curb JD (2001) Cholesterol and all-cause mortality in elderly people from the Honolulu Heart Program: a cohort study. Lancet 358:351–353
Iribarren C, Jacobs, DR Jr, Sidney S, Claxton AJ, Feingold KR (1998) Cohort study of serum total cholesterol and in-hospital incidence of infectious diseases. Epidemiol Infect 121:335–347
Gordon BR, Parker TS, Levine DM, Saal SD, Wang JC, Sloan BJ, Barie PS, Rubin AL (2001) Relationship of hypolipidemia to cytokine concentrations and outcomes in critically ill surgical patients. Crit Care Med 29:1563–1568
Chien JY, Jerng JS, Yu CJ, Yang PC (2005) Low serum level of high-density lipoprotein is a poor prognostic factor for severe sepsis. Crit Care Med 33:1688–1693
Vanni HE, Gordon BR, Levine DM, Sloan BJ, Stein DR, Yurt RW, Saal SD, Parker TS (2003) Cholesterol and interleukin-6 concentrations relate to outcomes in burn-injured patients. J Burn Care Rehabil 24:133–141
Heller AR, Rössler S, Litz RJ, Stehr SN, Heller SC, Koch R, Koch T (2006) Omega-3 fatty acids improve the diagnosis-related clinical outcome. Crit Care Med 34:972–979
Chenaud C, Merlani PG, Roux-Lombard P, Burger D, Harbarth S, Luyasu S, Graf JD, Dayer JM, Ricou B (2004) Low apolipoprotein A-I level at intensive care unit admission and systemic inflammatory response syndrome exacerbation. Crit Care Med 32:632–637
Riemens SC, van Tol A, Sluiter WJ, Dullaart RPF (1998) Plasma phospholipid transfer protein activity is related to insulin resistance: impaired acute lowering by insulin in obese NIDDM patients. Diabetologia 41:929–934
Fielding CJ, Reaven GM, Fielding PE (1982) Human noninsulin-dependent diabetes. Identification of a defect in plasma cholesterol transport normalized in vivo by insulin and in vitro by selective immunoadsorption of apolipoprotein E. Proc Natl Acad Sci USA 79:6365–6369
Oppenheimer MJ, Sundquist K, Bierman EL (1989) Downregulation of high-density lipoprotein receptor in human fibroblasts by insulin and IGF-1. Diabetes 38:117–122
Lay SL, Krief S, Farnier C, Lefrère I, Le Liepvre X, Bazin R, Ferré P, Dugail I (2001) Cholesterol, a cell size-dependent signal that regulates glucose metabolism and gene expression in adipocytes. J Biol Chem 276:16904–16910
Van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P, Bouillon R (2001) Intensive insulin therapy in critically ill patients. N Engl J Med 345:1359–1367
Flegel WA, Wolpl A, Mannel DN, Northoff H (1989) Inhibition of endotoxin-induced activation of human monocytes by human lipoprotein. Infect Immun 57:2237–2254
Harris HW, Johnson JA, Wigmore SJ (2002) Endogenous lipoproteins impact the response to endotoxin in humans. Crit Care Med 30:23–31
Sprong T, Netea MG, van der Ley P, Verver-Jansen TJG, Jacobs LEH, Stalenhoef A, van der Meer JWM, van Deuren M (2004) Human lipoproteins have divergent neutralizing effects on E. coli LPS, N. meningitidis LPS, and complete Gram-negative bacteria. J Lipid Res 45:742–749
Kitchens RL, Wolfbauer G, Albers JJ, Munford RS (1999) Plasma lipoproteins promote the release of bacterial lipopolysaccharide from the monocyte cell surface. J Biol Chem 274:34116–34122
Kitchens RL, Thompson PA, Viriyakosol S, O'Keefe GE, Munford RS (2001) Plasma CD14 decreases monocyte responses to LPS by transferring cell-bound LPA to plasma lipoproteins. J Clin Invest 108:485–493
Vreugdenhil ACE, Snoek AMP, Van't Veer C, Greve JWM, Buurman WA (2001) LPS-binding protein circulates in association with apoB-containing lipoproteins and enhances endotoxin-LDL/VLDL interaction. J Clin Invest 107:224–234
Landmann R, Zimmerli W, Sansano S, Link S, Hahn A, Glauser MP, Calandra T (1995) Increased soluble CD14 is associated with high mortality in gram-negative septic shock. J Infect Dis 171:639–644
Levine DM, Parker TS, Donnelly TM, Walsh A, Rubin AL (1993) In vivo protection against endotoxin by plasma high density lipoprotein. Proc Natl Acad Sci USA 90:12040–12044
McDonald M, Dhadly P, Cockerill GW, Cuzzocrea S, Mota-Filipe H, Hinds CJ, Miller NE, Thiemermann C (2003) Reconstituted high-density lipoprotein attenuates organ injury and adhesion molecule expression in a rodent model of endotoxic shock. Shock 20:551–557
Pajkrt D, Lerch PG, van der Poll T, Levi M, Illi M, Doran JE, Arnet B, van den Ende A, tenCate JW, Deventer SJ (1997) Differential effects of reconstituted high-density lipoprotein on coagulation, fibrinolysis and platelet activation during human endotoxemia. Thromb Haemost 77:303–307
Netea MG, Curfs JH, Demacker PN, Van der Meer JW, Kullberg BJ (1999) Infusion of lipoproteins into volunteers enhances the growth of Candica albicans. Clin Infect Dis 28:1148–1151
Heyland DK, MacDonald S, Keefe L, Drover JW (1998) Total parenteral nutrition in the critically ill patient: a meta-analysis. JAMA 16:280:2013–2019
Lekka ME, Liokatis S, Nathanail C, Galani V, Nakos G (2003) The impact of intravenous fat emulsion administration in acute lung injury. Am J Respir Crit Care Med 169:638–644
Koch T, Heller A (2002) Effects of intravenous fish oil on pulmonary integrity and function. Clin Nutr 21:41–45
Kelbel I, Koch T, Prechtl A, Heller A, Schlotzer E, Schiefer G, Neuhof H (1999) Effects of parenteral application of fish oil versus soy oil emulsions on bacterial clearance functions. Beitr Infusionsther Transfusionsmed 26:226–232
Gupta H, Dai L, Datta G, Garber DW, Grenett H, Li Y, Mishra V, Palgunachari MN, Handattu S, Gianturco SH, Bradley WA, Anantharamaiah GM, White CR (2005) Inhibition of lipopolysaccharide-induced inflammatory responses by an apolipoprotein AI mimetic peptide. Circ Res 97:236–243
Navab M, Anantharamaiah GM, Reddy ST, van Lenten BJ, Data G, Garber D, Fogelman AM (2004) Human apolipoprotein A-I and A-I mimetic peptide: potential for atherosclerosis reversal. Curr Opin Lipidol 15:654–649
Huber J, Vales A, Mitulovic G, Blumer M, Schmid R, Witztum JL, Binder BR, Leitinger N (2002) Oxidized membrane vesicles and blebs from apoptotic cells contain biologically active oxidized phospholipids differentially regulate endothelial binding of monocytes and neutrophils. Arterioscler Thromb Vasc Biol 22:101–107
Berliner JA, Subbanagounder G, Leitinger N, Watson AD, Vora D (2001) Evidence for a role of phospholipids oxidation products in atherogenesis. Trends Cardiovasc Med 11:142–147
Eligini S, Brambilla M, Banfi C, Camera M, Sironi L, Barbieri SS, Auwerx J, Tremoli E, Colli S (2002) Oxidized phospholipids inhibit cyclooxygenase-2 in human macrophages via nuclear factor-κB/IκB-and ERK2-dependent mechanisms. Cardiovasc Res 55:406–415
Chung SW, Kang BY, Kim SH, Pak YK, Cho D, Trinchieri G, Kim TS (2000) Oxidized low density lipoprotein inhibits interleukin-12 production in lipopolysaccharide-activated mouse macrophages via direct interactions between peroxisome proliferators-activated receptor-gamma and nuclear factor-κB. J Biol Chem 275:32681–32687
Hamilton TA, Major JA, Armstrong D, Tebo JM (1998) Oxidized LDL modulates activation of NF-κB in mononuclear phagocytes by altering degradation of IκBs. J Leukoc Biol 64:667–674
Bochkov VN, Kadl A, Huber J, Gruber F, Binder BR, Leitinger N (2002) Protective role of phospholipid oxidation products in endotoxin-induced tissue damage. Nature 419:77–81
Walton, KA Cole AL, Yeh M, Subbanagounder G, Krutzik SR, Modlin RL, Lucas RM, Nakai J, Smart EJ, Vora DK, Berliner JA (2003) Specific phospholipid oxidation products inhibit ligand activation of Toll-like receptors 4 and 2. Arterioscler Thromb Vasc Biol 23:1197–1203
Grundt H, Nilsen DW, Hetland O, Aarsland T, Baksaas I, Grande T, Woie L (1995) Improvement of serum lipids and blood pressure during intervention with n-3 fatty acids was not associated with changes in insulin levels in subjects with combined hyperlipidaemia. J Intern Med 237:249–259
Leaf A (1990) Cardiovascular effects of fish oils. Beyond the platelet. Circulation 82:624–628
Mensink RP, Katan MB (1990) Effect of dietary trans fatty acids on high-density and low-density lipoprotein cholesterol levels in healthy subjects. N Engl J Med 323:439–445
MacLean CH, Newberry SJ, Mojica WA, Khanna P, Issa AM, Suttorp MJ, Lim YW, Traina SB, Hilton L, Garland R, Morton SC (2006) Effects of omega-3 fatty acids on cancer risk: a systematic review. JAMA 295:403–415
Pscheidl E, Schywalsky M, Tschaikowsky K, Boke-Prols T (2000) Fish oil-supplemented parenteral diets normalize splanchnic blood flow and improve killing of translocated bacteria in a low-dose endotoxin rat model. Crit Care Med 28:1489–1496
Weiss G, Meyer F, Matthies B, Pross M, Koenig W, Lippert H (2002) Immunomodulation by perioperative administration of n-3 fatty acids. Br J Nutr 87(Suppl 1):S89–S94
Brown AJ, Pang E, Roberts DC (1991) Persistent changes in the fatty acid composition of erythrocyte membranes after moderate intake of n-3 polyunsaturated fatty acids: study design implications. Am J Clin Nutr 54:668–673
Lehr HA, Hubner C, Finckh B, Nolte D, Beisiegel U, Kohlschutter A, Messmer K (1991) Dietary fish oil reduces leukocyte/endothelium interaction following systemic administration of oxidatively modified low density lipoprotein. Circulation 84:1725–1731
Urakaze M, Hamazaki T, Makuta M, Ibuki F, Kobayashi S, Yano S, Kumagai A (1987) Infusion of fish oil emulsion: effects on platelet aggregation and fatty acid composition in phospholipids of plasma, platelets, and red blood cell membranes in rabbits. Am J Clin Nutr 46:936–940
Lee TH, Sethi T, Crea AE, Peters W, Arm JP, Horton CE, Walport MJ, Spur BW (1988) Characterization of leukotriene B3: comparison of its biological activities with leukotriene B4 and leukotriene B5 in complement receptor enhancement, lysozyme release and chemotaxis of human neutrophils. Clin Sci (Lond) 74:467–475
Needleman P, Raz A, Minkes MS, Ferrendelli JA, Sprecher H (1979) Triene prostaglandins: prostacyclin and thromboxane biosynthesis and unique biological properties. Proc Natl Acad Sci USA 76:944–948
Sperling RI, Robin JL, Kylander KA, Lee TH, Lewis RA, Austen KF (1987) The effects of N-3 polyunsaturated fatty acids on the generation of platelet-activating factor-acether by human monocytes. J Immunol 139:4186–4191
Weber C, Aepfelbacher M, Lux I, Zimmer B, Weber P. C (1991) Docosahexaenoic acid inhibits PAF and LTD4 stimulated [Ca2+]i-increase in differentiated monocytic U937 cells. Biochim Biophys Acta 1133:38–45
Lee JY, Sohn KH, Rhee SH, Hwang D (2001) Saturated fatty acids, but not unsaturated fatty acids, induce the expression of cylooxygenase-2 mediated through Toll-like receptor 4. J Biol Chem 276:16683–16689
Lee JY, Ye J, Gao Z, Youn HS, Lee WH, Zhao L, Sizemore N, Hwang DH (2003) Reciprocal modulation of Toll-like receptor-4 signaling pathways involving MyD88 and phosphatidylinositol 3-kinase/Akt by saturated and polyunsaturated fatty acids. J Biol Chem 278:37041–37051
Lee JY, Zhao L, Youn HS, Wheatherill AR, Tapping R, Feng L, Lee WH, Fitzgerald KA, Hwang DH (2004) Saturated fatty acid activates but polyunsaturated fatty acid inhibits Toll-like receptor 2 dimerization with Toll-like receptor 6 or 1. J Biol Chem 279:16971–16979
Li H, Ruan XZ, Powis SH, Fernando R, Mon WY, Wheeler DC, Moorhead JF, Varghese Z (2005) EPA and DHA reduce LPS-induced inflammation responses in HK-2 cells: evidence for a PPAR-γ-dependent mechanism. Kidney Int 67:867–874
Ertel W, Morrison MH, Ayala A, Chaudry IH (1993) Modulation of macrophage membrane phospholipids by n-3 polyunsaturated fatty acids increases interleukin 1 release and prevents suppression of cellular immunity following hemorrhagic shock. Arch Surg 128:15–20
Bertolini G, Iapichino G, Radrizzani D, Facchini, Simini B, Bruzzone P, Zanforlin G, Tognoni G (2003) Early enteral immunonutrition in patients with severe sepsis: results of an interim analysis of a randomized multicentre clinical trial. Intensive Care Med 29:834–840
Faist E, Hartl WH, Baue AE (1994) Immune mechanisms of post-traumatic hyperinflammation and sepsis. Immun Infekt 22:203–213
Falconer JS, Fearon KC, Ross JA, Elton R, Wigmore SJ, Garden OJ, Carter DC (1995) Acute-phase protein response and survival duration of patients with pancreatic cancer. Cancer 75:2077–2082
Friedman G, Silva E, Vincent JL (1998) Has the mortality of septic shock changed with time? Crit Care Med 26:2078–2086
Gadek JE, DeMichele SJ, Karlstad MD, Pacht ER, Donahoe M, Albertson TE, Van Hoozen C, Wennberg AK, Nelson JL, Noursalehi M (1999) Effect of enteral feeding with eicosapentaenoic acid, gamma-linolenic acid, and antioxidants in patients with acute respiratory distress syndrome. Enteral Nutrition in ARDS Study Group. Crit Care Med 27:1409–1420
Galban C, Montejo JC, Mesejo A, Marco P, Celaya S, Sanchez-Segura JM, Farre M, Bryg DJ (2000) An immune-enhancing enteral diet reduces mortality rate and episodes of bacteremia in septic intensive care unit patients. Crit Care Med 28:643–648
Pontes–Arruda A, Aragao AM, Albuquerque JD (2006) Effects of enteral feeding with eicosapentaenoic acid, gamma–linolenic acid, and antioxidants in mechanically ventilated patients with severe sepsis and septic shock. Crit Care Med 34(9):2325–2333
Heller AR, Rossel T, Gottschlich B, Tiebel O, Menschikowski M, Litz RJ, Zimmermann T, Koch T (2004) Omega-3 fatty acids improve liver and pancreas function in postoperative cancer patients. Int J Cancer 111:611–616
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is discussed in the editorial available at: http://dx.doi.org/10.1007/s00134-006-0434-9.
Rights and permissions
About this article
Cite this article
Wendel, M., Paul, R. & Heller, A.R. Lipoproteins in inflammation and sepsis. II. Clinical aspects. Intensive Care Med 33, 25–35 (2007). https://doi.org/10.1007/s00134-006-0433-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-006-0433-x