Abstract
Background
The management of suspected ventilator-associated pneumonia (VAP) is traditionally based on either a clinical or a “bacteriological” approach. Direct examination of specimens may provide a valuable help to both approaches.
Objective
The objective was to test an algorithm for the management of suspected pneumonia based on severity criteria and Gram stains of specimens, including early treatment of patients with a positive Gram stain of a protected telescoping catheter specimen and awaiting culture results in patients with either Gram stains of endotracheal aspirate and protected specimen both being negative, or only positive endotracheal aspirate (EA), except in the presence of the severity criteria.
Methods
Observational study including 76 first episodes of suspected pneumonia in 4 units. We assessed the rates of correctly diagnosed episodes and of those treated empirically (i.e., ultimately confirmed pneumonia), or appropriately not receiving such therapy (non-confirmed pneumonia), based on protected specimen culture results.
Results
When adhered to, the algorithm allowed early appropriate management in 80% of patients, including 83% of those with confirmed pneumonia, and 74% of those without confirmed infection. The rate of appropriately managed episodes using this algorithm was significantly higher than that using a strategy based on the modified clinical pulmonary infection score (CPIS) alone (80 vs. 50%, p < 0.001). The empiric regimen was adequate in 86% of confirmed episodes.
Conclusion
A strategy based on severity criteria and Gram stain examination of respiratory tract specimens allows correct identification and appropriate management of more than 80% of mechanically ventilated patients who need or do not need therapy for suspected pneumonia.
Similar content being viewed by others
Introduction
The challenge facing clinicians during the management of ventilator-acquired pneumonia (VAP) includes both the requirement for early appropriate antimicrobial therapy when necessary [1], and the need to avoid unnecessary antibiotic prescriptions in patients not needing such therapy [2, 3]. Indeed, the impact on outcome of patients of providing early and appropriate antibiotic therapy for pneumonia has been recently emphasized [4, 5, 6]. Thus, strategies contributing to an early accurate diagnosis of pneumonia in mechanically ventilated patients are clinically important.
The so-called clinical and microbiological approaches to the management of suspected pneumonia remain in opposition [7]. In the former approach [7, 8], the decision to treat and maintain therapy is based on the clinical probability of pneumonia (defined from the usual clinical and radiological criteria), which may include calculation of the Clinical Pulmonary Infection Score (CPIS), as originally described by Pugin et al. [9] or modified [10, 11].However, the clinical criteria for VAP are overly sensitive, which results in excessive antibiotic use [12]. Even refining the score by including Gram stains of respiratory secretion specimens in its calculation may result in over-diagnosing VAP [10]. Other authors emphasize that microbiological findings from selected respiratory tract secretions samples [3, 13, 14] should play a central role in the definitive diagnosis of pneumonia, allowing unnecessary antibiotic use to be reduced while ensuring treatment for patients who really need it [3, 15, 16]. However, culture results are available late relative to the clinical suspicion of pneumonia, and several studies suggest that changes in therapy based on such results do not improve the outcome of patients [5, 6, 17]. Simple techniques allowing pneumonia to be confirmed or ruled out at an early stage of management would thus be clinically useful.
The potential value of Gram stain examination of respiratory tract samples has been rarely assessed. In previous studies describing protected sampling techniques such as the protected telescoping catheter (PTC), Gram stain examination of specimens showed poor sensitivity and high specificity for detecting culture-positive samples [14]. Conversely, the sensitivity of Gram staining of non-protected specimens (endotracheal aspirate, EA) appears high, while its specificity is poor [18]. Therefore, the two techniques could be complementary in the diagnostic approach. Blot et al. [19] have suggested that the combination of direct examination of the two samples would help in the early management of suspected VAP. In the light of their results, they proposed a decision tree using Gram stain examination of both EA and PTC, to guide empiric therapy when needed, and to avoid to a large extent unnecessary therapy.
The aim of this prospective observational multicenter study was to confirm the value of the combination of Gram stain examination of both EA and PTC for the early diagnosis of VAP and to assess the performance of the algorithm proposed by Blot et al. in the initial management of suspected VAP.
Materials and methods
The study was performed in 4 ICUs (2 medical ICUs, 1 surgical ICU, and 1 mixed ICU) between January 2002 and December 2003. The study was approved by the Ethics Committee of the Société de Réanimation de Langue Française, and informed consent was waived.
The following parameters were recorded in patients with clinically suspected pneumonia (please refer to the ESM for details): demographic data, ICU admission diagnosis and severity [20], and severity scores [21, 22] at the time of suspected pneumonia. The CPIS was also calculated on the day of inclusion (see ESM, Table S1) according to Singh et al. [11], and after including EA Gram stain examination (CPIS-EA Gram) or PTC Gram stain examination (CPIS-PTC Gram) [10]. Severity criteria for VAP were defined as extensive lung involvement or severe hypoxemia (PaO2/FiO2 ratio less than 200), or occurrence of severe sepsis or shock.
Sampling techniques and definitions
Lower respiratory tract samples were obtained on the day of clinical suspicion of pneumonia. An EA was first obtained for Gram staining only. A PTC was then performed as previously described [14], for Gram staining and quantitative culture (ESM, Figure S1). The sensitivity, specificity, predictive positive and negative values of EA and PTC Gram stains were calculated according to a positive culture of the PTC, defined as the recovery of at least 103 cfu/ml of a potential pathogen [10, 14, 19, 23].
Algorithm tested
As proposed by Blot et al. [19], four situations were considered, each corresponding to a recommendation for initiating therapy (Fig. 1):
-
1.
When the PTC Gram stain was positive, pneumonia was considered very likely; empiric therapy was initiated, and therapy secondarily adapted to PTC culture results to maintain, modify or stop therapy;
-
2.
When EA Gram stain examination was negative (as well as direct examination of PTC), the probability of pneumonia was low, and empiric antimicrobial treatment was withheld pending PTC culture results;
-
3.
The Gram stain of PTC was negative and Gram stain of EA was positive:
-
a)
Empiric antimicrobial treatment was introduced if patients had severity criteria;
-
b)
Empiric antimicrobial treatment could be withheld in the absence of such criteria.
-
a)
Empiric antibiotic regimens
A recommended first-line empiric antimicrobial regimen was defined at each center, based on local epidemiological data, the time of occurrence of suspected VAP relative to initiation of mechanical ventilation (early-onset, i.e., before day 7; or late-onset, after 7 or more days), recent hospitalization, and prior antibiotic therapy [7, 24] (see the ESM for details).
Analysis
The primary end-point was the accuracy of the decisional tree in terms of the decision to treat or not (i.e., to predict the presence of VAP). Secondary end-points were the adequacy of empirical therapy, based on the algorithm proposed, according to final PTC culture results and susceptibility of the organisms recovered. This was examined both according to therapy actually administered and when considering 100% adherence to the recommended regimens. Mortality rate at 15 days and duration of MV and of ICU stay were examined in patients with confirmed VAP and according to the adequacy of the empiric antibiotic regimen actually administered.
Data were recorded on Epi-Info 2000 (CDC, Atlanta, GA, USA) and statistical analyses conducted using Statview Software (SAS Institute, Berkeley, CA, USA). Continuous variables were described by their means and standard deviations (SD), and compared using Student's t test or Mann–Whitney U-test, when appropriate. Qualitative variables were described by their frequency and compared using the chi-squared test. A value of p < 0.05 was considered significant.
Results
Patients
We studied 78 patients with suspected VAP, 76 of whom were evaluable (2 patients were excluded because of missing data). The 76 patients had a mean age of 59 ± 15 years, a mean ICU admission SAPS II of 42 ± 17, and 13 (17%) patients had shock. Their overall mean duration of MV and length of ICU stay were 30 ± 29 and 43 ± 36 days respectively, and 32 patients (42%) died in the ICU. Patients had received MV for 10 ± 8.8 days at the time of VAP suspicion (Table 1); 44 episodes (58%) were late-onset; and 57 (75%) patients had received antibiotics for 2 days or more within the preceding 15-day period (see the ESM for details).
Microbiological results and concordance with the algorithm tested
The direct examinations of EA and PTC were positive in 53 (70%) and 36 (47%) episodes respectively. Forty-one (54%) PTC specimen cultures grew 103 cfu or more, including 30 (73%) monomicrobial and 11 (27%) polymicrobial episodes (ESM, Table S2). Potentially resistant pathogens were recovered from 16 (29%) patients. Gram stain examination and PTC culture results were concordant in 35 (92%) cases, partially so in 2 (5%), and discordant in 1.
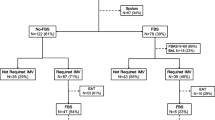
Figure 2 shows the distribution of patients according to the results of Gram stain examinations and the final diagnosis (see the ESM for details). Of the 36 (47%) patients with a positive PTC Gram stain examination, 30 (83%) had confirmed VAP, and of the 21 patients with negative Gram stain examination of both EA and PTC, only 3 had positive PTC culture. In the remaining 19 (25%) episodes, EA Gram stain examination was positive whereas PTC Gram stain examination was negative; 8 (42%) had positive PTC cultures, including 4 of the 7 (57%) patients with severity criteria and 4 of the remaining 12 (33%) without such criteria.
Number of episodes and number of corresponding VAP confirmed by PTC culture in each group according to the algorithm tested
Using this approach, 34 out of 41 (83%) confirmed VAP episodes would appropriately be treated empirically, while 26 out of 35 (74%) not needing therapy are not treated empirically
Combining EA and PTC Gram stain examinations according to the algorithm tested resulted in sensitivity, specificity, and positive and negative predictive values for VAP of 83%, 74%, 79%, and 79% respectively (Fig. 2, Table 2), thus allowing the need for empiric therapy of 34 out of 41 (83%) patients with PTC culture-confirmed VAP to be correctly predicted, as well as the lack of need for such therapy in 26 out of 35 (74%) episodes of non-confirmed VAP.
Predictive accuracy of alternative strategies
The above results were compared with treatment decisions expected from a clinical strategy based on the CPIS scores, simplified or including Gram stain of EA or PTC [10, 11]. Taking a modified CPIS higher than 6 as indicative of a probable VAP justifying antimicrobial therapy, the corresponding sensitivity, specificity, and positive and negative predictive values of each of these different CPIS calculations are shown in Table 2, in comparison to the algorithm tested (see ESM for details).
The overall rate of correct decisions to initiate (or not) empiric therapy according to the strategy tested (79%) was significantly higher than with management strategies using the simplified CPIS (36 out of 76 [47%]; p < 0.01) or CPIS-EA Gram stain (45 out of 76 [59%]; p = 0.014). Compared with the strategy using the CPIS-PTC Gram stain [10], there were still more correct decisions overall with the strategy tested (79% vs. 46 out o f76 [61%]; p = 0.022). Fewer patients with VAP eventually confirmed by PTC culture would not receive empiric therapy with the strategy tested (7 out of 41; 17%) than with a strategy based on the simplified CPIS (28 out of 41 [68%]; p < 0.001); compared with the CPIS-Gram EA and PTC strategies, this difference was not statistically significant (p = 0.59 and p = 0.13 respectively; Table 3).
Observance of the therapeutic strategy
Twenty nine of the 36 (81%) episodes with positive PTC Gram stains were actually treated empirically by the attending physicians (Table 3). Only 1 of the 21 episodes with negative EA and PTC Gram stains received empirical therapy, and 20 did not receive such therapy. In the 19 episodes with a positive EA Gram stain together with a negative PTC Gram stain, antibiotics were administered empirically in 6 out of 7 episodes with severity criteria; in their absence (12 episodes), empiric antibiotics were not administered.
Overall, empiric therapy was initiated by the attending physicians in 35 out of 43 (81%) episodes, justifying therapy according to the algorithm tested, and 32 out of 33 (97%) episodes not justifying such therapy were not treated empirically. Thus, the algorithm tested was not adhered to in 9 (12%) episodes (8 with a positive PTC Gram stain, and 1 with both negative Gram stains). The antibiotic regimens recommended by the protocol were adequate in 36 (88%) of the 41 episodes with positive PTC cultures; unanticipated resistance was associated with the 5 episodes in which the recommended regimen was not effective (see ESM for details).
Outcomes of patients
The mean duration of mechanical ventilation was 30.1 ± 29 days overall, and it was non-significantly higher in patients with confirmed VAP (33.5 ± 33.7 days) than in patients without confirmed VAP (26.1 ± 22.9 days; p = 0.27). Their overall mean length of stay in the ICU was 43.2 ± 36.3 days. Patients with confirmed VAP stayed significantly longer in the ICU than patients without confirmed VAP (57.1 ± 33.7 days vs. 31.9 ± 26.7 days; p = 0.01).
The overall 15-day and ICU mortality rates were 18% (14 out of 76) and 42% (32 out of 76 patients). The 15-day mortality rate was 17% (7 out of 41) in patients with confirmed VAP, and 20% (7 out of 35) in patients with non-confirmed VAP. Survival at 15 days was similar between patients receiving initially appropriate (20 out of 26, 77%) and those receiving inappropriate empiric antimicrobial therapy or no empiric therapy (10 out of 15, 67%; p = 0.5).
Discussion
Our findings suggest that a management strategy based on Gram stains of respiratory tract secretion specimens (both protected and non-protected specimens) allows rapid and correct identification of 80% of patients with suspected VAP needing or not needing initial empiric therapy. Our results also confirm that clinical strategies based on CPIS calculation alone do not perform as well in this regard [10], although patients with confirmed pneumonia are better identified using a modified CPIS incorporating Gram stain results than when using the modified CPIS described by Singh et al. [11].
The debate continues as to which of the so-called clinical or microbiological approaches is preferred—a somewhat artificial debate, as most physicians actually combine the two—for initial management of patients with suspected VAP. The recent ATS/IDSA guideline incorporates and contrasts the two strategies [7], noting that the “clinical strategy” is overly sensitive and likely entails more false-positives than the “microbiological” one in terms of identification of patients requiring antibiotic therapy, whereas the major criticism of the latter strategy is the potential problem of false-negatives.
Within the context of this prospective multicenter study, our results confirm the findings from the previous single-center study by Blot et al. [19] suggesting that a combined clinical and microbiological approach, taking into account the severity of presentation of suspected VAP and the results of Gram stains of protected and non-protected specimens, could help the decision to initiate empiric therapy. When adhered to, the algorithm tested allowed both initiating therapy in over 80% of patients with eventually confirmed pneumonia and withholding therapy in over 70% of those not needing it. Moreover, using a simple algorithm for empiric selection of drugs, adequate therapy could be administered to most patients treated, a result comparable to those obtained in recent studies in which a uniform pre-specified protocol was used for initial empiric therapy of suspected VAP, although with much broader spectrum regimens in most instances [25].
It is noteworthy that the protocol tested gave more accurate results than one based on clinical evaluation and CPIS calculation. Compared with confirmed VAP based on PTC cultures, the modified CPIS (> 6) had suboptimal predictive values of only about 50%. Although the operative characteristics of the modified CPIS improved when incorporating Gram stain results of respiratory tract specimens into the score calculation, its accuracy remained relatively low, in accordance with our previous findings when using BAL as the reference standard [10]. In that study, we also reported that a modified CPIS incorporating the results of Gram stains of BAL or PTC fluid marginally improved the performance of the score to diagnose BAL-confirmed pneumonia.
Our strategy allowed early identification of 83% patients with confirmed VAP, thus leaving 17% (7 out of 41) of patients with VAP not identified early and receiving delayed therapy for up to 24 h following clinical suspicion (Fig. 2). Among these 7 patients with negative PTC Gram stains, 4 had a positive EA Gram stain. It could thus be argued that using a positive EA Gram stain to decide to initiate empiric therapy would result in avoiding some “missed opportunities” for early therapy; however, this would also result in treating 11 more patients unnecessarily, for an overall 43% (15 out of 35) unnecessary therapy rate of patients without confirmed pneumonia, whereas only 9 (26%) would receive such therapy according to the strategy tested.
According to the protocol, only 1 of the patients receiving delayed therapy had severity criteria. In this small selected group of patients with a low to moderate probability of VAP, the outcome was not worse than that of other patients with confirmed pneumonia who received early appropriate therapy. However, appropriate therapy was provided to all these patients following early culture results within 24 h of sampling. The acceptable lag time between clinical suspicion of VAP and administration of appropriate therapy in such patients has not been determined. Despite a recent study suggesting the reverse [26], our results do not therefore support the concept that patients with mild to moderate disease severity have a worse outcome when not immediately treated upon clinical suspicion of pneumonia. However, given the small number of these patients, our data should be interpreted with caution and further experience is needed in this regard.
Central to the interpretation of our results are the criteria used to diagnose and confirm pneumonia in our patients. We used blind PTC quantitative culture to define pneumonia, and it can be argued that using this criterion biased the results against clinical evaluation. However, the PTC technique—a technique, which, despite its minimal invasiveness, still uses protected sampling and quantitative cultures—has now been compared in several studies with more widely accepted tests such as BAL, and has been shown to provide similar accuracy to other protected or non-invasive techniques cultured quantitatively [10, 14, 23]. It is most convenient when bronchoscopy is impractical or not available [27]. Nevertheless, PTC could have suboptimal sensitivity, and some patients included in the group with non-confirmed pneumonia may have had broncho-pulmonary infection. We believe this occurred rarely, as indirectly suggested from the outcome of patients with and without confirmed pneumonia by our criteria; the latter subgroup did not appear to have a worse outcome despite the withholding or withdrawing of antibiotic therapy. Nevertheless, the groups were of limited size, and further experience with this approach is needed to confirm its safety.
Although the management strategy examined in this study takes culture results of protected and quantitative specimens as the reference standard, it does allow for integration of clinical information, such as a high clinical probability of pneumonia and severity of its presentation, which corresponds to clinical reasoning in practice. Clearly, patients presenting with severity criteria should receive empiric therapy. In these patients, microbiological information are used only to help in the selection of drugs (Gram stain), and culture results to streamline therapy; this is mostly a “clinical” approach. In patients without severity criteria—the majority of patients with suspected VAP (71% of episodes in our series)—more guidance can be sought from microbiological information, including withholding of therapy in patients with low/moderate probability of pneumonia when Gram stains are negative, as mentioned in the recent ATS/IDSA guidelines [7]. Instead of the clinical probability of pneumonia, we used in this study severity criteria as a major clinical component for decision-making. An alternative algorithm to the one we tested could incorporate for example a modified CPIS including Gram stain results [28] (ESM, Figure S2). In terms of number of episodes appropriately treated, this approach would provide similar sensitivity (83%) to those shown in Fig. 2, but twice as many patients (24%) would be treated unnecessarily.
When based on the regimens recommended for empiric therapy, the actual therapy administered to our patients was adequate (according to PTC culture results) in 83% of patients treated. It is noteworthy that this result was obtained despite using a relatively conservative approach in the selection of drugs administered, based on epidemiological data recorded in the participating units, which confirms the importance of repeated monitoring of local epidemiological and susceptibility surveillance [29]. However, a few patients received inadequate initial therapy because of unanticipated species or resistance. A higher adequacy rate of initial therapy might be achieved by using routinely broader-spectrum empirical regimens in patients with clinically suspected pneumonia [30]; however, this approach is debatable, since it entails unnecessary therapy in many patients who do not need such therapy, increasing the risk of VAP and antibiotic resistance [24].
References
Iregui M, Ward S, Sherman G, Fraser VJ, Kollef MH (2002) Clinical importance of delays in the initiation of appropriate antibiotic treatment for ventilator-associated pneumonia. Chest 122:262–268
Kollef MH, Fraser VJ (2001) Antibiotic resistance in the intensive care unit. Ann Intern Med 134:298–314
Chastre J, Fagon JY (2002) State of the art: ventilator-associated pneumonia. Am J Respir Crit Care Med 165:867–903
Kollef MH, Sherman G, Ward S, Fraser VJ (1999) Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest 115:462–474
Luna CM, Vujacich P, Niederman MS, Vay C, Gherardi C, Matera J, Jolly EC (1997) Impact of BAL data on the therapy and outcome of ventilator-associated pneumonia. Chest 111:676–685
Rello J, Gallego M, Mariscal D, Sonora R, Vallés J (1997) The value of routine microbial investigation in ventilator-associated pneumonia. Am J Respir Crit Care Med 156:196–200
Niederman M, Craven DE, Bonten MJ, Chastre J, Craig WA, Fagon JY, Hall JB, Jacoby GA, Kollef MH, Luna CM, Mandell LA, Torres A et al. (2005) Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171:388–416
Grossman RF, Fein AM (2000) Evidence-based assessment of diagnostic tests for ventilator-associated pneumonia. Executive summary. Chest 117:177S–181S
Pugin J, Auckenthaler R, Mili N, Janssens JP, Lew PD, Suter PM (1991) Diagnosis of ventilator-associated pneumonia by bacteriologic analysis of bronchoscopic and non-bronchoscopic “blind” bronchoalveolar lavage fluid. Am Rev Respir Dis 143:1121–1129
Fartoukh M, Maitre B, Honore S, Cerf C, Zahar JR, Brun-Buisson C (2003) Diagnosing pneumonia during mechanical ventilation: the clinical pulmonary infection score revisited. Am J Respir Crit Care Med 168:173–179
Singh N, Rogers P, Atwood CW, Wagener MM, Yu VL (2000) Short-course empiric antibiotic therapy for patients with pulmonary infiltrates in the intensive care unit. A proposed solution for indiscriminate antibiotic prescription. Am J Respir Crit Care Med 162:505–511
Luyt CE, Chastre J, Fagon JY (2004) Value of the clinical pulmonary infection score for the identification and management of ventilator-associated pneumonia. Intensive Care Med 30:844–852
Bregeon F, Papazian L, Thomas P, Carret V, Garbe L, Saux P, Drancourt M, Auffray JP (2000) Diagnostic accuracy of protected catheter sampling in ventilator-associated bacterial pneumonia. Eur Respir J 16:969–975
Pham LH, Brun-Buisson C, Legrand P, Rauss A, Verra F, Brochard L, Lemaire F (1991) Diagnosis of nosocomial pneumonia in mechanically ventilated patients: comparison of a plugged telescoping catheter with the protected specimen brush. Am Rev Respir Dis 143:1055–1061
Bonten MJ, Bergmans DC, Stobberingh EE, van der Geest S, de Leeuw PW, van Tiel F, Gaillard CA (1997) Implementation of bronchoscopic techniques in the diagnosis of ventilator-associated pneumonia to reduce antibiotic use. Am J Respir Crit Care Med 156:1820–1824
Heyland DK, Cook DJ, Marshall JC, Heule M, Guslits B, Lang J, Jaeschke R, The Canadian Critical Care Trials Group (1999) The clinical utility of invasive diagnostic techniques in the setting of ventilator-associated pneumonia. Chest 115:1076–1084
Kollef MH, Ward S (1998) The influence of mini-BAL cultures on patient outcomes. Implications for the antibiotic management of ventilator-associated pneumonia. Chest 113:412–420
Jourdain B, Novara A, Joly-Guillou M-L, Dombret MC, Calvat S, Trouillet JL, Gibert C, Chastre J, Joly-Guillou ML (1995) Role of quantitative cultures of endotracheal aspirates in the diagnosis of nosocomial pneumonia. Am J Respir Crit Care Med 152:241–246
Blot F, Raynard B, Chachaty E, Tancrede C, Antoun S, Nitenberg G (2000) Value of gram stain examination of lower respiratory tract secretions for early diagnosis of nosocomial pneumonia. Am J Respir Crit Care Med 162:1731–1737
Le Gall JR, Lemeshow S, Saulnier F (1993) A new simplified acute physiology score (SAPS II) based on a European-North American multicenter study. JAMA 270:2957–2963
Vincent JL, de Mendonca A, Cantraine F, Moreno R, Takala J, Suter PM, Sprung CL, Colardyn F, Blecher S (1998) Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: results of a multicenter, prospective study. Working group on “sepsis-related problems” of the European Society of Intensive Care Medicine. Crit Care Med 26:1793–1800
Bone RC, Balk RA, Cerra FB, Dellinger EP, Fein AM, Knaus WA, Schein RM, Sibbald WJ, the ACCP/SCCM Consensus Conference Committee (1992) Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Chest 101:1656–1662
Mentec H, May-Michelangeli L, Rabbat A, Varon E, Le Turdu F, Bleichner G (2004) Blind and bronchoscopic sampling methods in suspected ventilator-associated pneumonia: a multicentre prospective study. Intensive Care Med 30:1319–1326
Trouillet JL, Chastre J, Vuagnat A, Joly-Guillou M-L, Combaux D, Dombret MC, Gibert C (1998) Ventilator-associated pneumonia caused by potentially drug-resistant bacteria. Am J Respir Crit Care Med 157:531–539
Micek ST, Ward S, Fraser VJ, Kollef MH (2004) A randomized controlled trial of an antibiotic discontinuation policy for clinically suspected ventilator-associated pneumonia. Chest 125:1791–1799
Clec'h C, Timsit JF, De Lassence A, Azoulay E, Alberti C, Garrouste-Org, Mourvilier B, Troche G, Tafflet M, Tuil O, Cohen Y (2004) Efficacy of adequate early antibiotic therapy in ventilator-associated pneumonia: influence of disease severity. Intensive Care Med 30:1327–1333
Hubmayr RD (2002) Statement of the 4th International Consensus Conference in Critical Care on ICU-Acquired Pneumonia—Chicago, Illinois, May 2002. Intensive Care Med 28:1521–1536
Brun-Buisson C, Fartoukh M, Lechapt E, Honoré S, Zahar JR, Maitre B (2005). Contribution of blinded, protected quantitative specimens to the diagnostic and therapeutic management of ventilator-associated pneumonia. Chest 128:533–544
Rello J, Sa-Borges M, Correa H, Leal SR, Baraibar J (1999) Variations in etiology of ventilator-associated pneumonia across four treatment sites: implications for antimicrobial prescribing practices. Am J Respir Crit Care Med 160:608–613
Ibrahim EH, Ward S, Sherman G, Schaiff R, Fraser VJ, Kollef MH (2001) Experience with a clinical guideline for the treatment of ventilator-associated pneumonia. Crit Care Med 29:1109–1115
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Veinstein, A., Brun-Buisson, C., Derrode, N. et al. Validation of an algorithm based on direct examination of specimens in suspected ventilator-associated pneumonia. Intensive Care Med 32, 676–683 (2006). https://doi.org/10.1007/s00134-006-0077-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-006-0077-x