Abstract
Objective
A negative effect of hyperglycemia on clinical outcome has been proposed for patients treated under intensive care conditions. So far, however, the safety and effect of fat based nutrition regimen, especially of olive oil based emulsions, have not been systematically studied.
Design and setting
Prospective, open-labeled, randomized, pilot study in a 14-bed surgical intensive care unit.
Patients and interventions
Thirty-three consecutive, severe multiple trauma patients (injury severity score 31.6±11.5) were included; 18 (L group) received a parenteral lipid-based nutrition; 15 (G group) were given a standard parenteral glucose-based nutrition.
Measurements and results
The energy expenditure (EE) showed no difference between groups and no significant difference between the energy intake/EE ratio. The daily mean energy intake was lower in the L group (17.9±6.3 kcal/kg) than in the G group (22.3±4.2 kcal/kg). Triglycerides and nitrogen balance showed no significant differences between groups. The L group had significantly lower blood glucose (L 7.4±1.6, G 8.7±1.6 mmol/l), carbon dioxide production, and minute volume and shorter duration of mechanical ventilation (L 13.0±8.9, G 20.4±7.0 days), and stay in the ICU (L 17.9±11.2, G 25.1±7.0 days).
Conclusions
Our findings suggest a good tolerance, a decrease in blood glucose, clinically relevant shortening of ICU stay, and shorter time on mechanical ventilation for patients treated with olive oil based than with conventional glucose-heavy nutrition.
Similar content being viewed by others
Introduction
Hyperglycemia is a well known concern in multiple-trauma patients [1, 2, 3, 4, 5]. As a result of the hormonal and metabolic changes that accompany the “stress response” hyperglycemia frequently occurs in multiple trauma patients, and blood glucose levels have recently been proposed to be a main outcome predictor [1]. In the setting of multiple trauma and other severe acute disorder states hyperglycemia appears to predispose to infections, polyneuropathy, catabolism, multiorgan failure, and death [2, 3, 4, 5]. Prior work has shown a reduction in mortality and morbidity by lowering the level of blood glucose using intensive insulin therapy in patients after surgery [1]. The aim of this pilot study was to test the safety of an olive oil lipid-based vs. a conventional glucose-based parenteral/enteral nutrition regimen and its effect on blood glucose, duration of mechanical ventilation, and length of intensive care unit (ICU) stay. For lipid emulsions based on soybean oil an increased susceptibility to infections has been suggested [7, 8]. We therefore recorded infectious complications and measured the expression of human leukocyte antigen group DR (HLA-DR) on blood monocytes.
Patients and methods
The baseline characteristics of the 33 randomized patients are shown in Table 1. The study was performed between August 2001 and December 2002 with approval from the Ethics Committee of the University of Witten/Herdecke, Germany. The study design was open-label, prospective, and randomized. Informed consent was obtained from a guardian and/or relative. Inclusion criteria were major trauma and Injury Severity Score of 16 or higher, requirement of mechanical ventilation, patient age between 18 and 80 years, and admission within 24 h after trauma. Exclusion criteria were unstable hemodynamics, uncorrected lactate acidosis, known disorder of lipid metabolism, severe obesity (body mass index ≥ 40), known alcoholism, and allergy to the used lipid emulsions.
All study patients were included on the day of the trauma (study day 0). After meeting the inclusion criteria, and informed consent had been obtained, the patients were randomly assigned to receive either a lipid-based (L group) or a glucose-based (G group) nutrition. The study nutrition started on the day (study day 1) following the trauma with 50% of the energy demand, increasing to 75% on day 2, and 100% on day 3. L group patients received a parenteral lipid-based nutrition with a lipid/glucose ratio (percentage kcal/percentage kcal of nonprotein energy) of 75/25; the remaining 15 patients (G group) were given a standard parenteral glucose-based nutrition with a lipid/glucose ratio of 37/63. No other sources of glucose (i.e., intravenous solutions, minibags) were given. Both groups received 20% of their parenteral energy supply as amino acids. To avoid a high intake of n-6-fatty acids L group patients received a fat emulsion based on olive oil (ClinOleic 20%, Baxter, Germany); in the G group a standard soybean derived emulsion (Lipofundin N 20%, B. Braun, Germany) was given. The parenteral nutrition was given continuously over 24 h. Enteral nutrition was started on day 1 with a daily maximum of 5 kcal/kg during the first 6 days. The L group also received an enteral diet containing high fat based on olive oil (lipid/glucose 60/40; Glucerna, Abbott, Germany); the G group received an enteral standard diet (lipid/glucose 44/56; Fresubin HP energy, Fresenius, Germany). After day 6 the parenteral energy supply was reduced while the enteral energy was increased dependent on the patients gastrointestinal tolerability. The same enteral diet products (L group Glucerna, G group Fresubin HP energy) were given throughout the study. Gastrointestinal tolerability was assumed when gastric residual volumes were below 200 ml after a 3-h enteral nutrition intermission. Until the gastrointestinal tolerability was reached, a “trickle feed” (20 ml enteral solution plus 20 ml water) was given. The enteral nutrition was given in hourly boluses between 8 a.m. and 10 p.m.
The daily energy requirement was estimated by continuous indirect calorimetry (DeltaTrak II, Datex, Finland) for at least 8 h per day using the respirator mode. On day 1 the indirect calorimetry was started before giving nutrition support. Patients were extubated after 24 h of spontaneous breathing. After extubation both the indirect calorimetry and the mechanical ventilation ended. Because we did not use the canopy for indirect calorimetry in spontaneously breathing patients, the last measured energy requirement value was carried forward for the following days.
Blood triglycerides were measured each morning under continuous infusion conditions. Blood glucose was calculated as the mean of multiple daily measurements (minimum of three, maximum of six measurements) using point-of-care devices (AVL OMNI 9, Roche Diagnostics). Intravenous insulin was given if the blood glucose exceeded 10 mmol/l. After decreasing the blood glucose below 10 mmol/l the infusion of insulin was stopped. The given values were calculated only for days on which insulin was given. Nitrogen balance was calculated by assessing the daily N-intake and urea excretion in the 24-h urine. Carbon dioxide production and minute volume were estimated by using the continuous indirect calorimetry while the patients were on respirator therapy.
By study design the staff responsibilities for nutrition therapy and intensive care therapy were held separate. The ward physician was not involved in the nutrition therapy. In turn, all decisions regarding extubation or discharge of the patients were taken by the ward physician unaware of the nutrition regimen.
The two groups did not differ significantly in age, gender, body mass index, need for transfusion, number of surgical procedures, or clinical scores. Injury to the head and chest were main cause of prolonged artificial ventilation. Neither Abbreviated Injury Scale subscore differed significantly between groups at baseline (head: L group 3.1±1.7 and G group 2.7±2.1, p=0.57; chest: L group 2.4±1.8 and G group 2.7±1.8, p=0.73).
The expression of HLA-DR on CD14+ monocytes was evaluated on days 1, 3, 5, 7, and 14 after trauma by fluorescence activated cell scanning; for this we used antibodies labeled on a protein/fluorophore ratio of 1/1 (QuantiBRITE reagents, BD Biosciences, Heidelberg, Germany). The measurement of multilevel calibrated QuantiBRITE fluorescent beads allows the construction of a calibration curve for antigen quantification. Fifteen healthy individuals with a mean age of 32.8±2.8 years (range 23–67) served as controls.
The clinical outcome at discharge from the acute care hospital was determined by the Early Rehabilitation Barthel Index (ERI). The modified ERI assesses the need for medical or geriatric care and disability on a scale ranging from minus 325 to plus 100 points. A value of plus 100 indicates complete independence in all daily activities [9]. The mortality was recorded at 6 months after the trauma.
All values are given as mean ±standard deviation. All analyses were performed on an intention-to-treat basis. Statistical analyses used the software package SPSS 11.0 (SPSS, Munich, Germany). Graphs show mean ±standard error of the mean. Differences in baseline and outcome parameters were compared using the unpaired Student’s t test, Mann-Whitney U test, or Fisher’s exact test as appropriate.
Results
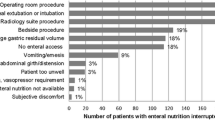
The resting energy expenditure (REE, measured on day 0) and the energy expenditure (EE) on the following days was similar in the two groups (Table 2). The total energy intake (EI) is shown in Fig. 1. No significant difference between the EI/EE ratio was observed (L 0.75±0.17, G 0.86±0.26, p=0.24). The cumulative daily total mean energy intake was significantly lower in the L group (L 17.9±6.3, G 22.3±4.2 kcal/kg, p=0.03). There was no significant difference between groups in daily enteral energy intake (L 4.5±5.2, G 5.9±4.6 kcal/kg, p=0.41). Three patients were intolerant to enteral feeding within the first 3 days (one in the L group, two in the G group). The daily parenteral glucose intake (L 0.8±0.3, G 2.0±0.6 g/kg, p<0.001) was higher in the G group. The daily parenteral lipid intake was higher in the L group (L 0.8±0.3, G 0.5±0.1 g/kg, p<0.001). The daily intravenous amino acid supply showed no significant difference between groups (L 0.7±0.2, G 0.8±0.2 kcal/kg, p=0.17). Eight patients were discharged from the ICU before day 14 (seven in the L group, one in the G group). Reasons for discharge were clinical improvement (six in the L group, one in the G group). One patient in the L group was excluded from the study after disclosure of a fat metabolism disorder (protocol violation). One patient in the L group died on day 3. The number of treated patients at each day is indicated in the graphs.
Mean blood glucose levels in the L group were significantly lower than in the G group (Table 2). Whereas in the L group blood glucose was stable at approx. 7 mmol/l, glucose values increased in the G group after day 4 to stay above 9 mmol/l until day 11 (Fig. 2). In the L group 5 of 18 patients required insulin and in the G group 9 of 15 (p=0.09). The mean use of insulin in the L group was 55.8±46.0 IE and in the G group 65.3±42.0 IE (p=0.70). One diabetic patient in the L group had a history of 90 IE insulin per day before trauma. Of the daily recorded minimal blood glucose measurements (n=380 in the L group, n=412 in the G group) one value in an L group patient was under 3.5 mmol/l.
Blood triglyceride levels did not differ significantly between groups (Table 2). Nitrogen balance was negative in both groups and showed no significant difference (Table 2). The carbon dioxide production, total respiratory quotient, and minute volume were significantly reduced in the lipid group (Table 2). Four patients in the lipid and two in the glucose group had no indirect calorimetry. No patient had to be excluded from indirect calorimetry assessment for reasons of high fraction of inspired oxygen.
The duration of mechanical ventilation and length of stay in the ICU was significantly shorter in the L group. Infectious complications were less frequent in the L group (Fig. 3). The length of stay in the acute care hospital did not differ significantly between groups (Table 3). The modified ERI at discharge from the acute care hospital showed no significant difference between groups (L 2.5±120, G 25±64, p=0.99). Within 6 months after trauma four patients in the L group and one in the G group died (p=0.35). One patient in the L group died due to his severe head trauma on day 3. Another patient in the L group died due to sepsis after bronchopneumonia on day 19. Two patients in the L group died on day 41 and 50, and the patient in the G group died on day 47 at a nursing home. In the latter three cases the cause of death was unspecified.
Monocytic HLA-DR on admission was significantly lower than in controls (9,890±4,010 ABC vs. 29,881±8,060 ABC, p<0.001). There was no difference between groups in monocytic HLA-DR expression at admission. The time course of monocytic HLA-DR expression is shown in Fig. 4; monocytic HLA-DR expression increased to values in the lower range of healthy volunteers on day 14. Patients in the L group showed a faster, albeit statistically nonsignificant, improvement in monocytic HLA-DR than those in the G group.
HLA-DR expression on CD14+ blood monocytes in the lipid (circles) and the glucose group (triangle). All results are expressed as antibodies bound per cell (ABC, mean ±SEM). Gray area Values of age-matched healthy volunteers with 25th percentile (P25), 50th percentile (P50), and 75th percentile (P75); n number of patients
Discussion
Our findings show a good tolerance of an olive oil based high lipid nutrition regimen in multiple trauma patients. Further, our data confirm prior reports showing a decrease in blood glucose and CO2 production [10, 11], good tolerance [10, 11, 12, 13], and reduced time on mechanical ventilation [10] under lipid-based nutrition in ICU treated patients. Furthermore, our findings of abbreviated ICU times may support the notion of a positive clinical effect of lower blood glucose regimen [1].
The main causes of ventilator support in multiple trauma patients are chest/lung and craniocerebral traumata, these have a similar frequency and often coexisting in our patients. The differential effects of central and/or peripheral respiratory failure could therefore not be determined in our study. Prior work has suggested an improved pulmonary function especially in patients with preexisting lung disease under a high-fat, low-carbohydrate diet [14, 15]. In general the objectives of mechanical ventilation in severe trauma patients are an adequate delivery of oxygen and elimination of CO2. Elevated CO2 production is accompanied by an increase in minute ventilation [16], higher risk of pulmonary overdistention, higher mortality, and longer time on respirator [17]. Therefore a lower CO2 production can be considered beneficial. In our study the observed decrease in CO2 production was 14% (95% confidence interval, CI, 2–26%), and the decrease in minute volume was 13% (95% CI 1–25%). Assuming a total combustion of 2,000 kcal (1,500 parenteral, 500 enteral), according to our study protocol per day CO2 production would have been 325 and 349 l in L and G group patients, respectively (expected difference of 7%, 95% CI 6–8%). Given the difference in expected vs. observed point estimates (however, not significant by 95% CI) the effect of factors such as severity and pattern of injury, body temperature, caloric intake and use of sedation as well as an effect of the lower, i.e., hypocaloric, energy intake in the L group may be considered.
Despite lower glucose levels in our L group cases (as expected with lower glucose intakes), blood glucose values still ranged above normal, leaving open the possibility for further beneficial effects of more forceful approaches to blood glucose normalization in severe trauma cases. Further studies using lipid-based nutrition also found slightly elevated blood glucose values ranging from 6 to 8 mmol/l (compared to glucose-based nutrition with values from 8 to 10 mmol/l) [11, 13]. Studies using intensive insulin therapy achieved blood glucose values below 6 mmol/l [1]; i.e., within the range of normal. Furthermore, the work by Tappy et al. [11, 18] has shown in critical illness an independence of endogenous glucose production from parenteral [11] and enteral [18] glucose supply.
Lipid emulsions have been suggested to influence inflammation processes [19], affecting the level of metabolic activation including CO2 production. The underlying mechanism is due mainly to the type and quantity of administered fatty acids which act as precursor in the eicosanoid metabolism. Modifying the n-6/n-3 ratio has both rapid and persistent effects on inflammatory and immune responses [20]. The lipid emulsion used in our L group patients is containing 80% olive oil and 20% soybean oil. It was developed with the rationale to decrease the polyunsaturated fatty acids content of soybean emulsions and replace it with mainly oleic acid (C18:1 n-9). This preparation is considered not to affect membrane composition and therefore to have little effect on eicosanoid production and the immune response [21] followed by a reduced need for antioxidants. In preterm infants, who are often exposed to oxidative stress, an improvement in the antioxidant status using an olive oil based lipid emulsion has been shown [22]. Our nutrition regimen in the L group patients resulted in a significantly increased monounsaturated fatty acid intake accompanied by a decreased polyunsaturated fatty acid intake.
The lower rate of infectious complications in the L group is in contrast to previous studies mainly using soybean derived lipid emulsion [7, 23, 24]. The authors offer the argument that the known negative immune effects of n-6 fatty acids [8, 25] may be avoided with the n-9 fatty acids used in our study.
Since the principal aim of this pilot study was to investigate the tolerance of such a regimen, we decided to investigate the monocytic HLA-DR expression—a general measure of immunodeficiency after major trauma—instead of analyzing fatty acid metabolites. Prior work proposed an association of low monocytic HLA-DR expression with the risk of infection and clinical outcome [26]. In our pilot study the difference in improvement in monocytic HLA-DR expression between groups, although not significant, suggests a possible positive effect of olive oil lipid-based nutrition [19, 27].
Prior work suggested negative effects of long-chain triglyceride emulsions on the reticuloendothelial system function [28], an induction of hypertriglyceridemia, and adverse impacts on pulmonary gas diffusion [29]. The disturbances in reticuloendothelial system function and hypertriglyceridemia have been considered to be pronounced in critical illness and have been associated with the rapid and intermittent infusion of the lipid emulsions [30]. With the use of indirect calorimetry for the determination of the daily energy goal and a continuous lipid infusion regimen we minimized the risk of an overdose of intravenous lipids. Despite the high percentage of lipid calories in our olive-oil based nutritional regimen, the mean hourly lipid doses of 0.03 g/kg given in the L group were lower than the 0.11 g/kg lipid infusion doses considered toxic by prior authors [31]. The negative changes in pulmonary gas diffusion under the infusion of lipid emulsions have been linked to the supplementation of prostaglandin precursors as linoleic acid [30]. One goal of our study was to reduce the linoleic acid intake.
Regarding clinical outcome the modified ERI at discharge from the acute care hospital did not show a significant difference between groups. Our mortality point estimate of 12% (95% CI 1–23%) was similar to the 10% given in the trauma register of the German Society of Traumatology [32].
Our findings suggest a good tolerance of a high lipid nutrition based on olive-oil resulting in a decrease in blood glucose, a clinically relevant shortening of ICU stay, and shortening of the time on mechanical ventilation for severe trauma patients treated with lipid-based than with conventional glucose-heavy nutrition regimen. Further studies in larger patient samples are needed to test the effect of lipid-based nutrition on clinical outcome.
References
Berghe G van den, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P, Bouillon R (2001) Intensive insulin therapy in the surgical intensive care unit. N Engl J Med 345:1359–1367
Fietsam R Jr, Bassett J, Glover JL (1991) Complications of coronary artery surgery in diabetic patients. Am Surg 57:551–557
Gore DC, Chinkes DL, Hart DW, Wolf SE, Herndon DN, Sanford AP (2002) Hyperglycemia exacerbates muscle protein catabolism in burn-injured patients. Crit Care Med 30:2438–2442
Ortiz A, Ziyadeh FN, Neilson EG (1997) Expression of apoptosis-regulatory genes in renal proximal tubular epithelial cells exposed to high ambient glucose and in diabetic kidneys. J Investig Med 45:50–56
Said G, Goulon-Goeau C, Slama G, Tchobroutsky G (1992) Severe early-onset polyneuropathy in insulin-dependent diabetes mellitus. A clinical and pathological study. N Engl J Med 326:1257–1263
Huschak G, zur Nieden K, Stuttmann R (2003) Effects of lipid based nutrition on clinical outcome of patients after severe trauma. Clin Nutr 22 Suppl 1:S1–114
Heyland DK, MacDonald S, Keefe L, Drover JW (1998) Total parenteral nutrition in the critically ill patient: a meta-analysis. JAMA 280:2013–2019
Yaqoob P (1998) Lipids and the immune response. Curr Opin Clin Nutr Metab Care 1:153–161
Schönle PW (1995) The Early Rehabilitation Barthel Index—an early rehabilitation-oriented extension of the Barthel Index. Rehabilitation (Stuttg) 34:69–73
al Saady NM, Blackmore CM, Bennett ED (1989) High fat, low carbohydrate, enteral feeding lowers PaCO2 and reduces the period of ventilation in artificially ventilated patients. Intensive Care Med 15:290–295
Tappy L, Schwarz JM, Schneiter P, Cayeux C, Revelly JP, Fagerquist CK, Jequier E, Chiolero R (1998) Effects of isoenergetic glucose-based or lipid-based parenteral nutrition on glucose metabolism, de novo lipogenesis, and respiratory gas exchanges in critically ill patients. Crit Care Med 26:860–867
Kohlhardt SR, Smith RC, Kee AJ (1994) Metabolic response to a high-lipid, high-nitrogen peripheral intravenous nutrition solution after major upper-gastrointestinal surgery. Nutrition 10:317–326
Kohlhardt SR, Smith RC, Kee AJ (1994) Metabolic evaluation of a 75% lipid/25% glucose high nitrogen solution for intravenous nutrition. Eur J Surg 160:335–344
Cai B, Zhu Y, Ma Y, Xu Z, Zao Y, Wang J, Lin Y, Comer GM (2003) Effect of supplementing a high-fat, low-carbohydrate enteral formula in COPD patients. Nutrition 19:229–232
Sheman MS (2001) Parenteral nutrition and cardiopulmonary disease. In: Rombeau JL, Rolandelli RH (eds) Clinical nutrition: parenteral nutrition. Saunders, Philadelphia, pp 335–352
Weinberger SE, Schwartzstein RM, Weiss JW (1989) Hypercapnia. N Engl J Med 321:1223–1231
The Acute Respiratory Distress Syndrome Network (2000) Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 342:1301–1308
Tappy L, Berger M, Schwarz JM, McCamish M, Revelly JP, Schneiter P, Jequier E, Chiolero R (1999) Hepatic and peripheral glucose metabolism in intensive care patients receiving continuous high- or low-carbohydrate enteral nutrition. JPEN J Parenter Enteral Nutr 23:260–267
Carpentier YA, Dupont IE (2000) Advances in intravenous lipid emulsions. World J Surg 24:1493–1497
Endres S, Ghorbani R, Kelley VE, Georgilis K, Lonnemann G, van der Meer JW, Cannon JG, Rogers TS, Klempner MS, Weber PC (1989) The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N Engl J Med 320:265–271
Yaqoob P, Knapper JA, Webb DH, Williams CM, Newsholme EA, Calder PC (1998) Effect of olive oil on immune function in middle-aged men. Am J Clin Nutr 67:129–135
Göbel Y, Koletzko B, Böhles HJ, Engelsberger I, Forget D, Le Brun A, Peters J, Zimmermann A (2003) Parenteral fat emulsions based on olive and soybean oils: a randomized clinical trial in preterm infants. J Pediatr Gastroenterol Nutr 37:161–167
Battistella FD, Widergren JT, Anderson JT, Siepler JK, Weber JC, MacColl K (1997) A prospective, randomized trial of intravenous fat emulsion administration in trauma victims requiring total parenteral nutrition. J Trauma 43:52–58
Garrel DR, Razi M, Lariviere F, Jobin N, Naman N, Emptoz-Bonneton A, Pugeat MM (1995) Improved clinical status and length of care with low-fat nutrition support in burn patients. JPEN J Parenter Enteral Nutr 19:482–491
Alexander JW (1998) Immunonutrition: the role of omega-3 fatty acids. Nutrition 14:627–633
Hershman MJ, Cheadle WG, Wellhausen SR, Davidson PF, Polk HC Jr (1990) Monocyte HLA-DR antigen expression characterizes clinical outcome in the trauma patient. Br J Surg 77:204–207
Torrinhas RS, Gidlund M, Goto H, Sales M, Oliviera P, Jacintho TM, Waitzberg DL (2002) Olive oil based lipid emulsion does not alter the expression of surface molecules with immunological functions on human monocytes/macrophages (MO). Clin Nutr Suppl 21:P-040
Seidner DL, Mascioli EA, Istfan NW, Porter KA, Selleck K, Blackburn GL, Bistrian BR (1989) Effects of long-chain triglyceride emulsions on reticuloendothelial system function in humans. JPEN J Parenter Enteral Nutr 13:614–619
Hwang TL, Huang SL, Chen MF (1990) Effects of intravenous fat emulsion on respiratory failure. Chest 97:934–938
Driscoll DF, Adolph M, Bistrian BR (2001) Lipid emulsions in parenteral nutrition. In: Rombeau JL, Rolandelli RH (eds) Clinical nutrition: parenteral nutrition. Saunders, Philadelphia, pp 35–59
Klein S, Miles JM (1994) Metabolic effects of long-chain and medium-chain triglyceride emulsions in humans. JPEN J Parenter Enteral Nutr 18:396–397
Bardenheuer M, Obertacke U, Waydhas C, Nast-Kolb D (2000) Epidemiologie des Schwerverletzten. Eine prospektive Erfassung der präklinischen und klinischen Versorgung. Unfallchirurg 103:355–363
Acknowledgements
Parts of the manuscript were presented at the 25th Congress of the European Society of Parenteral and Enteral Nutrition (ESPEN) at Cannes, France [6]
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was funded by the German Berufsgenossenschaften (Employers’ Liability Insurance Association) and Baxter Deutschland GmbH.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Huschak, G., zur Nieden, K., Hoell, T. et al. Olive oil based nutrition in multiple trauma patients: a pilot study. Intensive Care Med 31, 1202–1208 (2005). https://doi.org/10.1007/s00134-005-2727-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-005-2727-9