Summary
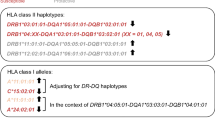
In Finland the haplotype A2, Cw1, B56, DR4, DQ8 is the third most common haplotype in insulin-dependent diabetic (IDDM) patients and has the highest haplotype-specific absolute risk for IDDM. Cw1, B56, DR4, DQ8 haplotypes containing HLA-A alleles other than A2 are infrequent in the population and are not associated with IDDM. Comparison of the A2 and non-A2 haplotypes at the DNA level showed that they were identical at HLA-B,-DR, and -DQ loci. Evidence that class I alleles confer susceptibility to IDDM was obtained from the two HLA-C, -B, -DR and -DQ haplotypes most frequently found in IDDM patients in Finland. A24, A3 and A2 on the Cw3, B62, DR4, DQ8 haplotype, and A28, A2 and A1 on the Cw7, B8, DR3, DQ2 were all found to be associated with IDDM. In Finland these seven haplotypes, including A2, Cw1, B56, DR4, DQ8, account for 33% of diabetic haplotypes and 10.3% of non-diabetic haplotypes (p<0.00001). The contribution of the class I region to IDDM susceptibility was also apparent in those IDDM patients lacking the disease-predisposing class II alleles. Significantly more non-DR3/non-DR4 IDDM patients (47 of 55) possessed two of the IDDM-associated HLA-A alleles compared to non-DR3/non-DR4 control subjects (40 of 58; p=0.038). Moreover, IDDM patients confirmed by oligotyping as unable to form a ‘diabetes-susceptibility’ DQ heterodimer, tended to possess two diabetes-associated HLA-A alleles (12 of 13) compared to control subjects (12 of 20; p=0.056).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- IDDM:
-
Insulin-dependent diabetes mellitus
- NIDDM:
-
non-insulin-dependent diabetes mellitus
- PCR:
-
polymerase chain reaction
- SSO:
-
sequence specific oligonucleotide
- TNF:
-
tumour necrosis factor
References
Cudworth AG, Woodrow JC (1976) Genetic susceptibility in diabetes mellitus: analysis of the HLA association. BMJ 2: 846–848
Sachs JA, Cudworth AG, Jaraquemada D, Gorsuch AN, Festenstein H (1980) Type 1 diabetes and the HLA-D locus. Diabetologia 18: 41–43
Raum D, Awdeh Z, Yunis EJ, Alper CA, Gabbay KH (1984) Etended major histocompatibility complex haplotypes in type 1 diabetes mellitus. J Clin Invest 74: 449–454
Tienari PJ, Tuomilehto-Wolf E, Tuomilehto J, Peltonen L, and the DIME study group (1992) HLA haplotypes in type 1 (insulin-dependent) diabetes mellitus: molecular analysis of the HLA-DQ locus. Diabetologia 35: 254–260
Hägglöf B, Holmgren G, Holmlund G, Lindblom B, Olaisen B, Teisberg P (1986) Studies of HLA, factor B(Bf), complement C2 and C4 haplotypes in type 1 diabetic and control families from Northern Sweden. Hum Hered 36: 201–212
Ronningen KS, Spurkland A, Iwe T, Vartdal F, Thorsby E (1991) Distribution of HLA-DRB1, -DQA1 and -DQB1 alleles and DQA1-DQB1 genotypes among Norwegian patients with insulin-dependent diabetes mellitus. Tissue Antigens 37: 105–111
Wolf E (1989) Different linkage disequilibria in type 1 diabetic and ‘healthy’ HLA haplotypes. Pediatr Adolesc Endocrinol 18: 270–274
Contu L, Deschamps I, Lestradet H et al. (1982) HLA haplotype study of 53 juvenile insulin-dependent diabetic (IDD) families. Tissue Antigens 20: 123–140
Cambon-de Mouzon A, Ohayon E, Hauptmann G (1982) HLA-A, B, C, DR antigens, Bf, C4 and glyoxylase 1 (GLO) polymorphism in French Basques with insulin-dependent diabetes mellitus (IDDM). Tissue Antigens 19: 366–379
De Jongh BM, Bruining GJ, GMTH Schreuder et al. (1984) HLA and GM in insulin-dependent diabetes in the Netherlands: report on a combined multiplex family and population study. Hum Immunol 10: 5–21
Tuomilehto-Wolf E, Podar T, Adojan B, Kalits I, Tuomilehto J (1991) Can the difference in incidence of type 1 diabetes between Estonia and Finland be partly explained by genetic reasons? Diabetologia 24 [Suppl 2]: A65 (Abstract)
Rjasanowski I, Vogt L, Michaelis D, Keilacker H, Kohnert K (1993) The frequency of diabetes in children of type 1 diabetic parents. Diabete Metab Paris 19: 173–177
Genetic analysis of complex traits: insulin-dependent diabetes mellitus and affective disorders 1989 Proceedings of a workshop, Chantilly, France, September 2–5, 1987. Genet Epidemiol 6: 1–310
Hors J, Deschamps I, Schuler E (1986) Genetic Analysis Workshop IV: some clinical, pathological and etiological aspects of two HLA-related diseases. Genet Epidemiol 6 [Suppl 1]: 291–297
Thomson G, Robinson WP, Kuhner MK et al. (1988) Genetic heterogeneity, modes of inheritance, and risk estimates for a joint study of Caucasians with insulin-dependent diabetes mellitus. Am J Hum Genet 43: 799–816
Khalil I, d' Auriol L, Gobet M et al. (1990) A combination of HLA-DQβ Asp 57-negative and HLA-DQα Arg 52 confers susceptibility to insulin-dependent diabetes mellitus. J Clin Invest 85: 1315–1319
Tuomilehto-Wolf E, Tuomilehto J, Hitman GA and the DiMe study group (1992) DQA1 and DQB1 heterodimers in insulin-dependent diabetes mellitus. A genetic-epidemiology study in Finland. Ann Med 24: 533–538
Tuomilehto J, Lounamaa R, Tuomilehto-Wolf E et al. (1992) Epidemiology of childhood diabetes mellitus in Finland — background of a nationwide study of type 1 (insulindependent) diabetes mellitus. Diabetologia 35: 70–76
Kawasaki ES, Wang AM (1989) Detection of gene expression. In: Ehrlich HA (ed) PCR technology principles and applications for DNA amplification. Stockton Press, New York, pp 89–97
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. 2nd edn. Cold Spring Harbor Laboratory Press, New York
Balnaves ME, Nasioulas S, Dahl H-HM, Forrest S (1991) Direct PCR from CVS and blood lysates for detection of cystic fibrosis and Duchenne muscular dystrophy deletions. Nucleic Acids Res 19: 1155
Tsuji K, Aizawa M, Sasazuki T (eds) (1992) HLA 1991. Proceedings of the eleventh international histocompatibility workshop and conference. Oxford Science Publications
Nomura N, Ota M, Tsuji K, Inoka H (1991) HLA-DQB1 genotyping by a modified PCR-RFLP method combined with group-specific primers. Tissue Antigens 38: 53–59
Maeda M, Murayama N, Ishii H et al. (1989) A simple and rapid method for HLA-DQA1 genotyping by digestion of PCR-amplified DNA with allele specific restriction enzymes. Tissue Antigens 34: 290–298
Nedospasov SA, Udalova IA, Kuprash DV, Turetskaya RL (1991) DNA sequence polymorphism at the human tumor necrosis factor (TNF) locus. Numerous TNF/lymphotoxin alleles tagged by two closely linked microsatellites in the upstream region of the lymphotoxin (TNF-β) gene. J Immunol 147: 1053–1059
Alper CA, Boenisch T, Watson L (1972) Genetic polymorphism in human glycine-rich beta-glycoprotein. J Exp Med 135: 68–80
Awdeh ZL, Alper CA (1980) Inherited structural polymorphism of the fourth component of human complement. Proc Natl Acad Sci USA 77: 3576–3580
Zemmour J, Parham P (1991) HLA class I nucleotide sequences, 1991. Tissue Antigens 37: 174–180
Hildebrand WH, Madrigal JA, Little AM, Parham P (1992) HLA-Bw22: a family of molecules with identity to HLA-B7 in the α1-helix. J Immunol 148: 1155–1162
Jongeneel CV, Briant L, Udalova IA, Sevin A, Nedospasov SA, Cambon-Thomsen A (1991) Extensive genetic polymorphism in the human tumor necrosis factor region and relation to extended HLA haplotypes. Proc Natl Acad Sci USA 88: 9717–9721
Todd JA, Bell JI, McDevitt HO (1987) HLA-DQβ gene contibutes to susceptibility and resistance to insulin-dependent diabetes mellitus. Nature (London) 329: 599–604
Bottazzo GF, Dean BM, McNally JM, MacKay EH, Swift PGF, Gamble DR (1985) In situ characterization of autoimmune phenomena and expression of HLA molecules in the pancreas in diabetic insulitis. N Engl J Med 313: 353–360
Sibley RK, Sutherland DER (1987) Pancreas transplantation an immunohistologic and histopathologic examination of 100 grafts. Am J Pathol 128: 151–170
Santamaria P, Nakhleh RE, Sutherland DE, Barbosa JJ (1992) Characterization of T lymphocytes infiltrating human pancreas allograft affected by isletitis and recurrent diabetes. Diabetes 41: 53–61
Hayakawa M, Yokono K, Nagata M et al. (1991) Morphological analysis of selective destruction of pancreatic β-cells by cytotoxic T lymphocytes in NOD mice. Diabetes 40: 1210–1217
Nagata M, Yoon J-W (1992) Studies on autoimmunity for T-cell mediated β-cell destruction. Distinct difference in β-cell destruction between CD4+ and CDS+ T-cell clones derived from lymphocytes infiltrating the islets of NOD mice. Diabetes 41: 998–1008
Nagata M, Yokono K, Hayakawa M et al. (1989) Destruction of pancreatic islet cells by cytotoxic T lymphocytes in nonobese diabetic mice. J Immunol 143: 1155–1162
Taki T, Nagata M, Ogawa W et al. (1991) Prevention of cyclophosphamide-induced and spontaneous diabetes in NOD/Shi/Kbe mice by anti-MHC class I Kd monoclonal antibody. Diabetes 40: 1203–1209
Foulis AK, Farquharson MA, Hardman R (1987) Aberrant expression of class II major histocompatibility complex molecules by B cells and hyperexpression of class I major histocompatibility complex molecules by insulin containing islets in type 1 (insulin-dependent) diabetes mellitus. Diabetologia 30: 333–343
Faustman D, Coe C (1991) Prevention of xenograft rejection by masking donor HLA class I antigens. Science 252: 1700–1703
Faustman D, Li X, Lin HY et al. (1991) Linkage of faulty major histocompatibility complex class I to autoimmune diabetes. Science 254: 1756–1761
Fu Y, Nathan DM, Li F, Li X, Faustman DL (1993) Defective major histocompatibility complex class I expression on lymphoid cells in autoimmunity. J Clin Invest 91: 2301–2307
Van der Poel KJ, Pool J, Goulmy E, van Rood JJ (1983) Differential recognition of the serologically defined HLA-A2 antigen by allogeneic cytotoxic T cells. II. Definition of three HLA-A2 subtypes by CTLs. Immunogenetics 17: 599–608
Sheehy MJ, Scharf SJ, Rowe JR et al. (1988) A diabetessusceptible HLA haplotype is best defined by a combination of HLA-DR and -DQ alleles. J Clin Invest 83: 830–835
Kobayashi T, Tamemoto K, Nakanishi K, Kato N, Okubo M (1993) Immunogenetic and clinical characterization of slowly progressive IDDM. Diabetes Care 16: 780–788
Nakanishi K, Kobayashi T, Murase T et al. (1993) Association of HLA-A24 with complete β-cell destruction in IDDM. Diabetes 42: 1086–1093
Williams RC, Knowler WC, Butler WJ (1981) HLA-A2 and type 2 (insulin-dependent) diabetes mellitus in Pima Indians: an association of allele frequency with age. Diabetologia 21: 460–463
Briggs BR, Jackson WPU, Dutoit ED, Botha MC (1980) The histocompatibility (HLA) antigen distribution in diabetes in southern African blacks (Xhosa). Diabetes 29: 68–71
Tuomilehto-Wolf E, Tuomilehto J, Hitman GA et al. (1993) Genetic susceptibility to non-insulin dependent diabetes mellitus and glucose intolerance are located in the HLA region. BMJ 307: 155–159
Author information
Authors and Affiliations
Consortia
Rights and permissions
About this article
Cite this article
Fennessy, M., Metcalfe, K., Hitman, G.A. et al. A gene in the HLA class I region contributes to susceptibility to IDDM in the Finnish population. Diabetologia 37, 937–944 (1994). https://doi.org/10.1007/BF00400951
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00400951