Abstract
Aims/hypothesis
We hypothesised that progression of islet autoimmunity and type 1 diabetes mellitus differs among races/ethnicities in at-risk individuals.
Methods
In this study, we analysed the data from the Type 1 Diabetes TrialNet Pathway to Prevention Study. We studied 4873 non-diabetic, autoantibody-positive relatives of individuals with type 1 diabetes followed prospectively (11% Hispanic, 80.9% non-Hispanic white [NHW], 2.9% non-Hispanic black [NHB] and 5.2% non-Hispanic other [NHO]). Primary outcomes were time from single autoantibody positivity confirmation to multiple autoantibody positivity, and time from multiple autoantibody positivity to type 1 diabetes mellitus diagnosis.
Results
Conversion from single to multiple autoantibody positivity was less common in Hispanic individuals than in NHW individuals (HR 0.66 [95% CI 0.46, 0.96], p = 0.028) adjusting for autoantibody type, age, sex, Diabetes Prevention Trial Type 1 Risk Score and HLA-DR3-DQ2/DR4-DQ8 genotype. In participants who screened positive for multiple autoantibodies (n = 2834), time to type 1 diabetes did not differ by race/ethnicity overall (p = 0.91). In children who were <12 years old when multiple autoantibody positivity was determined, being overweight/obese had differential effects by ethnicity: type 1 diabetes risk was increased by 36% in NHW children (HR 1.36 [95% CI 1.04, 1.77], p = 0.024) and was nearly quadrupled in Hispanic children (HR 3.8 [95% CI 1.6, 9.1], p = 0.0026). We did not observe this interaction in participants who were ≥12 years old at determination of autoantibody positivity, although this group size was limited. No significant differential risks were observed between individuals of NHB and NHW ethnicity.
Conclusions/interpretation
The risk and rate of progression of islet autoimmunity were lower in Hispanic compared with NHW at-risk individuals, while significant differences in the development of type 1 diabetes were limited to children <12 years old and were modified by BMI.
Similar content being viewed by others

Introduction
Type 1 diabetes mellitus is a chronic autoimmune condition characterised by beta cell destruction leading to insulin deficiency. Studies of the natural history and pathogenesis of type 1 diabetes have shown that it is a disease continuum with variable progression along well-defined stages: presymptomatic beta cell autoimmunity with normoglycaemia; presymptomatic beta cell autoimmunity with dysglycaemia and symptomatic beta cell autoimmunity with dysglycaemia [1]. A wealth of data has been generated on genetic, immunological and metabolic risk factors that enable us to predict type 1 diabetes risk and design studies to intervene early in the autoimmune process, before the onset of symptoms. In genetically susceptible children positive for multiple autoantibodies, the 10 year risk of developing type 1 diabetes is 70%, with lifetime risk reaching 100% [2]. A predictive score (the Diabetes Prevention Trial–Type 1 Risk Score [DPTRS]) has been proposed to estimate the type 1 diabetes risk in at-risk individuals [3].
The growing public health impact of studies examining racial/ethnic differences is underscored by recent data demonstrating that the increase in type 1 diabetes incidence disproportionally affects racial and ethnic minorities [4]. However, much of the knowledge on type 1 diabetes pathogenesis stems from studies primarily conducted in the non-Hispanic white (NHW) population and generalisability to other races/ethnicities has not been established. The incidence of type 1 diabetes in children varies by race/ethnicity (e.g. the SEARCH study reported 27, 19 and 14.8 new incidences per 100,000 person-years in 2012, respectively, in NHW, non-Hispanic black [NHB] and Hispanic participants [4]). While a limited number of studies showed that there are significant racial/ethnic differences in genetic, immunological, metabolic and clinical characteristics [5,6,7,8,9,10,11,12,13], the risk and rate of progression of islet autoimmunity and type 1 diabetes development have not been compared among different racial/ethnic groups. A full understanding of these and associated factors may inform the design of future prediction models and prevention trials and, eventually, clinical care.
Type 1 Diabetes TrialNet is a National Institutes of Health (NIH)-funded international consortium of clinical research centres aiming to prevent or delay type 1 diabetes. Relatives of individuals with type 1 diabetes are offered screening for the presence of islet autoantibodies and, if positive, enrolment in the Pathway to Prevention (PTP) study; if eligible, participation in prevention studies is offered [14]. Inclusion of an increasing number of individuals of minority racial/ethnic background provided us with a unique opportunity to compare the natural course prior to development of type 1 diabetes in those of Hispanic and NHW ethnicity.
We hypothesised that the progression of islet autoimmunity and type 1 diabetes significantly differs among races/ethnicities in at-risk individuals. This study aimed to compare the rates and risk factors of progression of islet autoimmunity and type 1 diabetes development among races/ethnicities in at-risk individuals.
Methods
Design and settings
We analysed data from the Type 1 Diabetes TrialNet PTP study. The TrialNet PTP study screened relatives of individuals with type 1 diabetes with the aim of identifying participants for monitoring and/or prevention studies. First-degree relatives (1–45 years old) and second-degree relatives (1–20 years old) of individuals with type 1 diabetes were eligible for PTP screening; of note, due to rescreen guidelines and allowable timeframes, PTP participants could be identified as autoantibody positive after 45 years of age. Eligible relatives were tested for the presence of islet autoantibodies, including glutamic acid decarboxylase 65 (GAD65) autoantibody, islet antigen 2 (IA-2) autoantibody and micro-insulin autoantibody (mIAA), followed by islet cell autoantibody (ICA) if they were positive for ≥1 autoantibody(ies) at the initial screening test [15]. Additionally, zinc transporter 8 (ZnT8) autoantibody measurement was performed consistently from 2012 onwards in participants with ≥1 positive autoantibody(ies) at the initial screening test [16]. Participants who were negative for all tested autoantibodies were eligible for yearly rescreening until 18 years of age. Participants confirmed positive for a single autoantibody on a consecutive visit within 1 year were defined as single confirmed autoantibody positive. Those positive for two or more autoantibodies at any screening or follow-up were defined as multiple autoantibody positive. Single confirmed autoantibody-positive and multiple autoantibody-positive participants were offered enrolment in ‘monitoring’ and, if eligible and interested, prevention studies. Baseline risk assessments included OGTT, HbA1c measurement and HLA typing. Participants positive for multiple autoantibodies were monitored semi-annually throughout the PTP; single confirmed autoantibody-positive participants were monitored semi-annually until 2012, and then annually. This monitoring includes OGTT, HbA1c measurement and autoantibody testing. Details of the screening and follow-up processes have previously been described [15, 16]. All participants and/or their parents provided written informed consent and assent, as appropriate, approved by local Institutional Review Boards.
Inclusion and exclusion criteria
Between 22 March 2004 and 31 July 2017, 182,145 relatives were screened in the TrialNet PTP study at 21 clinical centres and approximately 100 collaborating clinical sites in the USA, Canada, UK, Finland, Italy, Germany, Australia and New Zealand. A total of 5703 autoantibody-positive participants who had at least one follow-up visit were identified in the TrialNet PTP–Monitoring Cohort. Exclusion criteria included fasting blood glucose <2.8 mmol/l or ≥7 mmol/l, 2 h OGTT blood glucose ≥11.1 mmol/l, type 1 diabetes at first monitoring visit, missing fasting or 2 h OGTT blood glucose data and, for the current analysis, unknown or missing ethnicity data. Individuals with glucose <2.8 mmol/l were excluded because of the potential for data quality problems and those with fasting glucose ≥7 mmol/l and 2 h OGTT glucose ≥11.1 mmol/l were excluded because of suspected type 1 diabetes.
Race and ethnicity categorisation
Race and ethnicity categories were based on self-report and on standard NIH classifications and definitions [17]. Individuals who listed more than one race were categorised as multiracial. We evaluated individuals based on these NIH-defined groups and also on composite race/ethnicity groups. Specifically, participants were assigned to one of the following four racial/ethnic groups: Hispanic, NHW, NHB and non-Hispanic other [NHO]. Non-Hispanic multiracial participants were included in the ‘NHO’ category.
Anthropometric measures and laboratory analyses
BMI
BMI was calculated using data from the first monitoring visit. BMI percentiles (BMI%iles) were calculated for all participants ≥2 years old. For adults over 20 years old, BMI%iles were calculated by imputing 20 as their age to be able to evaluate BMI%iles as a continuous measure across all participants. Classification of an individual as overweight was defined as a BMI ≥85th but <95th percentile, and obesity was defined as a BMI ≥95th percentile adjusting for age and sex according to Centers for Disease Control and Prevention criteria. Because of the very limited number of underweight participants, all participants with a BMI <85th percentile were considered to be lean.
HLA typing
HLA genotyping was performed at TrialNet HLA Laboratory at the Barbara Davis Center, which receives whole blood from clinical sites and extracts DNA using the AutoGen QuickGene-610 instrument. In this analysis, participants were classified by the presence or absence of the highest risk genotype (i.e. DR3-DQ2 [DRB1*03:01–DQA1*05:01–DQB1*02:01] and DR4-DQ8 [DQA1*03:01–DQB1*03:02 with DRB1*04:01, DRB1*04:02 or DRB1*04:05]). Further information on HLA typing is provided in electronic supplemental material (ESM) Methods.
Autoantibody assays
GAD65, IA-2, mIAA and ZnT8 autoantibodies were measured by radioimmunoassay in the TrialNet Core Laboratory at the Barbara Davis Center for Childhood Diabetes in Denver, CO, USA. During the 2015 Islet Autoantibody Standardization Program Workshop, respective sensitivities and specificities were 52% and 100% for mIAA, 82% and 99% for GAD65 autoantibody, 72% and 100% for IA-2 autoantibody and 70% and 97% for ZnT8 autoantibody [18]. ICA positivity was tested by indirect immunofluorescence in the Diagnostic Referral Laboratories at the University of Florida. An ICA value greater than 5 Juvenile Diabetes Foundation units was considered positive [19].
OGTT
The glycaemic status of the participants was tested with an OGTT (oral glucose dose 1.75 g/kg, maximum 75 g) after an overnight fast. C-peptide (nmol/l) and glucose (mmol/l) measurements were performed in the fasted state and at 30, 60, 90 and 120 min. The trapezoid method was used to calculate the AUC (nmol/l × min) for C-peptide.
Diagnosis of diabetes
Diabetes was diagnosed according to TrialNet Natural History Study of the Development of Type 1 Diabetes Protocol (TrialNet Protocol TN01): fasting plasma glucose ≥7.0 mmol/l, 2 h plasma glucose during an OGTT ≥11.1 mmol/l, a random plasma glucose ≥11.1 mmol/l with symptoms of hyperglycaemia or presence of unequivocal hyperglycaemia including acute metabolic decompensation (diabetic ketoacidosis). The first three criteria were required to be met on two occasions, with a strong preference that at least one of the two testing occasions included an OGTT. HbA1c level ≥48 mmol/mol (≥6.5%) from a laboratory that used The National Glycohemoglobin Standardization Program certified assay standardised to The Diabetes Control and Complications Trial was also accepted as a confirmatory criterion.
DPTRS
A metabolic risk score was calculated for each individual based on a model including loge-BMI, age, loge-fasting C-peptide and post-challenge glucose and C-peptide sums from 2 h OGTT at baseline assessment [3]. This score was used to compare races/ethnicities both as continuous variable and dichotomised variable (<6.5 and ≥6.5). For the purpose of this analysis, we used <6.5 and ≥6.5 to define low and high DPTRS, respectively, based on the previously published differential diabetes risk [20].
Statistical methods
Descriptive analyses were used to summarise characteristics across all participants as well as within single confirmed autoantibody- and multiple autoantibody-positive cohorts. Characteristics were compared between the race/ethnicity composite groups using Kruskal–Wallis tests for continuous variables and χ2 tests for categorical/dichotomised factors, where Fisher exact tests were used as appropriate with small numbers in subset groups. At-risk individuals who enrolled in prevention trials were censored at the time of their entry into the trial. Primary outcomes for these analyses were time to multiple positive autoantibodies in single confirmed autoantibody positive participants and time to type 1 diabetes diagnosis in multiple autoantibody-positive participants. Time to multiple positive autoantibodies was defined as the time from single confirmed autoantibody positive determination to the time when two or more positive autoantibodies were identified. Time to progression to type 1 diabetes diagnosis was defined as the time from participants being identified as having multiple positive autoantibodies to the time when they were diagnosed with type 1 diabetes. Those who had not progressed at their last follow-up visit were censored at that time point. The two analysis cohorts were not mutually exclusive, as single confirmed autoantibody-positive participants who subsequently converted to multiple positive autoantibodies were included in the multiple autoantibody-positive cohort after that time. Kaplan–Meier methods were used to estimate the proportion of participants who had not had an event (e.g. development of type 1 diabetes) by a certain time. Estimated event rates were also calculated using cumulative incidence analyses. Univariate and multivariable Cox regression models were used to evaluate prognostic utility of the various markers and factors in relation to time to progression to multiple positive autoantibodies or type 1 diabetes diagnosis for the single confirmed and multiple positive autoantibody cohorts, respectively. Cumulative incidences of events of interest were graphed and also adjusted for identified factors of interest using the methodology of Therneau et al [21] and Nieto et al [22]. Multivariable models employed a hybrid variable selection approach based on backwards selection and all subsets regression approaches. Age at autoantibody determination was evaluated as a continuous measure; however, an optimal cut-point for age was identified using recursive partitioning analyses (rpart package in R) [23], which uses a tree-based method and iteratively evaluates all possible cut-points of age that best differentiated participants’ prognosis in relation to, for instance, time to progression to type 1 diabetes [24, 25]. Statistical significance was determined at p < 0.05. All analyses were performed using the statistical program R version 3.4.1 for Windows [26].
Results
A total of 4873 TrialNet PTP participants, comprising 11% Hispanic, 80.9% NHW, 2.9% NHB and 5.2% NHO, were followed prospectively. At screening, 2039 participants (42%) were single confirmed autoantibody positive while 2834 (58%) were positive for multiple autoantibodies. Median follow-up for single to multiple autoantibody conversion was 1.9 years (interquartile range 0.7–4.2 years) and for progression to type 1 diabetes was 1.0 years (interquartile range 0.4–2.9 years) in event-free participants. A total of 363/2039 (18%) participants progressed from single to multiple autoantibody positivity. Across all 4873 participants, 591 (12%) progressed to type 1 diabetes during follow-up (65 single confirmed and 526 multiple autoantibody positive) (Fig. 1). The estimated cumulative rate of type 1 diabetes incidence at 5 years was 39% (95% CI 36, 42) in the multiple autoantibody-positive cohort and 26.4% (95% CI 24, 29) across all autoantibody-positive participants.
We observed differences in the distributions of baseline characteristics between the race/ethnicity groups (Table 1). The pairwise comparisons of baseline characteristics for NHB vs NHW, and NHO vs NHW, were not included because there were no significant differences in primary outcomes (i.e. time to multiple positive autoantibodies in single confirmed autoantibody-positive participants and time to type 1 diabetes in participants with multiple positive autoantibodies) between these respective groups.
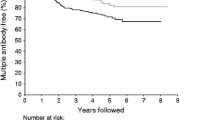
In progression from single confirmed to multiple positive autoantibodies, we found that Hispanic ethnicity, age at screening, sex, DPTRS and HLA-DR3-DQ2/DR4-DQ8 were significant factors affecting time to progression (ESM Table 1). Hispanic ethnicity was significantly associated with a protective effect for conversion to multiple positive autoantibodies (HR 0.66 [95% CI 0.46, 0.96], p = 0.028; ESM Table 1) after adjustment for positive autoantibody type, age, sex, DPTRS, obesity and HLA-DR3-DQ2/DR4-DQ8 genotype (Fig. 2). This lower likelihood of Hispanic participants to progress to multiple positive autoantibody status compared with NHW participants was also observed in those who did not have HLA-DR3-DQ2/DR4-DQ8, even after adjustment for potential confounders (HR 0.63 [95% CI 0.42, 0.94], p = 0.024).
Cumulative incidence of multiple autoantibody positivity in Hispanic vs NHW individuals in the TrialNet PTP cohort with single confirmed autoantibody positivity at screening (p = 0.01). Curves are adjusted for autoantibody type, age, sex, DPTRS and HLA-DR3-DQ2/DR4-DQ8 status. Ab+, autoantibody positive. Solid line, NHW; dashed line, Hispanic
In participants who were positive for multiple autoantibodies at screening, time to type 1 diabetes did not differ significantly by race/ethnicity in the overall cohort (p = 0.91), or between Hispanic vs NHW participants after adjusting for age, sex, DPTRS, number of autoantibodies, HLA-DR3-DQ2/DR4-DQ8 genotype and BMI (ESM Table 2). Cut-point analyses identified 12 years as the appropriate cut-off age in relation to time to progression to type 1 diabetes. In the overall cohort, there was a significant three-way interaction between age, being overweight/obese vs not, and Hispanic vs NHW (p = 0.006; ESM Table 2). Stratified analyses showed that in children <12 years old, ethnicity (Hispanic vs NHW) was a significant effect modifier on the effects of being overweight/obese on cumulative incidence and rate of progression to type 1 diabetes (p = 0.025). In children <12 years old at the time of multiple autoantibody determination, although it was not significantly different, lean Hispanic participants appeared to have a lower rate of progression to type 1 diabetes (HR 0.65 [95% CI 0.36, 1.17], p = 0.15; Fig. 3) than lean NHW participants after adjusting for sex, number of autoantibodies, DPTRS and HLA-DR3-DQ2/DR4-DQ8. However, in this age group, the state of being overweight/obese increased the risk of type 1 diabetes by 36% in NHW children (HR 1.36 [95% CI 1.04, 1.77], p = 0.024; ESM Table 3), while the risk was almost quadrupled in Hispanic participants (HR 3.8 [95% CI 1.6, 9.1], p = 0.0026; ESM Table 3), even after adjusting for sex, number of autoantibodies, DPTRS and HLA-DR3-DQ2/DR4-DQ8 (Fig. 3). In participants ≥12 years old at multiple autoantibody determination, there were no significant differences among Hispanic vs NHW ethnicities. However, we observed a significant interaction between the BMI%ile as a continuous measure and Hispanic ethnicity (p = 0.012). Although BMI%ile was not a significant factor in NHW participants ≥12 years old (HR 0.997 [95% CI 0.991, 1.004], p = 0.38), we noted that it had a significant effect (HR 0.96 [95% CI 0.94, 0.99], p = 0.007) in Hispanic participants ≥12 years old even with the relatively limited number of participants in that group. However, with only 13 events in this restricted cohort, we consider this an interesting and hypothesis-generating observation that warrants further investigation.
Cumulative incidence of type 1 diabetes in TrialNet PTP participants <12 years old with multiple positive autoantibodies, by ethnicity (i.e. Hispanic vs NHW) and BMI groups. Curves are adjusted for sex, number of autoantibodies, DPTRS, and HLA DR3-DQ2/DR4-DQ8 status. Ab+, autoantibody positive. Solid line, lean NHW; dashed line, overweight/obese NHW; dotted line, lean Hispanic; dash–dotted line, overweight/obese Hispanic
To further assess the role and effect modification by Hispanic ethnicity on progression to type 1 diabetes, we also evaluated this outcome in the overall cohort of all autoantibody-positive participants (n = 4873). Findings were similar after adjusting for the number of autoantibodies and other potential confounders (Fig. 4, ESM Tables 4 and 5). No significant differential risks were observed between NHB and NHW ethnic groups.
Cumulative incidence of type 1 diabetes in TrialNet PTP participants <12 years old in the whole at-risk cohort (i.e. both single confirmed and multiple autoantibody-positive cohort), by ethnicity (i.e. Hispanic vs NHW) and BMI groups. Curves are adjusted for sex, number of autoantibodies, DPTRS and HLA-DR3-DQ2/DR4-DQ8 status. Ab+, autoantibody positive. Solid line, lean NHW; dashed line, overweight/obese NHW; dotted line, lean Hispanic; dash–dotted line, overweight/obese Hispanic
Discussion
In our study of the role of race and ethnicity in relation to the progression of islet autoimmunity and type 1 diabetes development in at-risk individuals from the TrialNet PTP cohort, we found that conversion from single to multiple autoantibody positivity was less common in Hispanic than in NHW individuals after adjustment for autoantibody type, age, sex, DPTRS, obesity and HLA-DR3-DQ2/DR4-DQ8. Although time from multiple autoantibody positivity to diagnosis of type 1 diabetes did not differ by race/ethnicity in the overall cohort, we found that Hispanic ethnicity as well as age (specifically <12 or ≥12 years) were significant effect modifiers on the influence of BMI%ile on rates of progression to type 1 diabetes. In children <12 years old at multiple autoantibody determination, an overweight or obese status was a significant factor for progression to type 1 diabetes in both Hispanic and NHW participants; however, this risk was much more pronounced in Hispanic participants than in NHW participants (respective HRs 3.8 and 1.36) even adjusting for sex, number of autoantibodies, DPTRS and HLA-DR3-DQ2/DR4-DQ8. There was also an indication that Hispanic ethnicity could modify the effects of BMI%ile in participants ≥12 years old at autoantibody determination.
Single to multiple autoantibody conversion is a crucial step in islet autoimmunity progression and type 1 diabetes risk [1]. After adjustment for confounding factors, we observed that progression to multiple autoantibody positivity was less common in Hispanic than in NHW participants. The slower progression in Hispanic individuals suggests a lower frequency of predisposing characteristics beyond those that we adjusted for (e.g. additional type 1 diabetes-linked HLA haplotypes or non-HLA genetic factors, or environmental factors). Further research is warranted to understand the basis of this ethnic difference and its impact on type 1 diabetes prediction models as well as type 1 diabetes prevention efforts. Finally, the lower relative ratio of type 1 diabetes development in single confirmed autoantibody-positive vs multiple autoantibody-positive participants in our cohort is similar to previous findings [1] and underscores the role of multiple islet autoantibody positivity in progression to type 1 diabetes [2].
Although there was no difference in time to type 1 diabetes diagnosis in the multiple autoantibody-positive cohort between the races/ethnicities overall, Hispanic participants appeared to be less likely to progress to type 1 diabetes than the NHW participants when considering lean children aged <12 years after adjustment for sex, number of autoantibodies, DPTRS and HLA-DR3-DQ2/DR4-DQ8. Differences in additional type 1 diabetes-associated HLA haplotypes and/or non-HLA genetic factors, as well as epigenetic or environmental factors, may explain the lower relative risk and rate of progression in this group. The significant role of Hispanic ethnicity on modifying the effects of BMI in this age group was best illustrated through the greater detrimental effect of elevated BMI in Hispanic vs NHW children aged <12 years. This phenomenon may be related to the limitation of BMI in estimating per cent body fat and adiposity, which disproportionally affect US Hispanic individuals [27]. Accordingly, BMI has been shown to increase the risk of cardiovascular disease in Hispanic individuals more than in non-Hispanic individuals [28]. Higher adiposity and consequently higher insulin resistance for a given BMI%ile in the Hispanic individuals might have contributed to this disproportional detrimental effect.
Similarly, the lack of clear ethnic differences in progression in individuals aged ≥12 years could be due to lower statistical power because of a smaller sample size or fewer incidences of progression in individuals ≥12 years old, as expected since it is known that older age is associated with slower progression from multiple autoantibody positivity to type 1 diabetes [2, 29, 30]. The ethnic/racial differences in progression to type 1 diabetes in older individuals may be identified in future studies in larger cohorts with a longer observation period. On the contrary, others have reported that high-risk autoantibody profiles (presence of IA2 and/or ZnT8) and genetic factors (HLA class I and non-HLA genes), but not age, are independent risk factors in progression from multiple autoantibody positivity to type 1 diabetes [31,32,33,34].
Our data provide insight into the impact of race/ethnicity in type 1 diabetes progression and may be valuable for the design of predictive models and prevention trials. Future studies aimed at identifying factors (e.g. genetic, epigenetic, environmental, etc.) contributing to slower progression in Hispanic individuals will advance our understanding of the natural history of type 1 diabetes and may have a significant impact on the prevention of type 1 diabetes. Such analyses may also allow for the determination of categorical diabetes subtypes with important therapeutic implications.
We observed that NHW individuals were more likely to have multiple positive autoantibodies when compared with Hispanic individuals, similar to the findings of a previous study conducted in individuals with newly diagnosed type 1 diabetes [35]. The higher incidence of islet autoimmunity observed in NHW individuals could be due to the different distribution of type 1 diabetes-associated HLA haplotypes/genotypes, with increased frequency of susceptibility [6] and decreased frequency of protective types [36] when compared with Hispanic ethnicity.
Counselling family members regarding type 1 diabetes risk is an integral part of modern diabetes care [37]. If confirmed, our findings highlight that the differential effect of race/ethnicity may need to be taken into consideration when counselling at-risk family members. Seroconversion from single confirmed to multiple positive autoantibodies (i.e. presymptomatic phase of type 1 diabetes) was less common in Hispanic participants (vs NHW participants) in this study. Additionally, type 1 diabetes risk appeared to be lower in Hispanic children younger than 12 years without elevated BMI, and in this group, elevated BMI was more detrimental in Hispanic than NHW participants with regard to type 1 diabetes risk. Hence, during counselling of Hispanic families with at-risk children <12 years old, it might be advisable to include the warning that being overweight/obese increases the children’s risk for progression to type 1 diabetes to a much greater extent than the risk in NHW children. This knowledge may encourage Hispanic families to make healthy lifestyle changes. Furthermore, this finding has important public health implications due to the higher prevalence of overweight/obesity status [38] and greater annual rate of increase in type 1 diabetes incidence in Hispanic compared with NHW individuals [4].
Our study had the following limitations: lack of adjustment for other HLA [39, 40] and non-HLA loci [40, 41] that are known to be associated with type 1 diabetes development despite adjustment for the presence of the highest risk genotype; the observational nature of the study allowed identification of associations without implications on causality and the smaller number of participants in minority groups other than Hispanic limited our ability to delineate racial/ethnic differences in those groups. Longer follow-up of the participants will increase the precision of the estimates of progression. Because of the design of the TrialNet PTP study, the duration of positivity of single or multiple autoantibodies prior to screening is not known. However, the racial differences in the proportion of multiple autoantibody-positive individuals at screening (60% in NHW vs 48% in Hispanic individuals, Table 1) reflects the differential rates of early progression. Thus, the difference in progression from single to multiple autoantibody positivity between Hispanic and NHW participants could have been more robust if participants were followed since birth in a different study design. Our study focused on racial/ethnic differences in progression of islet autoantibodies (from single to multiple) and development of type 1 diabetes; analyses of differences in the first appearance of autoantibodies will require cohorts that follow individuals from birth. Additionally, caution should be taken in applying our data to the determination of type 1 diabetes risk in the general population, as our study participants were relatives of individuals with type 1 diabetes and thus were at increased risk for the development of type 1 diabetes.
The major strengths of this study were the relatively large sample size, including a significant number of Hispanic individuals, and the availability of comprehensive type 1 diabetes predictive data (e.g. HLA-DR3-DQ2/DR4-DQ8, DPTRS, etc.). These characteristics enabled us to compare the races/ethnicities for outcome measures while adjusting for confounding factors.
In conclusion, progression of islet autoimmunity, from single to multiple positive autoantibodies, was less common in Hispanic than in NHW individuals, while differences in progression from multiple autoantibodies to type 1 diabetes were limited to children <12 years old and were modified by BMI. Further research is warranted to investigate factors playing a role in the racial/ethnic heterogeneity of type 1 diabetes pathogenesis. Better insight into these factors will allow for adequate counselling of at-risk individuals and for the development of prediction models and design of prevention trials.
Data availability
The datasets generated during and/or analysed during the current study are available in the NIDDK Central Repository.
Abbreviations
- BMI%ile:
-
BMI percentile
- DPTRS:
-
Diabetes Prevention Trial–Type 1 Risk Score
- GAD65:
-
Glutamic acid decarboxylase 65
- IA-2:
-
Islet antigen 2
- ICA:
-
Islet cell autoantibody
- mIAA:
-
Micro-insulin autoantibody
- NHW:
-
Non-Hispanic white
- NHB:
-
Non-Hispanic black
- NHO:
-
Non-Hispanic other
- NIH:
-
National Institutes of Health
- PTP:
-
Pathway to Prevention
- ZnT8:
-
Zinc transporter 8
References
Insel RA, Dunne JL, Atkinson MA et al (2015) Staging presymptomatic type 1 diabetes: a scientific statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care 38:1964–1974
Ziegler AG, Rewers M, Simell O et al (2013) Seroconversion to multiple islet autoantibodies and risk of progression to diabetes in children. JAMA 309:2473–2479
Sosenko JM, Krischer JP, Palmer JP et al (2008) A risk score for type 1 diabetes derived from autoantibody-positive participants in the diabetes prevention trial-type 1. Diabetes Care 31:528–533
Mayer-Davis EJ, Lawrence JM, Dabelea D et al (2017) Incidence trends of type 1 and type 2 diabetes among youths, 2002-2012. N Engl J Med 376:1419–1429
Barinas-Mitchell E, Pietropaolo S, Zhang YJ et al (2004) Islet cell autoimmunity in a triethnic adult population of the Third National Health and Nutrition Examination Survey. Diabetes 53:1293–1302
Black MH, Lawrence JM, Pihoker C et al (2013) HLA-associated phenotypes in youth with autoimmune diabetes. Pediatr Diabetes 14:121–128
Gay EC, Hamman RF, Carosone-Link PJ et al (1989) Colorado IDDM Registry: lower incidence of IDDM in Hispanics. Comparison of disease characteristics and care patterns in biethnic population. Diabetes Care 12:701–708
Kostraba JN, Cruickshanks KJ, Neville TG et al (1992) Clinical characteristics of IDDM in Hispanics and non-Hispanic whites. Little evidence of heterogeneity by ethnicity. Diabetes Care 15:1303–1309
Libman IM, Pietropaolo M, Arslanian SA, LaPorte RE, Becker DJ (2003) Evidence for heterogeneous pathogenesis of insulin-treated diabetes in black and white children. Diabetes Care 26:2876–2882
Morahan G (2012) Insights into type 1 diabetes provided by genetic analyses. Current Opinion in Endocrinology, Diabetes, and Obesity 19:263–270
Tosur M, Redondo MJ (2018) Heterogeneity of type 1 diabetes: the effect of ethnicity. Curr Diabetes Rev 14:266–272
Gandhi K, Tosur M, Schaub R, Haymond MW, Redondo MJ (2017) Racial and ethnic differences among children with new-onset autoimmune type 1 diabetes. Diabet Med 34:1435–1439
Redondo MJ, Libman I, Cheng P et al (2018) Racial/ethnic minority youth with recent-onset type 1 diabetes have poor prognostic factors. Diabetes Care 41:1017–1024
Skyler JS, Greenbaum CJ, Lachin JM et al (2008) Type 1 diabetes TrialNet—an international collaborative clinical trials network. Ann N Y Acad Sci 1150:14–24
Mahon JL, Sosenko JM, Rafkin-Mervis L et al (2009) The TrialNet Natural History Study of the Development of Type 1 Diabetes: objectives, design, and initial results. Pediatr Diabetes 10:97–104
Meah FA, DiMeglio LA, Greenbaum CJ et al (2016) The relationship between BMI and insulin resistance and progression from single to multiple autoantibody positivity and type 1 diabetes among TrialNet Pathway to Prevention participants. Diabetologia 59:1186–1195
NIH (2015) Racial and ethnic categories and definitions for NIH diversity programs and for other reporting purposes. NOT-OD-15-089. Available from https://grants.nih.gov/grants/guide/notice-files/NOT-OD-15-089.html, last accessed 24 May 2018
Fouts A, Pyle L, Yu L et al (2016) Do electrochemiluminescence assays improve prediction of time to type 1 diabetes in autoantibody-positive TrialNet subjects? Diabetes Care 39:1738–1744
Deschamps I, Boitard C, Hors J et al (1992) Life table analysis of the risk of type 1 (insulin-dependent) diabetes mellitus in siblings according to islet cell antibodies and HLA markers. An 8-year prospective study. Diabetologia 35:951–957
Sosenko JM, Skyler JS, Mahon J et al (2011) Validation of the Diabetes Prevention Trial-Type 1 Risk Score in the TrialNet Natural History Study. Diabetes Care 34:1785–1787
Therneau TM, Crowson CS, Atkinson EJ (2015) Adjusted survival curves. Available from https://cran.r-project.org/web/packages/survival/vignettes/adjcurve.pdf, last accessed 24 May 2018
Nieto FJ, Coresh J (1996) Adjusting survival curves for confounders: a review and a new method. Am J Epidemiol 143:1059–1068
Therneau TM, Atkinson EJ (2018) rpart: recursive partitioning and regression trees. R package version 4.1-12. Available from https://CRAN.R-project.org/package=rpart (last accessed 24/5/2018)
Xu P, Krischer JP, Type 1 Diabetes TrialNet Study Group (2016) Prognostic classification factors associated with development of multiple autoantibodies, dysglycemia, and type 1 diabetes—a recursive partitioning analysis. Diabetes Care 39:1036–1044
Redondo MJ, Geyer S, Steck AK et al (2018) TCF7L2 genetic variants contribute to phenotypic heterogeneity of type 1 diabetes. Diabetes Care 41:311–317
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available from https://www.R-project.org/, last accessed 24 May 2018
Wong WW, Strizich G, Heo M et al (2016) Relationship between body fat and BMI in a US Hispanic population-based cohort study: results from HCHS/SOL. Obesity 24:1561–1571
Jayawardene WP, Lohrmann D, Dickinson S, Talagala S, Torabi M (2017) Clinical measures of obesity and cumulative cardiometabolic risk in adolescents. Clin Obes 7:11–21
Steck AK, Dong F, Waugh K et al (2016) Predictors of slow progression to diabetes in children with multiple islet autoantibodies. J Autoimmun 72:113–117
Krischer JP, Liu X, Lernmark A et al (2017) The influence of type 1 diabetes genetic susceptibility regions, age, sex, and family history on the progression from multiple autoantibodies to type 1 diabetes: a TEDDY study report. Diabetes 66:3122–3129
Gorus FK, Balti EV, Messaaoui A et al (2017) Twenty-year progression rate to clinical onset according to autoantibody profile, age, and HLA-DQ genotype in a registry-based group of children and adults with a first-degree relative with type 1 diabetes. Diabetes Care 40:1065–1072
Balke EM, Balti EV, Van der Auwera B et al (2018) Accelerated progression to type 1 diabetes in the presence of HLA-A*24 and -B*18 is restricted to multiple islet autoantibody-positive individuals with distinct HLA-DQ and autoantibody risk profiles. Diabetes Care 41:1076–1083
Achenbach P, Hummel M, Thumer L, Boerschmann H, Hofelmann D, Ziegler AG (2013) Characteristics of rapid vs slow progression to type 1 diabetes in multiple islet autoantibody-positive children. Diabetologia 56:1615–1622
Lipponen K, Gombos Z, Kiviniemi M et al (2010) Effect of HLA class I and class II alleles on progression from autoantibody positivity to overt type 1 diabetes in children with risk-associated class II genotypes. Diabetes 59:3253–3256
Aviles-Santa L, Maclaren N, Raskin P (2004) The relationship between immune-mediated type 1 diabetes mellitus and ethnicity. J Diabetes Complicat 18:1–9
Erlich HA, Zeidler A, Chang J et al (1993) HLA class II alleles and susceptibility and resistance to insulin dependent diabetes mellitus in Mexican-American families. Nat Genet 3:358–364
Chiang JL, Kirkman MS, Laffel LM, Peters AL, Type 1 Diabetes Sourcebook Authors (2014) Type 1 diabetes through the life span: a position statement of the American Diabetes Association. Diabetes Care 37:2034–2054
Ogden CL, Carroll MD, Fryar CD, Flegal KM (2015) Prevalence of obesity among adults and youth: United States, 2011-2014. NCHS Data Brief No. 219, November 2015. Available from https://www.cdc.gov/nchs/products/databriefs/db219.htm, last accessed on 24 May 2018)
Ilonen J, Kiviniemi M, Lempainen J et al (2016) Genetic susceptibility to type 1 diabetes in childhood—estimation of HLA class II associated disease risk and class II effect in various phases of islet autoimmunity. Pediatr Diabetes 17(Suppl 22):8–16
Redondo MJ, Steck AK, Pugliese A (2017) Genetics of type 1 diabetes. Pediatr Diabetes 19:346–353
Steck AK, Xu P, Geyer S et al (2017) Can non-HLA single nucleotide polymorphisms help stratify risk in TrialNet relatives at risk for type 1 diabetes? J Clin Endocrinol Metab 102:2873–2880
Funding
The sponsor of the trial was the Type 1 Diabetes TrialNet Study Group. The Type 1 Diabetes TrialNet Study Group is a clinical trials network funded by the NIH through the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases and The Eunice Kennedy Shriver National Institute of Child Health and Human Development, through cooperative agreements U01 DK061010, U01 DK061034, U01 DK061042, U01 DK061058, U01 DK085465, U01 DK085453, U01 DK085461, U01 DK085466, U01 DK085499, U01 DK085504, U01 DK085509, U01 DK103180, U01 DK103153, U01 DK085476, U01 DK103266, U01 DK103282, U01 DK106984, U01 DK106994, U01 DK107013, U01 DK107014, UC4 DK106993, and the JDRF. The contents of this article are solely the responsibility of the authors and do not necessarily represent the official views of the NIH or the JDRF.
Author information
Authors and Affiliations
Consortia
Contributions
All authors are members of the Type 1 Diabetes TrialNet Study Group and, as such, contributed to the data used in this article. MT and MJR designed the study. All authors contributed to result interpretation. SMG performed statistical analyses. MT wrote the initial draft and edited the manuscript. SMG, HR, IL, DAB and MJR reviewed and edited the manuscript. MJR is the guarantor of this work, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
The authors declare that there is no duality of interest associated with this manuscript.
Prior presentation
Part of this study was presented as an oral presentation at the 77th Scientific Sessions of the American Diabetes Association, San Diego, CA, USA, 9–13 June 2017.
Additional information
A complete list of the Type 1 Diabetes TrialNet Study Group can be found in the electronic supplementary material.
Electronic supplementary material
ESM
(PDF 458 kb)
Rights and permissions
About this article
Cite this article
Tosur, M., Geyer, S.M., Rodriguez, H. et al. Ethnic differences in progression of islet autoimmunity and type 1 diabetes in relatives at risk. Diabetologia 61, 2043–2053 (2018). https://doi.org/10.1007/s00125-018-4660-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-018-4660-9