Abstract
Aims/hypothesis
Atherosclerosis and cardiovascular diseases are often present at the time of diagnosis of type 2 diabetes mellitus. Whether subclinical atherosclerosis can be detected in the pre-diabetic (borderline fasting hyperglycemia) state is not clear. This study investigated the association of impaired fasting glucose (IFG) and coronary artery calcification (CAC), a marker of subclinical atherosclerosis, among participants without a history of coronary heart disease or manifest diabetes mellitus.
Methods
Study participants (aged 45–75 years) of the population-based Heinz Nixdorf Recall Study were categorised into those with normal fasting glucose (glucose <6.1 mmol/l) and those with IFG (glucose ≥6.1 to <7.0 mmol/l), excluding participants with a history of CHD or diabetes mellitus. CAC was assessed by electron-beam computed tomography, and risk factors were assessed by extended interviews, anthropometric measurements and laboratory tests. Various CAC cut-off points were used in multiple logistic and ordinal logistic regression models to estimate ORs and 95% CIs.
Results
Of the 2,184 participants, more men had IFG than did women (37% vs 22%). Participants with IFG showed a higher prevalence of CAC > 0 (men OR 1.90, 95% CI 1.33–2.70; women 1.63, 1.23–2.15). Risk factor adjustment weakened this association in both sexes (men 1.63, 1.12–1.36; women 1.26, 0.93–1.70). When the age- and sex-specific 75th percentile was used as the cut-off point for CAC, the association further decreased in men (1.10, 0.81–1.50), but became stronger in women (1.41, 1.02–1.94).
Conclusions/interpretation
These data support the hypothesis that CAC is already present in the pre-diabetic state and that IFG has a modest and independent impact on the atherosclerotic process. Biological sex appears to modify the association between IFG and CAC.
Similar content being viewed by others
Introduction
Diabetes mellitus is a major risk factor for cardiovascular diseases (CVD) [1]. With the repeatedly predicted worldwide increase of type 2 diabetes mellitus [2] a concomitant increase of CVD might be expected. Diabetes mellitus and CVD are two closely related chronic disease entities. The underlying causes of both diseases, insulin resistance and atherosclerosis, are characterised by a long asymptomatic developmental period, starting early in life.
Since the incidence of CVD apparently does not depend on the duration of diabetes mellitus [3], one may speculate that an impaired glucose homeostasis occurs many years before a clinical diagnosis of diabetes mellitus. The World Health Organization has recently summarised the developments in and the reasoning behind changes in classification to include impaired glucose tolerance (IGT) and impaired fasting glucose (IFG) as states occurring before the diagnosis of diabetes [4].
In a meta-analysis a pooled relative risk for incident diabetes mellitus of 4.7 was found in participants with IFG [5]. However, for non-fatal and fatal CVD among participants with IFG the evidence is less consistent, with relative risks ranging between 1.2 and 1.3 compared with participants with normal fasting glucose (NFG) [6].
Several concepts have been developed to explain the observation of higher CVD risk in participants with elevated fasting blood glucose. Some suggest that hyperglycaemia induces a large number of alterations in vascular tissue that potentially promote atherosclerosis [7]; others support the idea that glucose itself is not directly involved in the pathogenesis of cardiovascular disease but correlates with other risk factors that potentially contribute to macrovascular complication [8].
A number of clinical investigations have shown that coronary artery calcification (CAC) is a valid indicator for subclinical atherosclerosis [9]. Previous studies using non-invasive electron-beam computed tomography (EBCT) as a method for the detection and quantification of CAC have found a higher plaque burden in participants with diabetes mellitus [10–14]. However, uncertainty remains regarding the relationship between IFG and CAC in the general population without diabetes mellitus.
The aim of this study was to investigate the association of IFG with the extent of coronary artery calcification in middle aged men and women without overt coronary artery disease (CAD) or diabetes mellitus. For this purpose, we analysed cross-sectional data of a large ongoing population-based cohort of EBCT-screened participants with NFG or IFG.
Methods
Study population
The study base of the ongoing Heinz Nixdorf Recall (Risk Factors, Evaluation of Coronary Calcium and Lifestyle) Study are men and women of the general German population living in three adjacent cities of the densely populated Ruhr metropolitan area in Germany. The rationale and design of the study have been described in detail [15]. Briefly, 4,814 participants aged 45–75 years, randomly selected from mandatory lists of residents of Essen, Bochum and Mülheim, were recruited between 2000 and 2003. The baseline response was 56% [16]. The study was approved by the institutional local ethics committees and comprises extended quality management procedures, including a certification according to DIN ISO 9001:2000. Informed consent was obtained from all participants.
Blood analysis
Standardised enzymatic methods were used to determine total cholesterol, HDL-cholesterol, LDL-cholesterol and triacylglycerol. Serum for glucose determination was immediately centrifuged at the research centre and analysed enzymatically by the hexokinase method. All analyses were done within 12 h at the central laboratory of the University Hospital of Essen, Germany.
Glucose status
NFG was defined as fasting plasma glucose <6.1 mmol/l, IFG as fasting plasma glucose ≥6.1 but <7.0 mmol/l [17]. Fasting was defined as no energy intake for at least 8 h.
CAC
CAC was assessed by non-enhanced electron-beam CT scans (C-150 scanner, GE Imatron, San Francisco, CA, USA), as previously described in detail [15]. CAC was defined as a hyperattenuating focus of at least four contiguous pixels with a CT density ≥130 Hounsfield units. The area of each focus was measured and the CAC score was determined using the methods of Agatston et al. [18].
Risk factor assessment
Smoking, medical history and socioeconomic status were assessed by trained personnel with standardised computer-assisted face-to-face interviews. Assessment of current use of medication was performed by asking participants to bring along all medication (including packages) taken in the last 7 days. Blood pressure was measured using an automated oscillometric blood pressure device (HEM-705CP-E, Omron Healthcare Europe, Hoofdorp, the Netherlands) with participants in sitting position, using the mean of the second and third value of three measurements [19]. Body mass index (BMI) was calculated from standardised measurements of height and weight with participants in light underwear without shoes.
Diagnostic conventions
A history of manifest coronary artery disease was assessed by self reports of previous events (myocardial infarction and/or revascularisation of coronary arteries including balloon dilatation and coronary bypass surgery). Participants were classified as having diabetes if they reported a history of diagnosis of this disease or intake of glucose-lowering drugs. Antihypertensive therapy included the following classes of drugs or any combination of them: diuretics, β-blockers, angiotensin-converting enzyme inhibitors, angiotensin-receptor antagonists, calcium channel blockers, centrally active antihypertensive drugs and hydralazine. Lipid-lowering medication included HMG CoA reductase inhibitors (statins), fibrates, bile acid sequestrants and nicotinic acid derivatives.
Statistical analyses
Analyses were performed on a subgroup of the study population with NFG or IFG, excluding participants with a self-reported history of CAD (n = 327), known and unknown diabetes mellitus (n = 581), with a fasting state less than 8 h (n = 1,629) or lacking data for CAC (n = 93). The final study group consisted of 2,184 (1,200 women, 984 men) study participants. We did not find evidence for differences in the main population characteristics between those participants excluded because of non-fasting status or missing CAC data and those available for analysis. Given extreme sex differences in the distribution of the CAC score, all analyses were conducted separately for men and women.
We categorised CAC scores a priori into four risk groups: minimal (<10), mild (≥10 to <100), moderate (≥100 to <400) and extensive (≥400) atherosclerotic plaque burden [20]. Overweight (BMI ≥ 25 to <30 kg/m2) and obesity (BMI ≥ 30 kg/m2) were defined according to the WHO conventions [21]. To control for socioeconomic status, education was classified as total years of formal education, classified into ≤10 and >10 years of education.
The crude association between IFG and CAC was visualised by plotting the loge-transformed mean CAC score +1 against the fasting plasma glucose (see Electronic supplementary material [ESM] Fig. 1). Because of the high sample size, we categorised plasma glucose into sex-specific deciles of glucose. We used multiple logistic regression to analyse the association between IFG and CAC. The CAC score as outcome was divided into two groups on the basis of (1) the presence or absence of any CAC (CAC > 0) and (2) the age- and sex-specific 75th percentiles, determined by the CAC distribution of all study participants of the Heinz Nixdorf Recall Study not taking any cardiovascular medication [22]. Finally, ordinal logistic regression models were fitted to evaluate the association between an increase in level of CAC score, in order to study associations regardless of where the cut-off point was set for the ordinal outcome [23]. We used the proportional odds model (POM), which can be interpreted as an intermediate approach between a binary cut-off point and a continuous distribution model. The ordinal outcome has four levels according to the CAC risk groups (see above). The special feature of a POM is that the OR of a predictor can be interpreted as a summary of the ORs obtained from separate binary logistic regression models using all possible cut-off points of the ordinary outcome [24]. Whereas a binary logistic regression models a single logit, the POM models several logits. Therefore, since the outcome CAC has four levels (cut-off points), three logits were will be modelled. For a POM to be valid, the assumption that the ORs can be interpreted as a constant across all possible cut-off points of the outcome must be tested (in SAS 9.1 this is called the score test for the proportional odds assumption). Here the proportional odds assumption held true (χ 2 score tests >0.05), so the association between glucose status and CAC could be expressed as a single summary estimate (OR) across all the CAC risk categories. For all regression models, we initially fit a crude model (model 1), expanding the model by adjusting for age (model 2), age and BMI (model 3) and a final model with additional adjustment for smoking, HDL-cholesterol, total cholesterol and blood pressure (model 4). In addition we performed age-stratified analyses using model 4 for each age-group (45–54, 55–64 and 65–75 years). Statistical analyses were performed using SAS 9.1 (SAS Institute, Cary, NC, USA).
Results
Of 2,184 participants 29% (n = 633) had IFG. Among men a higher prevalence than in women was observed (37% vs 22%). Characteristics of the study population according to the glucose status are shown in Table 1. Participants with IFG were particularly characterised by a higher mean BMI and waist circumference than those with NFG. Accordingly, more participants with IFG were classified as being obese (BMI ≥ 30 kg/m2). In addition the participants with IFG were more likely to be older and hypertensive. Dyslipidaemia, smoking status and use of medications differed between sexes with regard to glucose status.
For both sexes a positive association between fasting plasma glucose levels and CAC can be shown (ESM Fig. 1). The prevalence of NFG and IFG by CAC risk group and age group is presented in Table 2. A higher proportion of men and women with NFG had no or only small amounts of CAC (<10) compared with those with IFG. On the other hand, participants with IFG were more likely to exhibit high amounts of CAC (≥400). However, in the age group 55–64 years as well as in the CAC risk-groups with values of CAC ≥10 to <100 and ≥10 to <400, this tendency is less pronounced, especially in men.
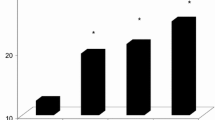
Results of the logistic regression analyses showed an association between the presence of CAC (>0) in participants with IFG compared with those with NFG (Fig. 1). In men, the crude OR was 1.90 (95% CI 1.33–2.70), in women 1.63 (1.23–2.15). Controlling for CVD risk factors weakened this association among men (1.63, 1.12–2.36) and women (1.26, 0.93–1.70). Stratification by age showed an association between IFG and CAC only in men in the youngest age group (OR 2.76, 1.51–5.04). A similar tendency, with the highest OR in the youngest age group, could be observed in women, but at a lower level (OR 1.36, 0.78–2.39).
Crude (model 1) and adjusted ORs for presence of any CAC (CAC >0) for men (a) and women (b). Vertical lines indicate 95% CI. Adjusted for age (model 2), model 2+BMI (model 3), and model 3+smoking, blood pressure, total cholesterol and HDL-cholesterol (model 4). Analyses were also done separately for age groups: the ORs of the fully adjusted model (model 4) are shown
Using a cut-off point for CAC above the 75th age- and sex-specific percentile revealed a stronger association between CAC and IFG in women than in men (Fig. 2). In men the association attenuated, whereas in women the OR remained elevated even in the fully adjusted model (OR 1.41, 1.02–1.94).
Crude (model 1) and adjusted ORs for a CAC score above the age- and sex-specific 75th percentile of the total sample of the Heinz Nixdorf Recall Study for men (a) and women (b). Vertical lines indicate 95% CI. Adjusted as in Fig. 1 without adjusting for age
Similar observations were found using a cut-off point independent model (Fig. 3). The POM generated elevated crude proportional odds in men (OR 1.34, 1.06–1.69) and women (OR 1.59, 1.21–2.09). In other words, men with IFG were approximately 30% (women 60%) more likely to be in the higher coronary calcification groups than those participants with NFG. Since the proportional odds assumption is met, this interpretation holds across the entire range to the CAC risk score groups. However, in the full model the observed associations became weaker among both men and women, with barely any association among men (OR 1.13, 0.89–1.44; women: 1.34, 1.01–1.78).
Crude (model 1) and adjusted ORs of the ordinal logistic regression analysis (POM), using as ordinal categories the CAC risk score groups: minimal (<10), mild (≥10–<100), moderate (≥100–<400) and extensive atherosclerotic calcification (≥400) for men (a) and women (b). Vertical lines indicate 95% CI. Adjusted as in Fig. 1
Adjustments for further potential confounders (i.e. socioeconomic status) did not substantially change these associations. Since we did not perform an OGTT, we were not able to exclude all participants with undiagnosed diabetes mellitus. We performed a sensitivity analysis to estimate the possible influence of these undetected cases. We calculated the number of undiagnosed diabetes mellitus cases, relying on several assumptions. (1) Since many studies could show a similar diagnostic performance of HbA1c to that of fasting glucose or an OGTT, we used HbA1c measurements of our cohort to define undiagnosed diabetes (although this is not recommended as a test for diabetes). However, since no actual reference value exists and many individuals with IGT may have normal or near normal glycated haemoglobin levels, we additionally used an elevated CAC score as a screening tool for undiagnosed diabetes. In a first step we excluded participants with HbA1c values above the sex-specific means of our subgroup with undiagnosed diabetes (detected by IFG) and recalculated our results. In a second step we excluded participants with additionally elevated CAC scores (above the sex-specific mean). The results of the two approaches did not differ and reduced only marginally our point estimates. (2) Assuming that 30% of people with diabetes are not diagnosed as such if only IFG is used, we conducted a sensitivity analysis excluding a random 30% of the study sample, repeating our analysis three times. With this approach we obtained slightly elevated but still comparable point estimates.
Discussion
Our study shows a positive association between impaired fasting glucose as a sign of a pre-diabetic state (borderline fasting hyperglycemia) and CAC as a marker of subclinical atherosclerosis in a population-based cohort of middle-aged men and women without overt CVD and diabetes mellitus. Adjustment for potential confounders did not substantially change these associations.
Our study has several strengths. (1) We screened in a large population-based sample for the prevalence and extent of CAC. The CAC score has been shown to correlate with traditional cardiovascular risk factors [25–27] and is strongly related to CHD events [28, 29]. (2) We excluded participants with a history of CVD as well as known and unknown diabetes. (3) The large sample size made it possible to study the association between IFG and CAC separately for men and women, which is especially important given the sex differences in relation to the CAC burden. (4) We conducted a consistent quality assurance with a comprehensive assessment of standard cardiovascular risk factors, lifestyle behaviours and medication use under one protocol from one single assessment centre. (5) Blood samples were prepared immediately and analysed in the same laboratory with the same machine by the same trained staff using the same methodology.
The strengths are balanced by a number of limitations. Although we tried to minimise errors due to pre-analytical and analytical conditions exposure misclassification is unavoidable, bearing in mind the following factors: (1) measurement errors of glucose of at least ±2%; (2) biological variation of about ±5% [30] (these first two factors result in an optimal reference change value [RCV] of 14% [6]); (3) probably incorrect statements of the study participants of their fasting status; (4) the narrow interval of IFG from 6.1 to 6.9 mmol/l; and (5) that we did not measure IGT by an OGTT. This last factor means that we could not distinguish between participants with IFG or IGT, and probably misclassified some participants with unknown diabetes as having NFG or IFG. However, we performed several sensitivity analyses by estimating the number of participants with unknown diabetes (see results). We found only marginally changed point estimates and concluded that the influence of these cases on our results seems negligible.
To our knowledge there are currently only two published studies reporting on glucose metabolism and CAC. The association between high fasting blood glucose in the non-diabetic range and CAC was evaluated in a study of 458 selected non-diabetic Brazilian men without CVD who presented for an obligatory cardiac risk assessment paid for by their employers [31]. Although different glucose and CAC risk categories were used, similar results were reported, demonstrating a relationship between fasting blood glucose and CAC in the non-diabetic range.
In a subsample of the Framingham Offspring Study, the risk for CAC in 325 men and women according to insulin resistance, normal glucose tolerance, impaired glucose tolerance or type 2 diabetes was assessed [32]. The ORs for subclinical atherosclerosis (defined as the upper CAC quartile, ranging from 170 to 3,266) tended to be higher in participants with IFG and/or IGT (fully adjusted OR 1.49; 95% CI 0.65–3.38). Because of their small sample size they could not conduct sex-specific analysis and their results had constrained analytic power. However, our results are in line with their observations, confirming their results in a practical sense.
Most studies that have investigated the impact of an abnormal glucose homeostasis on the risk of subclinical atherosclerosis or CVD have aimed to identify and explain differences in IFG and IGT. Furthermore, the majority of studies investigated atherosclerosis in the peripheral arteries, in particular the carotid intima–media. The results indicate a positive association between IFG/IGT and carotid intima–media thickness [33–35], corresponding to our results. One study investigated in 232 Japanese men the association between IFG and brachial ankle pulse wave velocity, a novel indicator of early stage atherosclerosis [36]. As in our study, IFG was closely related to pulse wave velocity regardless of age, blood pressure or BMI, providing further evidence of an independent association between IFG and atherosclerosis.
It is accepted meanwhile that participants with diabetes mellitus are at high risk of developing CVD and that CVD accounts for a large proportion of the excess mortality related to diabetes mellitus [37]. Less evident is the relationship between elevated blood glucose and the development of atherosclerosis, the underlying cause of CVD. Several studies have shown that CAC is higher in participants with diabetes mellitus than without diabetes mellitus [11–14, 38], suggesting that atherosclerosis already starts in the pre-diabetic state. This is in line with a meta-analysis of 38 prospective studies in which CVD incidence or mortality was predicted by elevated blood glucose levels among participants without diabetes, independently of traditional risk factors [39]. However, other investigators could not find an independent impact of elevated blood glucose on CVD [40–42]. So far, evidence for an involvement of high blood glucose in the pathogenesis of CVD is mixed and possible pathways are still unclear.
We found that the association between IFG and CAC was explained only in part by differences in medication use and CVD risk factors, but was considerably affected by age, sex and the cut-off point of the CAC score. For men a marked elevated OR could be found only when comparing no vs any CAC. When using the age- and sex-specific 75th percentile, the risk-factor-adjusted association between IFG and CAC vanished. Furthermore, stratification by age revealed that an association could be detected only in younger men (aged 45–54 years). This might be explained by the fact that men with no CAC were scarce and most likely to be found in the youngest age-group (age 45–54: 30%, 55–64: 15%, 65–75: 9%). In any case, these data show that the probability of no CAC is higher in those men with normal fasting glucose levels.
Compared with men, a higher proportion of women in every age-group had no CAC (65%, 46%, 32%), and the association between IFG and CAC > 0 was less pronounced in women. However, stronger associations could be detected using cut-off points representing higher accumulations of CAC. One explanation for these differences is that women exhibited a considerably lower burden of CAC than men. In addition, other investigators found that more men than women had IFG, whereas women more often had IGT [43–45], reflecting different physiological processes in men and women: IFG reflects the basal fasting state with an impaired insulin secretion, whereas IGT represents postprandial abnormalities with insulin resistance. In addition there might be a different impact of glucose on atherosclerotic processes in men and women. Results of the Framingham study showed qualitatively different relationships between glucose and mortality in men and women; there was a rapidly rising risk in men through the normal range of blood glucose and a flattening curve thereafter, whereas in women no relationship between the normal glucose range and mortality could be observed but there was a steep risk increase in the impaired glucose range [42]. These results emphasise the need for sex-specific analysis, at least when determining CAC as outcome.
In conclusion, in this subsample of the population-based Heinz Nixdorf Recall Study, men and women with IFG exhibited higher amounts of CAC than those with NFG. These data provide further evidence that CAC is already present in the pre-diabetic state and that a high normal plasma blood glucose has a modest and independent impact on the atherosclerotic process. The strength of association seems to differ between men and women, and this will be explored further in this ongoing study.
Abbreviations
- CAC:
-
coronary artery calcification
- CVD:
-
cardiovascular disease
- EBCT:
-
electron-beam computed tomography
- IFG:
-
impaired fasting glucose
- IGT:
-
impaired glucose tolerance
- NFG:
-
normal fasting glucose
- POM:
-
proportional odds model
References
Laakso M, Lehto S (1997) Epidemiology of macrovascular disease in diabetes. Diabetes Rev 5:294–315
Wild S, Roglic G, Green A, Sicree R, King H (2004) Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 27:1047–1053
Haffner SM, Stern MP, Hazuda HP, Mitchell BD, Patterson JK (1990) Cardiovascular risk factors in confirmed prediabetic individuals. Does the clock for coronary heart disease start ticking before the onset of clinical diabetes. JAMA 263:2893–2898
World Health Organization, International Diabetes Federation (2006) Definition and diagnosis of diabetes mellitus and intermediate hyperglycemia. Report of a WHO/IDF consultation. Available from http://www.who.int/diabetes/publications/Definition%20and%20diagnosis%20of%20diabetes_new.pdf, accessed 19 September 2008
The Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (1997) Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 20:1183–1197
Santaguida PL, Balion C, Hunt D et al (2005) Diagnosis, prognosis, and treatment of impaired glucose tolerance and impaired fasting glucose. Summary, Evidence Report/Technology Assessment No. 128. Available from http://www.ahrq.gov/downloads/pub/evidence/pdf/impglucose/impglucose.pdf, accessed 19 September 2008
Aronson D, Rayfield E (2002) How hyperglycemia promotes atherosclerosis: molecular mechanisms. Cardiovasc Diabetol 1:1. doi:10.1186/1475-2840-1-1
Reaven GM (2005) Insulin resistance, the insulin resistance syndrome, and cardiovascular disease. Panminerva Med 47:201–210
Greenland P, Bonow RO, Brundage BH et al (2007) ACCF/AHA 2007 Clinical expert consensus document on coronary artery calcium scoring by computed tomography in global cardiovascular risk assessment and in evaluation of patients with chest pain: A Report of the American College of Cardiology Foundation Clinical Expert Consensus Task Force Developed in Collaboration With the Society of Atherosclerosis Imaging and Prevention and the Society of Cardiovascular Computed Tomography. J Am Coll Cardiol 49:378–402
Olson JC, Edmundowicz D, Becker DJ, Kuller LH, Orchard TJ (2000) Coronary calcium in adults with type 1 diabetes: a stronger correlate of clinical coronary artery disease in men than in women. Diabetes 49:1571–1578
Mielke CH, Shields JP, Broemeling LD (2001) Coronary artery calcium, coronary artery disease, and diabetes. Diabetes Res Clin Pract 53:55–61
Schurgin S, Rich S, Mazzone T (2001) Increased prevalence of significant coronary artery calcification in patients with diabetes. Diabetes Care 24:335–338
Wolfe ML, Iqbal N, Gefter W et al (2002) Coronary artery calcification at electron beam computed tomography is increased in asymptomatic type 2 diabetics independent of traditional risk factors. J Cardiovasc Risk 9:369–376
Elkeles RS, Feher MD, Flather MD et al (2004) The association of coronary calcium score and conventional cardiovascular risk factors in type 2 diabetic subjects asymptomatic for coronary heart disease (The PREDICT Study). Diab Med 21:1129–1134
Schmermund A, Möhlenkamp S, Stang A et al (2002) Assessment of clinically silent atherosclerotic disease and established and novel risk factors for predicting myocardial infarction and cardiac death in healthy middle-aged subjects: rationale and design of the Heinz Nixdorf Recall Study. Risk factors, evaluation of coronary calcium and lifestyle. Am Heart J 144:212–218
Stang A, Moebus S, Dragano N et al (2005) Baseline recruitment and analyses of nonresponse of the Heinz Nixdorf Recall Study: identifiability of phone numbers as the major determinant of response. Eur J Epidemiol 20:489–496
World Health Organization (1999) Definition and diagnosis and classification of diabetes mellitus and its complications: Report of a WHO consultation. Part 1.32–33. Available from http://www.staff.ncl.ac.uk/philip.home/who_dmg.pdf, accessed 19 September 2008
Agatston AS, Janowitz WR, Hildner FJ et al (1990) Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 15:827–832
Stang A, Moebus S, Möhlenkamp S et al (2006) Algorithms for converting random-zero to automated oscillometric blood pressure values, and vice versa. Am J Epidemiol 164:85–94
Rumberger JA, Brundage BH, Rader DJ, Kondos G (1999) Electron beam computed tomographic coronary calcium scanning: a review and guidelines for use in asymptomatic persons. Mayo Clin Proc 74:243–252
World Health Organization (2000) Obesity: preventing and managing the global epidemic. Report on a WHO Consultation. Technical Report Series, No 894. Geneva, WHO
Schmermund A, Möhlenkamp S, Berenbein S et al (2006) Population-based assessment of subclinical coronary atherosclerosis using electron-beam computed tomography. Atherosclerosis 185:177–182
Bender R, Grouven U (1997) Ordinal logistic regression in medical research. J R Coll Physicians Lond 31:546–551
Scott SC, Goldberg MS, Mayo NE (1997) Statistical assessment of ordinal outcomes in comparative studies. J Clin Epidemiol 50:45–55
Möhlenkamp S, Lehmann N, Schmermund A et al (2003) Prognostic value of extensive coronary calcium quantities in symptomatic males—a 5-year follow-up study. Eur Heart J 24:845–854
Vliegenthart R, Oudkerk M, Song B et al (2002) Coronary calcification detected by electron-beam computed tomography and myocardial infarction. The Rotterdam Coronary Calcification Study. Eur Heart J 23:1596–1603
Hoff JA, Daviglus ML, Chomka EV et al (2003) Conventional coronary artery disease risk factors and coronary artery calcium detected by electron beam tomography in 30,908 healthy individuals. Ann Epidemiol 13:163–169
Budoff MJ, Achenbach S, Blumenthal RS et al (2006) Assessment of coronary artery disease by cardiac computed tomography: a scientific statement from the American Heart Association Committee on Cardiovascular Imaging and Intervention, Council on Cardiovascular Radiology and Intervention, and Committee on Cardiac Imaging, Council on Clinical Cardiology. Circulation 114:1761–1791
Erbel R, Möhlenkamp S, Kerkhoff G, Budde T, Schmermund A (2007) Non-invasive screening for coronary artery disease: calcium scoring. Heart 93:1620–1629
Hyltoft PP, Brandslund I, Jorgensen L et al (2001) Evaluation of systematic and random factors in measurements of fasting plasma glucose as the basis for analytical quality specifications in the diagnosis of diabetes. 3. Impact of the new WHO and ADA recommendations on diagnosis of diabetes mellitus. Scand J Clin Lab Invest 61:191–204
Nasir K, Santos R, Tufail K et al (2007) High-normal fasting blood glucose in non-diabetic range is associated with increased coronary artery calcium burden in asymptomatic men. Atherosclerosis 195:155–160
Meigs JB, Larson MG, D’Agostino RB et al (2002) Coronary artery calcification in type 2 diabetes and insulin resistance: the Framingham Offspring Study. Diabetes Care 25:1313–1319
Wagenknecht LE, D’Agostino RB, Haffner SM, Savage PJ, Rewers M (1998) Impaired glucose tolerance, type 2 diabetes, and carotid wall thickness: the Insulin Resistance Atherosclerosis Study. Diabetes Care 21:1812–1818
Haffner SM (1998) The importance of hyperglycemia in the nonfasting state to the development of cardiovascular disease. Endocr Rev 19:583–592
Faeh D, William J, Yerly P, Paccaud F, Bovet P (2007) Diabetes and pre-diabetes are associated with cardiovascular risk factors and carotid/femoral intima–media thickness independently of markers of insulin resistance and adiposity. Cardiovasc Diabetol 6:32. doi:10.1186/1475-2840-6-32
Ohnishi H, Saitoh S, Takagi S et al (2003) Pulse wave velocity as an indicator of atherosclerosis in impaired fasting glucose: The Tanno and Sobetsu Study. Diabetes Care 26:437–440
Lee WL, Cheung AM, Cape D, Zinman B (2000) Impact of diabetes on coronary artery disease in women and men: a meta-analysis of prospective studies. Diabetes Care 23:962–968
Hoff JA, Quinn L, Sevrukov A et al (2003) The prevalence of coronary artery calcium among diabetic individuals without known coronary artery disease. J Am Coll Cardiol 41:1008–1012
Levitan EB, Song Y, Ford ES, Liu S (2004) Is nondiabetic hyperglycemia a risk factor for cardiovascular disease? A meta-analysis of prospective studies. Arch Intern Med 164:2147–2155
Sorkin JD, Muller DC, Fleg JL, Andres R (2005) The relation of fasting and 2-h postchallenge plasma glucose concentrations to mortality: data from the Baltimore Longitudinal Study of Aging with a critical review of the literature. Diabetes Care 28:2626–2632
Grundy SM (2006) Diabetes and coronary risk equivalency: what does it mean. Diabetes Care 29:457–460
Levitzky YS, Pencina MJ, D’Agostino RB et al (2008) Impact of impaired fasting glucose on cardiovascular disease: the Framingham Heart Study. J Am Coll Cardiol 51:264–270
Borch-Johnsen K (2002) IGT and IFG. Time for revision. Diabet Med 19:707
Williams JW, Zimmet PZ, Shaw JE et al (2003) Gender differences in the prevalence of impaired fasting glycaemia and impaired glucose tolerance in Mauritius. Does sex matter. Diabet Med 20:915–920
Bock G, Dalla Man C, Campioni M et al (2006) Pathogenesis of pre-diabetes: mechanisms of fasting and postprandial hyperglycemia in people with impaired fasting glucose and/or impaired glucose tolerance. Diabetes 55:3536–3549
Chobanian AV, Bakris GL, Black HR et al (2003) The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA 289:2560–2572
Acknowledgements
We thank the Heinz Nixdorf Foundation (Chairman: G. Schmidt) for the generous support of the Heinz Nixdorf Study. The study is also supported by the German Ministry of Education and Science. An additional research grant was received from Imatron (South San Francisco, CA, USA), which produced the EBCT scanners, and GE-Imatron (South San Francisco, CA, USA) after the acquisition of Imatron. Assessment of psychosocial factors and neighbourhood-level information is funded by the German Research Council (DFG; Projects SI 236/8-1 and SI 236/9-1). We acknowledge the support of the Sarstedt (Nümbrecht, Germany) concerning laboratory equipment. The authors are indebted to the dedicated personnel of both the study centre and the EBT-scanner facilities. Advisory board: T. Meinertz, Hamburg, Germany (Chair), C. Bode, Freiburg, Germany; P.J. de Feyter, Rotterdam, The Netherlands; B. Güntert, Hall in Tirol, Austria; F. Gutzwiller, Bern, Switzerland; H. Heinen, Bonn, Germany; O. Hess, Bern, Switzerland; B. Klein, Essen, Germany; H. Löwel, Neuherberg, Germany; M. Reiser, Munich, Germany; G. Schmidt, Essen, Germany; M. Schwaiger, Munich, Germany; C. Steinmüller, Bonn, Germany; T. Theorell, Stockholm, Sweden; S.N. Willich, Berlin, Germany.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Author information
Authors and Affiliations
Consortia
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Fig. 1.
Scatter plot of the mean loge (CAC score + 1) and sex-specific deciles of fasting plasma glucose for men and women. Glucose group represents the fasting plasma glucose categorised into sex specific deciles. Diamonds, men; circles, women. Cutoff values for men in each glucose group are (mmol/l): 1, <5.03; 2, ≥5.03 to <5.42; 3, ≥5.42 to <5.63; 4, ≥5.63 to <5.80; 5, ≥5.80 to <5.93; 6, ≥5.93 to <6.10; 7, ≥ 6.10 to <6.29; 8, ≥6.29 to <6.58; 9, ≥6.58 to <7.11; 10, ≥9.56. For women the cutoff values for each glucose group are (mmol/l): 1, <4.79, 2, ≥4.79 to <5.14; 3, ≥5.14 to <5.33; 4, ≥5.33 to <5.47; 5, ≥5.47 to <5.61; 6, ≥5.61 to <5.76; 7, ≥ 5.76 to <5.93; 8, ≥5.93 to <6.15; 9, ≥6.15 to <6.51; 10, ≥8.05 (PDF 10.0 KB)
Rights and permissions
About this article
Cite this article
Moebus, S., Stang, A., Möhlenkamp, S. et al. Association of impaired fasting glucose and coronary artery calcification as a marker of subclinical atherosclerosis in a population-based cohort—results of the Heinz Nixdorf Recall Study. Diabetologia 52, 81–89 (2009). https://doi.org/10.1007/s00125-008-1173-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-008-1173-y