Abstract
Key message
Eight QTL for coleoptile length were identified in a genome-wide association study on a set of 893 wheat accessions, four of which are novel loci.
Abstract
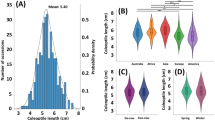
Wheat cultivars with long coleoptiles are preferred in wheat-growing regions where deep planting is practiced. However, the wide use of gibberellic acid (GA)-insensitive dwarfing genes, Rht-B1b and Rht-D1b, makes it challenging to breed dwarf wheat cultivars with long coleoptiles. To understand the genetic basis of coleoptile length, we performed a genome-wide association study on a set of 893 landraces and historical cultivars using 5011 single nucleotide polymorphism (SNP) markers. Structure analysis revealed four subgroups in the association panel. Association analysis results suggested that Rht-B1b and Rht-D1b genes significantly reduced coleoptile length, and eight additional quantitative trait loci (QTL) for coleoptile length were also identified. These QTL explained 1.45–3.18 and 1.36–3.11% of the phenotypic variation in 2015 and 2016, respectively, and their allelic substitution effects ranged from 0.31 to 1.75 cm in 2015, and 0.63–1.55 cm in 2016. Of the eight QTL, QCL.stars-1BS1, QCL.stars-2DS1, QCL.stars-4BS2, and QCL.stars-5BL1 are likely novel loci for coleoptile length. The favorable alleles in each accession ranged from two to eight with an average of 5.8 at eight loci in the panel, and more favorable alleles were significantly associated with longer coleoptile, suggesting that QTL pyramiding is an effective approach to increase wheat coleoptile length.





Similar content being viewed by others
Abbreviations
- FDR:
-
False discovery rate
- GWAS:
-
Genome-wide association study
- LD:
-
Linkage disequilibrium
- LnP(D):
-
Log probability of data
- MAGIC:
-
Multiparent advanced generation intercross
- MLM:
-
Mixed linear model
- NSGC:
-
National Small Grains Collection
- PCR:
-
Polymerase chain reaction
- QTL:
-
Quantitative trait loci
- SNP:
-
Single-nucleotide polymorphism
References
Addisu M, Snape JW, Simmonds JR, Gooding MJ (2009) Reduced height (Rht) and photoperiod insensitivity (Ppd) allele associations with establishment and early growth of wheat in contrasting production systems. Euphytica 166:249–267
Bai G, Das MK, Carver BF et al (2004) Covariation for microsatellite marker alleles associated with Rht8 and coleoptile length in winter wheat. Crop Sci 44:1187–1194
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265. doi:10.1093/bioinformatics/bth457
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B 57:289–300
Bonman JM, Babiker EM, Cuesta-Marcost A et al (2015) Genetic diversity among wheat accessions from the USDA National Small Grains Collection. Crop Sci 55:1243
Botwright TL, Rebetzke GJ, Condon AG, Richards RA (2005) Influence of the gibberellin-sensitive Rht8 dwarfing gene on leaf epidermal cell dimensions and early vigour in wheat (Triticum aestivum L.). Ann Bot 95:631–639
Bradbury PJ, Zhang Z, Kroon DE et al (2007) TASSEL: software for association mapping of complex traits in diverse samples. Bioinformatics 23:2633–2635. doi:10.1093/bioinformatics/btm308
Breseghello F, Sorrells ME (2006) Association mapping of kernel size and milling quality in wheat (Triticum aestivum L.) cultivars. Genetics 172:1165–1177. doi:10.1534/genetics.105.044586
Brown PR, Singleton GR, Tann CR, Mock I (2003) Increasing sowing depth to reduce mouse damage to winter crops. Crop Prot 22:653–660. doi:10.1016/S0261-2194(03)00006-1
Budak N, Baenziger PS, Eskridge KM et al (1995) Plant height response of semidwarf and nonsemidwarf wheats to the environment. Crop Sci 35:447–451
Chen W, Gao Y, Xie W et al (2014) Genome-wide association analyses provide genetic and biochemical insights into natural variation in rice metabolism. Nat Genet 46:714–721. doi:10.1038/ng.3007
Chia JM, Song C, Bradbury PJ et al (2012) Maize HapMap2 identifies extant variation from a genome in flux. Nat Genet 44:803–807. doi:10.1038/ng.2313
Dubcovsky J, Galvez AF, Dvořák J (1994) Comparison of the genetic organization of the early salt- stress-response gene system in salt-tolerant Lophopyrum elongatum and salt-sensitive wheat. Theor Appl Genet 87:957–964
Ellis M, Spielmeyer W, Gale K et al (2002) “ Perfect” markers for the Rht-B1b and Rht-D1b dwarfing genes in wheat. Theor Appl Genet 105(6–7):1038–1042
Ellis MH, Rebetzke GJ, Chandler P et al (2004) The effect of different height reducing genes on the early growth of wheat. Funct Plant Biol 31:583–589. doi:10.1071/fp03207
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Gale MD, Youssefian S (1985) Dwarfing genes in wheat. In: Russell GE (Ed). Progress in Plant Breeding, pp 1–35. Butterworths, London
Huang X, Zhao Y, Wei X et al (2012) Genome-wide association study of flowering time and grain yield traits in a worldwide collection of rice germplasm. Nat Genet 44:32–39
Kertho A, Mamidi S, Bonman JM et al (2015) Genome-wide association mapping for resistance to leaf and stripe rust in winter-habit hexaploid wheat landraces. PLoS One 10:e0129580. doi:10.1371/journal.pone.0129580
Keyes GJ, Paolillo DJ, Sorrells ME (1989) The effects of dwarfing genes Rht1 and Rht2 on cellular dimensions and rate of leaf elongation in wheat. Ann Bot 64:683–690
Korzun V, Roder MS, Ganal MW, Worland AJ, Law CN (1998) Genetic analysis of the dwarfing gene (Rht8) in wheat. Part I. Molecular mapping of Rht8 on the short arm of chromosome 2D of bread wheat (Triticum aestivum L.). Theor Appl Genet 96:1104–1109
Li P, Chen J, Wu P et al (2011) Quantitative trait loci analysis for the effect of dwarfing gene on coleoptile length and seedling root length and number of bread wheat. Crop Sci 51:2561–2568
Li G, Xu X, Bai G, Carver BF, Hunger R, Bonman M, Kolmer J, and Dong H (2016) Genome-wide association mapping reveals novel quantitative trait loci for leaf rust resistance in a worldwide collection of winter wheat. The Plant Genome (in press)
Maccaferri M, Sanguineti MC, Mantovani P et al (2010) Association mapping of leaf rust response in durum wheat. Mol Breed 26:189–228. doi:10.1007/s11032-009-9353-0
Maccaferri M, Zhang J, Bulli P, et al (2015) A genome-wide association study of resistance to stripe rust (Puccinia striiformis f. sp. tritici) in a worldwide collection of hexaploid spring wheat (Triticum aestivum L.). G3: Genes|Genomes|Genetics 5:449–465. doi: 10.1534/g3.114.014563
Murphy K, Balow K, Lyon SR, Jones SS (2008) Response to selection, combining ability and heritability of coleoptile length in winter wheat. Euphytica 164:709–718
O’Sullivan PA, Weiss GM, Friesen D (1985) Tolerance of spring wheat (Triticum aestivum L.) to trifluralin deep-incorporated in the autumn or spring. Weed Res 25:275–280
Peng J, Richards DE, Hartley NM, Murphy GP, Devos KM, Flintham JE, Beales J, Fish LJ, Worland AJ, Pelica F, Sudhakar D (1999) ‘Green revolution’genes encode mutant gibberellin response modulators. Nature 400(6741):256–261
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959. doi:10.1111/j.1471-8286.2007.01758.x
Rebetzke GJ, Richards RA, Fischer VM, Mickelson BJ (1999) Breeding long coleoptile, reduced height wheats. Euphytica 106:159–168. doi:10.1023/A:1003518920119
Rebetzke GJ, Appels R, Morrison AD et al (2001) Quantitative trait loci on chromosome 4B for coleoptile length and early vigour in wheat (Triticum aestivum L.). Aust J Agric Res 52:1221–1234. doi:10.1071/ar01042
Rebetzke GJ, Richards RA, Sirault XRR, Morrison AD (2004) Genetic analysis of coleoptile length and diameter in wheat. Aust J Agric Res 55:733–743
Rebetzke GJ, Richards RA, Fettell NA et al (2007) Genotypic increases in coleoptile length improves stand establishment, vigour and grain yield of deep-sown wheat. F Crop Res 100:10–23. doi:10.1016/j.fcr.2006.05.001
Rebetzke GJ, Verbyla AP, Verbyla KL et al (2014) Use of a large multiparent wheat mapping population in genomic dissection of coleoptile and seedling growth. Plant Biotechnol J 12:219–230. doi:10.1111/pbi.12130
Schillinger WF, Donaldson E, Allan RE, Jones SS (1998) Winter wheat seedling emergence from deep sowing depths. Agron J 90:582–586. doi:10.2134/agronj1998.00021962009000050002x
Spielmeyer W, Bonnett D, Ellis M et al (2001) Implementation of molecular markers to improve selection efficiency in CSIRO wheat breeding program. ‘Proceedings 10th Australian Wheat Breeders Assembly (Wheat Breeding Society of Australia)
Spielmeyer W, Hyles J, Joaquim P et al (2007) A QTL on chromosome 6A in bread wheat (Triticum aestivum) is associated with longer coleoptiles, greater seedling vigour and final plant height. Theor Appl Genet 115:59–66
Stockton RD, Krenzer EGJ, Solie J, Payton ME (1996) Stand establishment of winter wheat in Oklahoma: a survey. J Prod Agric 9:571–575
Sukumaran S, Dreisigacker S, Lopes M et al (2015) Genome-wide association study for grain yield and related traits in an elite spring wheat population grown in temperate irrigated environments. Theor Appl Genet 128:353–363. doi:10.1007/s00122-014-2435-3
Wang S, Wong D, Forrest K et al (2014) Characterization of polyploid wheat genomic diversity using a high-density 90,000 single nucleotide polymorphism array. Plant Biotechnol J 12:787–796. doi:10.1111/pbi.12183
Whan BR (1976a) The association between coleoptile length and culm length in semidwarf and standard wheats. Journal of the Australian Institute of Agricultural Science
Whan BR (1976b) The emergence of semidwarf and standard wheats, and its association with coleoptile length. Aust J Exp Agric 16:411–416
Worland T, Snape JW. 2001. Genetic analysis of worldwide wheat varietal improvement. P. – 100. In A.P. Bonjean and W.J. Angus (ed.) The world wheat book: A history of wheat breeding. Lavoisier Publishing, Paris
Worland AJ, Korzun V, Röder MS et al (1998) Genetic analysis of the dwarfing gene Rht8 in wheat. Part II. The distribution and adaptive significance of allelic variants at the Rht8 locus of wheat as revealed by microsatellite screening. Theor Appl Genet 96:1110–1120
Wright STC (1966) Growth and cellular differentiation in the wheat coleoptile (Triticum vulgare): II. Factors influencing the growth response to gibberellic acid, kinetin and indolyl-3-acetic acid. J Exp Bot 17:165–176
Yu JB, Bai GH (2010) Mapping quantitative trait loci for long coleoptile in Chinese wheat landrace Wangshuibai. Crop Sci 50:43–50. doi:10.2135/cropsci2009.02.0065
Yu J, Pressoir G, Briggs WH et al (2006) A unified mixed-model method for association mapping that accounts for multiple levels of relatedness. Nat Genet 38:203–208. doi:10.1038/ng1702
Yu LX, Morgounov A, Wanyera R et al (2012) Identification of Ug99 stem rust resistance loci in winter wheat germplasm using genome-wide association analysis. Theor Appl Genet 125:749–758. doi:10.1007/s00122-012-1867-x
Zanke CD, Ling J, Plieske J, Kollers S, Ebmeyer E, Korzun V, Argillier O, Stiewe G, Hinze M, Neumann K, Ganal MW (2014) Whole genome association mapping of plant height in winter wheat (Triticum aestivum L.). PLoS One 9(11):e113287
Zhang D, Bai G, Hunger RM et al (2011) Association study of resistance to Soilborne wheat mosaic virus in U.S. winter wheat. Phytopathology 101:1322–1329. doi:10.1094/PHYTO-02-11-0041
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by S. Dreisigacker.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, G., Bai, G., Carver, B.F. et al. Genome-wide association study reveals genetic architecture of coleoptile length in wheat. Theor Appl Genet 130, 391–401 (2017). https://doi.org/10.1007/s00122-016-2820-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-016-2820-1