Abstract
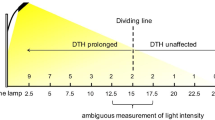
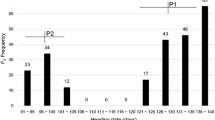
A late heading-time mutant line, HS276, which was induced by gamma-irradiation of seeds of the japonica rice (Oryza sativa L.) variety Gimbozu, exhibits an extremely long basic vegetative growth phase (BVP). A genetic analysis using the F2 population from the cross between HS276 and Gimbozu revealed that the late heading of HS276 is governed by a single recessive mutant gene. The subsequent analysis on heading responses of HS276 and Gimbozu to four photoperiods (12, 13, 14, and 15 h) and to the photoperiodic transfer treatment from a short photoperiod to a long photoperiod revealed that the mutant gene confers an extremely long BVP and increases photoperiod sensitivity under long photoperiod (14 and 15 h). The BVP durations of HS276 and Gimbozu were estimated at 30.1 and 16.0 days, respectively; the mutant gene, compared with its wild type allele, elongates the duration of BVP by 14 days. Linkage analysis showed that the mutant gene is located in the 129 kb region between the two INDEL markers, INDELAP0399_6 and INDELAP3487_2, on the distal part of the short arm of chromosome 6. None of the other BVP genes are located in this region; therefore, we declared this a newly detected mutant gene and designated it ef7. A recently established program to breed rice suitable for low latitudes, where short photoperiodic conditions continue throughout the year, aims to develop varieties with extremely long BVPs and weak photoperiod sensitivities; the mutant gene ef7, therefore, will be quite useful in these programs because it confers an extremely long BVP and little enhances photoperiod sensitivity under short photoperiod.





Similar content being viewed by others
References
Akagi H, Yokozeki Y, Inagaki A, Fujimura T (1997) Highly polymorphic microsatellites of rice consist of AT repeats, and a classification of closely related cultivars with these microsatellite loci. Theor Appl Genet 94:61–67
Baurle I, Dean C (2006) The timing of developmental transitions in plants. Cell 125:655–664
Chang TT, Vergara BS (1969) Component analysis of duration from seeding to heading in rice by the basic vegetative phase and the photoperiod-sensitive phase. Euphytica 18:79–91
Doi K, Yoshimura A (1998) RFLP mapping of a gene for heading date in an African rice. Rice Genet Newslett 15:148–149
Doi K, Izawa T, Fuse T, Yamanouchi U, Kubo T, Shimatani Z, Yano M, Yoshimura A (2004) Ehd1, a B-type response regulator in rice, confers short-day promotion of flowering and controls FT-like gene expression independently of Hd1. Genes Dev 18:926–936
Ellis RH, Collinson ST, Hudson D, Patefield WM (1992) The analysis of reciprocal transfer experiments to estimate the durations of the photoperiod-sensitive and photoperiod-insensitive phases of plant development: an example in soya bean. Ann Bot 70:87–92
Hayama R, Coupland G (2003) Shedding light on the circadian clock and the photoperiodic control of flowering. Curr Opin Plant Biol 6:13–19
Hosoi N (1981) Studies on meteorological fluctuation in the growth of rice plants. V. Regional differences of thermo-sensitivity, photo-sensitivity, basic vegetative growth and factors determining the growth duration of Japanese varieties. Jpn J Breed 31:239–250
Ichitani K, Okumoto Y, Tanisaka T (1998) Genetic analyses of low photoperiod sensitivity of rice cultivars from the northernmost regions of Japan. Plant Breed 117:543–547
Izawa T, Oikawa T, Sugiyama N, Tanisaka T, Yano M, Shimamoto K (2002) Phytochrome mediates the external light signal to repress FT orthologs in photoperiodic flowering of rice. Genes Dev 16:2006–2020
Khun LH, Hiraiwa M, Sato S, Motomura K, Murayama S, Adaniya S, Nose A, Ishimine Y (2004) Location of new gene for late heading in rice, Oryza sativa L. Using interchange homozygotes. Breed Sci 54:259–263
Khun LH, Motomura K, Murayama S, Adaniya S, Nose A (2005a) Linkage analysis of a lateness gene ef4 in rice, Oryza sativa L. Breed Sci 55:231–235
Khun LH, Motomura K, Murayama S, Adaniya S, Nose A (2005b) Trisomic analysis of a lateness gene ef2 in rice, Oryza sativa L. Breed Sci 55:35–39
Khun LH, Miyaji S, Motomura K, Murayama S, Adaniya S, Nose A (2006) Trisomic analysis of new gene for late heading in rice, Oryza sativa L. Euphytica 151:235–241
Komiya R, Ikegami A, Tamaki S, Yokoi S, Shimamoto K (2008) Hd3a and RFT1 are essential for flowering in rice. Development 135:767–774
Koornneef M, Peeters A (1997) Floral transition mutants in Arabidopsis. Plant Cell Environ 20:779–784
Kurata N, Nagamura Y, Yamamoto K, Harushima Y, Sue N, Wu J, Antonio BA, Shomura A, Shimizu T, Lin SY, Inoue T, Fukuda A, Shimano T, Kuboki Y, Toyama T, Miyamoto Y, Kirihara T, Hayasaka K, Miyao A, Monna L, Zhong HS, Tamura Y, Wang ZX, Momma T, Umehara Y, Yano M, Sasaki T, Minobe Y (1994) A 300 kilobase interval genetic map of rice including 883 expressed sequences. Nat Genet 8:365–372
Lee S, Kim J, Han J-J, Han M-J, An G (2004) Functional analyses of the flowering time gene OsMADS50, the putative SUPPRESSOR OF OVEREXPRESSION OF CO 1/AGAMOUS-LIKE 20 (SOC1/AGL20) ortholog in rice. Plant J 38:754–764
Matsubara K, Yamanouchi U, Wang ZX, Minobe Y, Izawa T, Yano M (2008) Ehd2, a rice ortholog of the maize ID1 gene, promotes flowering by upregulating Ehd1. Plant Physiol. doi: 10.1104/pp.108.125542
McCouch SR, Kochert G, Yu ZH, Wang ZY, Khush GS, Coffman WR, Tanksley SD (1988) Molecular mapping of rice chromosomes. Theor Appl Genet 76:815–829
Monden Y, Naito K, Okumoto Y, Saito H, Oki N, Tsukiyama T, Ideta O, Nakazaki T, Wessler SR, Tanisaka T (2009) High potential of a transposon mPing as a marker system in japonica × japonica cross in rice. DNA Res. doi:10.1093/dnares/dsp004
Monna L, Lin H, Kojima S, Sasaki T, Yano M (2002) Genetic dissection of a genomic region for a quantitative trait locus, Hd3, into two loci, Hd3a and Hd3b, controlling heading date in rice. Theor Appl Genet 104:772–778
Nishida H, Okumoto Y, Nakagawa H, Ichitani K, Inoue H, Tanisaka T (2001) Analysis of tester lines for rice (Oryza sativa L.) heading-time genes using reciprocal photoperiodic transfer treatments. Ann Bot 88:527–536
Nishida H, Inoue H, Okumoto Y, Tanisaka T (2002) A novel gene ef1-h conferring an extremely long basic vegetative growth period in rice. Crop Sci 42:348–354
Okumoto Y, Tanisaka T (1997) Trisomic analysis of a strong photoperiod-sensitivity gene E1 in rice (Oryza sativa L.). Euphytica 95:301–307
Park SJ, Kim SL, Lee S, Je BI, Piao HL, Park SH, Kim CM, Ryu CH, Park SH, Xuan YH, Colasanti J, An G, Han CD (2008) Rice Indeterminate 1 (OsId1) is necessary for the expression of Ehd1 (Early heading date 1) regardless of photoperiod. Plant J 56:1018–1029
Poonyarit M, Mackill DJ, Vergara BS (1989) Genetics of photoperiod sensitivity and critical daylength in rice. Crop Sci 29:647–652
Reeves PH, Coupland G (2001) Analysis of flowering time control in Arabidopsis by comparison of double and triple mutants. Plant Physiol 126:1085–1091
Saito H, Yuan Q, Okumoto Y, Doi K, Yoshimura A, Inoue H, Teraishi M, Tsukiyama T, Tanisaka T (2009) Multiple alleles at Early flowering 1 locus making variation in the basic vegetative growth period in rice (Oryza sativa L.). Theor Appl Genet. doi:10.1007/s00122-009-1040-3
Tamaki S, Matsuo S, Wong HL, Yokoi S, Shimamoto K (2007) Hd3a protein is a mobile flowering signal in rice. Science 316:1033–1036
Tanisaka T, Inoue H, Uozu S, Yamagata H (1992) Basic vegetative growth and photoperiod sensitivity of heading-time mutants induced in rice. Jpn J Breed 42:657–668
Tsai KH (1986) Genes controlling heading time found in a tropical Japonica variety. Rice Genet Newslett 3:71–73
Tsai KH (1991) Genes for late-heading and their interaction in the background of Taichung 65. Rice genetics II. Intern Rice Res Inst, Manila, pp 211–215
Wang S, Basten CJ, Zeng Z-B (2005) Windows QTL Cartographer 2.5. North Carolina State University, Raleigh. http://www.statgen.ncsu.edu/qtlcart/WQTLCart.html
Wu C, You C, Li C, Long T, Chen G, Byrne ME, Zhang Q (2008) RID1, encoding a Cys2/His2-type zinc finger transcription factor, acts as a master switch from vegetative to floral development in rice. PNAS 105(35):12915–12920
Xue W, Xing Y, Weng X, Zhao Y, Tang W, Wang L, Zhou H, Yu S, Xu C, Li X, Zhang Q (2008) Natural variation in Ghd7 is an important regulator of heading date and yield potential in rice. Nat Genet 40:761–767
Yamamoto T, Lin H, Sasaki T, Yano M (2000) Identification of heading date quantitative trait locus Hd6 and characterization of its epistatic interactions with Hd2 in rice using advanced backcross progeny. Genetics 154:885–891
Yano M, Ebitani T (2002) Development of a series of chromosome segment substitution lines and their utilization in the genetic analysis of quantitative traits in rice. NIAS Ann Rep 27–28
Yano M, Harushima Y, Nagamura Y, Kurata N, Minobe Y, Sasaki T (1997) Identification of quantitative trait loci controlling heading date in rice using a high-density linkage map. Theor Appl Genet 95:1025–1032
Yano M, Katayose Y, Ashikari M, Yamanouchi U, Monna L, Fuse T, Baba T, Yamamoto K, Umehara Y, Nagamura Y, Sasaki T (2000) Hd1, a major photoperiod sensitivity quantitative trait locus in rice, Is closely related to the Arabidopsis flowering time gene CONSTANS. Plant Cell 12:2473–2484
Yin X, Kropff MJ, Ynalvez MA (1997) Photoperiodically sensitive and insensitive phases of preflowering development in rice. Crop Sci 37:182–190
Yokoo M, Kikuchi F (1992) The Lm locus controlling photoperiod sensitivity in rice differs from E1, E2 and E3 loci. Jpn J Breed 42:375–381 (In Japanese, with English abstract)
Acknowledgments
We thank the Rice Genome Resource Center (RGRC) (Tsukuba, Japan) for providing the seeds of chromosome segment substitution lines. This work was supported by a grant from the Ministry of Agriculture, Forestry, and Fisheries of Japan (Integrated research project for plant, insect and animal using genome technology QTL-4001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Snape.
Rights and permissions
About this article
Cite this article
Yuan, Q., Saito, H., Okumoto, Y. et al. Identification of a novel gene ef7 conferring an extremely long basic vegetative growth phase in rice. Theor Appl Genet 119, 675–684 (2009). https://doi.org/10.1007/s00122-009-1078-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-009-1078-2