Abstract
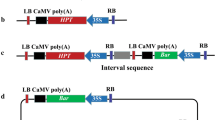
A negative selectable marker gene, codA, was successfully co-transformed with a GUS reporter gene to develop selectable marker gene-free transgenic plants. The pNC binary vector contained a T-DNA harboring the codA gene next to the nptII gene, while a second binary vector, pHG, contained a GUS reporter gene. Tobacco plants (Nicotiana tabacum cv. Samsun NN) were co-transformed via the mixture method with Agrobacterium tumefaciens LBA4404 strains harboring pNC and pHG, respectively. Seeds harvested from the co-transformants were sown on germination media containing 5-fluorocytosine (5-FC). Analysis of the progeny by GUS staining and PCR amplification revealed that all of the 5-FC-resistant R1 plants were codA free, and that the codA gene segregated independently of the GUS gene. Because codA-free seedlings developed normally on 5-FC-containing medium, we suggest that co-transformation with negatively selectable markers is a viable method for the production of easily distinguished, selectable marker gene-free transgenic plants.





Similar content being viewed by others
References
An G, Evert PR, Mitra A, Ha SB (1988) Plant molecular biology manual. Kluwer, Dordrecht
Chen H, Nelson RS, Sherwood JL (1994) Enhanced recovery of transformants of Agrobacterium tumefaciens after freeze-thaw transformation and drug selection. Biotechniques 16:664–669
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: version II. Plant Mol Biol Rep 1(4):19–21
Depicker A, Herman L, Jacobs A, Schell J, Montagu MV (1985) Frequencies of simultaneous transformation with different T-DNAs and their relevance to the Agrobacterium/plant cell interaction. Mol Gen Genet 201:477–484
Eklof S, Astot C, Sitbon F, Moritz T, Olsson O, Sanberg G (2000) Transgenic tobacco plants co-expressing Agrobacterium iaa and ipt genes have wild-type hormone levels but display both auxin- and cytokinin-overproducing phenotypes. Plant J 23:279–284
Endo S, Sugita K, Sakai M, Tanaka H, Ebinuma H (2002) Single-step transformation for generating marker-free transgenic rice using the ipt-type MAT vector system. Plant J 30:115–122
Framond AJD, Back EW, Chilton WS, Kayes L, Chilton M (1986) Two unlinked T-DNAs can transform the same tobacco plant cell and segregate in the F1 generation. Mol Gen Genet 202:125–131
Gleave AP, Mitra DS, Mudge SR, Morris BAM (1999) Selectable marker-free transgenic plants without sexual crossing: transient expression of cre recombinase and use of a conditional lethal dominant gene. Plant Mol Biol 40:223–235
Goldsbrough AP, Lastrella CN, Yoder JI (1993) Transposition mediated re-positioning and subsequent elimination of marker genes from transgenic tomato. Biotechnology 11:1286–1292
Horsch RB, Fry JE, Hoffman NL, Eichholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Jefferson RA, Kavanagh TA, Bevan MW (1987) β-Glucuronidase as a sensitive and versatile fusion marker in higher plants. EMBO J 6:3901–3907
Karlin-Neumann GA, Brusslan JA, Tobin EM (1991) Phytochrome control of the tms2 gene in transgenic Arabidopsis: a strategy for selecting mutants in the signal transduction pathway. Plant Cell 3:573–582
Komari T, Hiei Y, Saito Y, Murai N (1996) Vectors carrying two separate T-DNAs for co-transformation of higher plants mediated by Agrobacterium tumefaciens and segregation of transformants free from selection markers. Plant J 10:165–174
Koprek T, McElroy D, Louwerse J,Willams-Carrier R, Lemaux PG (1996) Negative selection systems for transgenic barley(Hordeum vulgare L.): comparison of bacterial codA- and cytochrome P450 gene-mediated selection. Plant J 19:719–726
McCormac AC, Fowler MR, Chen D, Elliot MC (2001) Efficient co-transformation of Nicotiana tabacum by two independent T-DNA, the effect of T-DNA size and implications for genetic separation. Transgenic Res 10:143–155
McKnight TD, Lillis MT, Simpson RB (1987) Segregation of genes transferred to one plant cell from two separate Agrobacterium strains. Plant Mol Biol 8:439–445
O’keefe DP, Tepperman JM, Dean C, Leto KJ, Erbes DL, Odell JT (1994) Plant expression of a bacterial cytochrome P450 that catalyzes activation of a sulfonylurea pro-herbicide. Plant Physiol 105:473–482
Petit A, Berkaloff A, Tempe J (1985) Multiple transformation of plant cells by Agrobacterium may be responsible for the complex organization of T-DNA in crown gall and hairy root. Mol Gen Genet 202:388–393
Puchta H (2000) Removing selectable marker genes: taking the shortcut. Trends Plant Sci 5:273–274
Sambrook J, Frisch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schlaman HRM, Hooykaas PJJ (1997) Effectiveness of the bacterial gene codA encoding cytosine deaminase as a negative selectable marker in Agrobacterium-mediated plant transformation. Plant J 11:1377–1385
Vain P, Afolabi AS, Worland B, Snape JW (2003) Transgene behaviour in populations of rice plants transformed using a new dual binary vector system: pGreen/pSoup. Theor Appl Genet 107:210–217
Werck-Reichhart D, Hehn A, Didierjean L (2000) Cytochromes P450 for engineering herbicide tolerance. Trends Plant Sci 5:116–123
Xiang C, Han P, Lutziger I, Wang K, Oliver DJ (1999) A mini binary vector series for plant transformation. Plant Mol Biol 40:711–717
Acknowledgements
This study was supported by a grant from BioGreen21 project from Rural Development Administration and CFGC project from Ministry of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Möllers
Rights and permissions
About this article
Cite this article
Park, J., Lee, Y.K., Kang, B.K. et al. Co-transformation using a negative selectable marker gene for the production of selectable marker gene-free transgenic plants. Theor Appl Genet 109, 1562–1567 (2004). https://doi.org/10.1007/s00122-004-1790-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-004-1790-x