Abstract
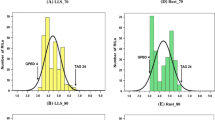
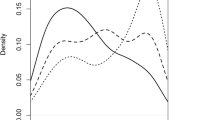
Resistance to Ascochyta blight of pea was genetically characterized by mapping quantitative trait loci (QTLs) using two crosses, 3147-A26 (A26, partially resistant) × cultivar Rovar (susceptible) and 3148-A88 (A88, partially resistant) × Rovar, with the aim of developing an increased understanding of the genetics of resistance and of identifying linked molecular markers that may be used to develop resistant germplasm. Molecular linkage maps for both crosses were aligned so that the results of QTL mapping could be compared. Ascochyta blight disease severity in response to natural epidemics was measured in field trials conducted in Western Australia and New Zealand. Eleven putative QTLs for Ascochyta blight resistance were identified from the A26 × Rovar population and 14 putative QTLs from the A88 × Rovar population. Six QTLs were associated with the same genomic regions in both populations. These QTLs reside on linkage groups II, III, IV, V, and VII (two QTLs). The severity of Ascochyta blight disease symptoms on pea increases during field epidemics as plants mature; therefore, QTLs for plant reproductive maturity were mapped. Six QTLs were detected for plant maturity in the A26 × Rovar population, while five plant maturity QTLs were mapped in the A88 × Rovar population. QTLs for plant maturity coincide with Ascochyta blight resistance QTLs in four genomic regions, on linkage groups II (two regions), III, and V. The plant maturity and Ascochyta blight resistance QTLs on III were linked in repulsion phase. Therefore, the coincidence of these QTLs may be explained by linkage of distinct loci for the two traits. The QTLs on linkage groups II and V were linked in coupling phase; therefore, linked QTLs for resistance and maturity may be present in these regions, or the Ascochyta blight resistance QTLs detected in these regions are the result of pleiotropic effects of plant-maturity genetic loci.


Similar content being viewed by others
References
Baggett JR, Hampton RO (1977) Oregon B-442-15 and B445-66 pea seedborne mosaic virus-resistant breeding lines. Hort Sci 12:506
Beasse CB, Ney B, Tivoli B (1999) Effects of pod infection by Mycosphaerella pinodes on yield components of pea (Pisum sativum). Ann Appl Biol 135:359–367
Beavis WD (1994) The power and deceit of QTL experiments: lessons from comparative QTL studies. In: 49th Annual Corn and Sorghum Industry Research Conference. American Seed Trade Association, Washington, pp 250–266
Bretag TW, Ramsey M (2001) Foliar diseases caused by fungi: Ascochyta spp. In: Kraft JM, Pfleger PM (eds) Pea disease compendium. American Pathological Society, St. Paul, pp 24–28
Churchill GA, Doerge RW (1994) Empirical threshold values for quantitative trait mapping. Genetics 138:963–971
Clulow SA, Matthews P, Lewis BG (1991) A pathotype classification of Mycosphaerella pinodes. J Phytopathol 131:322–332
Cullis BR, Lill WJ, Fisher JA, Read BJ, Gleeson AC (1989) A new procedure for the analysis of early generation variety trials. Appl Stat 38:361–375
Dirlewanger E, Isaac P, Ranade S, Belajouza M, Cousin R, Devienne D (1994) Restriction fragment length polymorphism analysis of loci associated with disease resistance genes and developmental traits in Pisum sativum L. Theor Appl Genet 88:17–27
Dudley JW (1993) Molecular markers in plant improvement: manipulation of genes affecting quantitative traits. Crop Sci 33:660–668
Frew TJ, Russell AC, Timmerman-Vaughan GM (2002) Sequence tagged site markers linked to the sbm1 gene for resistance to pea seedborne mosaic virus in pea. Plant Breed 121:512–516
Genstat Committee (1997) Genstat 5, release 4.1, reference manual supplement. Numerical Algorithms Group, Oxford
Genstat Committee (2002) Genstat, release 6.1, reference manual, Parts 1–3. VSN International, Oxford
Gervais L, Dedryver F, Morlais JY, Bodusseau V, Negre S, Bilous M, Groos C, Trottet M (2003) Mapping of quantitative trait loci for field resistance to Fusarium head blight in an European winter wheat. Theor Appl Genet 106:961–970
Gilmour AR, Cullis BR, Verbyla AP (1997) Accounting for natural and extraneous variation in the analysis of field experiments. J Agric Biol Environ Stat 2:269–273
Gilpin BJ, McCallum JA, Frew TJ, Timmerman-Vaughan GM (1997) A linkage map of the pea (Pisum sativum L.) genome containing cloned sequences of known function and expressed sequence tags (ESTs). Theor Appl Genet 95:1289–1299
Kempton RA, Gleeson AC (1997) Unreplicated trials. In: Kempton RA, Fox PN (eds) Statistical methods for plant variety evaluation. Chapman and Hall, London, pp 86–100
Knott CM (1987) A key for stages of development of the pea. Ann Appl Biol 111:233–244
Kraft JM, Dunne B, Goulden D, Armstrong S (1998) A search for resistance in peas to Mycosphaerella pinodes. Plant Dis 82:251–253
Lincoln S, Daly M, Lander E (1992) Constructing genetic maps with MAPMAKER/EXP 3.0. Whitehead Institute Technical Report, 3rd edn. Whitehead Institute, Cambridge
Ma ZQ, Steffenson BJ, Prom LK, Lapitan NLV, Ma ZQ (2000) Mapping of quantitative trait loci for fusarium head blight resistance in barley. Phytopath 90:1079–1088
McCallum J, Leite D, Pither-Joyce M, Havey MJ (2001) Expressed sequence markers for genetic analysis of bulb onion (Allium cepa L.). Theor Appl Genet 103:979–991
Melchinger AE, Utz HF, Schon CC (1998) Quantitative trait locus (QTL) mapping using different testers and independent population samples in maize reveals low power of QTL detection and large bias in estimates of QTL effects. Genetics 149:383–403
Mesterhazy A (1995) Types and components of resistance to Fusarium head blight of wheat. Plant Breed 114:377–386
Moreau L, Monod H, Charcosset A, Gallais A (1999) Marker-assisted selection with spatial analysis of unreplicated field trials. Theor Appl Genet 98:234–242
Murfet IC, Taylor SA (1999) Flowering gene ppd in pea: map position and disturbed segregation of allele ppd-2. J Hered 90:548–550
Onfroy C, Tivoli B, Coubiere R, Bouznad Z (1999) Cultural, molecular and pathogenic variability of Mycosphaerella pinodes and Phoma medicaginis var. pinodella isolates from dried pea (Pisum sativum) in France. Plant Pathol 48:218–229
Ooijen JW van (1992) Accuracy of mapping quantitative trait loci in autogamous species. Theor Appl Genet 84:803–811
Patterson HD, Thompson R (1971) Recovery of inter-block information when block sizes are unequal. Biometrika 58:545–554
de la Pena RC, Smith KP, Capettinin GJ, Muehlbauer GJ, Gallo-Meagher M, Dilly-Macky R, Somers DA, Rasmusson DC (1999) Quantitative trait loci associated with resistance to Fusarium head blight and kernel discoloration in barley. Theor Appl Genet 99:562–569
Timmerman-Vaughan GM, Frew TJ, Weeden NF (2000) Characterization and linkage mapping of R-gene analogous DNA sequences in pea (Pisum sativum L.). Theor Appl Genet 101:241–247
Timmerman-Vaughan GM, Frew TJ, Russell AC, Khan T, Butler R, Gilpin M, Murray S, Falloon K (2002) QTL mapping of partial resistance to field epidemics of Ascochyta blight of peas. Crop Sci 42:2100–2111
Utz HF, Melchinger AE (1994) Comparison of different approaches to interval mapping of quantitative trait loci. In: Van Ooijen JW, Jansen J (eds) Biometrics in plant breeding: applications of molecular markers, proceedings of the ninth meeting of the EUCARPIA section biometrics in plant breeding. CPRO-DLO, Wageningen, Netherlands, pp 195–204
Verbyla AP, Cullis BR, Kenward MG, Welham SJ (1999) The analysis of designed experiments and longitudinal experiments by using smoothing splines. J R Stat Soc Ser C (Appl Stat) 48:269–311
Visker MHPW, Keizer LCP, Van Eck HJ, Jacobsen E, Colon LT, Struik PC (2003) Can the QTL for late blight resistance on potato chromosome 5 be attributed to foliage maturity type? Theor Appl Genet 106:317–325
Visscher PM, Thompson R, Haley CS (1996) Confidence intervals in QTL mapping by bootstrapping. Genetics 143:1013–1020
Wang S, Basten CJ, Zeng Z-B (2002) QTL Cartographer, version 2.0. Statistical Genetics, North Carolina State University, Raleigh
Weeden NF, Swiecicki WK, Timmerman G, Ambrose M (1993) Guidelines for future mapping studies in Pisum. Pisum Genet 25:13–14
Weeden NF, Ellis THN, Timmerman-Vaughan GM, Sweicicki WK, Rozok SM, Bernikov VA (1998) A consensus linkage map for Pisum sativum. Pisum Genet 30:1–4
Weller JL, Reid JB, Taylor SA, Murfet IC (1997) The genetic control of flowering in pea. Trends Plant Sci 2:412–418
Wroth J (1998) Possible role for wild genotypes of Pisum spp. to enhance Ascochyta blight resistance in pea. Aust J Exp Agric 38:469–479
Wroth J (1999) Evidence suggests that Mycosphaerella pinodes infection of Pisum sativum is inherited as a quantitative trait. Euphytica 107:193–204
Xue AG, Warkentin TD, Gossen BD, Burnett PA, Vandenberg A, Rashid KY (1998) Pathogenic variation of western Canadian isolates of Mycosphaerella pinodes on selected Pisum genotypes. Can J Plant Pathol 20:189–193
Zhu H, Gilchrist L, Hayes P, Kleinhofs A, Kudrna D, Liu Z, Prom L, Steffenson B, Toojinda T, Vivar H (1999) Does function follow form? Principal QTLs for Fusarium head blight (FHB) resistance are coincident with QTLs for inflorescence traits and plant height in a doubled-haploid population of barley. Theor Appl Genet 99:1221–1232
Acknowledgements
The authors would like to thank Jeanne Jacobs and Tracy Williams for their critical reading of the manuscript and Stewart Armstrong and Robert Saunders (Southland) and Alan Harris (Western Australia) for management of field trials. This research was funded by the New Zealand Foundation for Research, Science and Technology and by the Australian Grains Research and Development Corporation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Langridge
Rights and permissions
About this article
Cite this article
Timmerman-Vaughan, G.M., Frew, T.J., Butler, R. et al. Validation of quantitative trait loci for Ascochyta blight resistance in pea (Pisum sativum L.), using populations from two crosses. Theor Appl Genet 109, 1620–1631 (2004). https://doi.org/10.1007/s00122-004-1779-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-004-1779-5